|
On the 26-31st March, the 14th International Conference on Alzheimer’s and Parkinson’s Diseases (or ADPD meeting) was held in Lisbon, Portugal. For 5 days – between 8:30am and 7:30pm each day – over 4000 researchers were able to attend lectures of new results and ideas, in any of 8 different auditoriums. Alternatively, they could wander among hundreds of research posters. It was a marathon effort, however, for all attendees. And a great deal of new results were shared. In today’s post, we will discussed what was presented at the 2019 ADPD meeting and what was actually learnt.
|
 Lisbon. Source: stmed
Lisbon. Source: stmed
Lisbon is a city, midway down the western coast of the Iberian Peninsula.
It is home to a little over 500,000 people (3 million in the wider metropolitan area), and it serves as the capital city for the Portuguese people.
 The Castelo de Sao Jorge, rises above Lisbon. Source: Wikipedia
The Castelo de Sao Jorge, rises above Lisbon. Source: Wikipedia
Interestingly, it is the 2nd oldest European capital city (after Athens), and has had a rich and fascinating history given its strategic location. But on the 1st November 1755, 20% of the population were killed and 85% of the city’s structures were destroyed by a terrible earthquake and subsequent tsunami, which resulted in the vast majority of the city being rebuilt.
The ‘new city’ is laid out in bairros de Lisboa (neighbourhoods of Lisbon) across a hilly landscape, providing views of the River Tagus at every vantage point. And while walking the steep cobblestoned streets is delightful, there is a system of vintage public trams that can take a lot of the leg work out of the effort.
 Source: Portugaltravelguide
Source: Portugaltravelguide
During the last week of March 2019, Lisbon was the site of the ADPD meeting.
What is the ADPD meeting?
The Alzheimer’s and Parkinson’s Diseases meeting is an annual international conference focused on the two most common neurodegenerative conditions and related neurological disorders. It is one of the big conferences of the year for researchers in the Alzheimer’s or Parkinson’s field.
And when I say big, I mean BIG!
It is a five day meeting, with each day starting around 8:30am and finishing at 7:30pm.
This year approximately 4000 researchers and clinicians from 73 countries around the world gathered in Lisbon to present and discuss new findings and clinical trial results.
The ADPD 2019 meeting was held at the Centro de Congressos de Lisboa:
There were more than 2200 scientific abstracts accepted by the ADPD organising committee, and the lecture sessions were spread between the 8 auditoriums that the conference centre houses.
Some of those lecture rooms were huge, with audience capacity for 1500 individuals:
While other auditoriums were some what smaller (and cosier).
In addition, there were hundreds of poster presentations being made across the week in a large, open display hall:
This is what a research conference looks like.
Was there any research of particular interest presented at the meeting?
Yes, there were a lot of really interesting presentations.
Some of the major take aways from the meeting included:
1. The Phase II Ambroxol headline results:
Ambroxol is a respiratory medication that has been found to have beneficial properties in models of Parkinson’s. Ambroxol promotes the clearance of mucus and eases coughing. It also has anti-inflammatory properties, reducing redness in a sore throat.
But there is evidence that this drug may be able to help with Parkinson’s.

Ambroxol. Source: Skinflint
Researchers believe that Ambroxol could be useful with Parkinson’s in two ways:
1. Ambroxol is believed to triggers exocytosis of lysosomes (Source). Exocytosis is the process by which waste is exported out of the cell. Lysosomes are the bags of digestive that rubbish and waste is put into inside a cell for recycling. By encouraging lysosomes to undergo exocytosis and spit their waste contents out of the cell – digested or not – Ambroxol helps the cell to remove waste efficiently. Less waste building up results in a healthier cell.

Exocytosis. Source: Socratic
2. Ambroxol has also been shown to increase levels of an enzyme called glucocerebrosidase (or GCase) in the brain (Source). GCase is involved with the breaking down of proteins in the lysosome. GCase is generated from instructions provided by a gene called GBA1. Mutations in this gene are one of the most common genetic risk factors for Parkinson’s (Click here to read a SoPD post on this topic).
So by administering Ambroxol to people with Parkinson’s, researchers are hoping to raise levels of GCase to help digest proteins and increase the excretion of this waste from cells. This would ideally keep cells healthier for longer and slow down the progression of Parkinson’s.
The “Ambroxol in Disease Modification in Parkinson Disease” (or AiM-PD) clinical trial was set up to test this idea (Click here to read more about the details of this study). The study was supported by the Cure Parkinson’s Trust (UK), the Van Andel Research Institute (USA) and the John Black Charitable Foundation.
The results look very encouraging, with the treatment being very well tolerated and accessing the brain. A larger study is now being proposed. Once the results are published in a month or so, you can expect a full report on the SoPD website.
2. The safety data from the ongoing Herantis Pharma Phase I clinical trial of CDNF
There is an on-going clinical trial in Finland and Sweden testing a compound called CDNF in people with Parkinson’s. The study is being conducted by a biotech company called Herantis Pharma.
CDNF (or Cerebral Dopamine Neurotrophic Factor) is a naturally produced protein – a neurotrophic factor – that the brain makes to support dopamine cells.
Neurotrophic factors (neurotrophic = Greek: neuron – nerve; trophikós – pertaining to food/to feed) are chemicals that nurture neurons and support growth. There are many types of neurotrophic factors, some having more beneficial effects on certain types of neurons and not other. GDNF (or glial cell line-derived neurotrophic factor) which has recently been tested in Parkinson’s is also one of these neurotrophic factors (Click here to read a recent SoPD post on this topic).
Herantis Pharma has produced this video explaining CDNF:
For readers interested in a bit more detail as to how CDNF work, I can recommend this recent review article from researchers involved with Herantis:
 Title: CDNF Protein Therapy in Parkinson’s Disease.
Title: CDNF Protein Therapy in Parkinson’s Disease.
Authors: Huttunen HJ, Saarma M.
Journal: Cell Transplant. [Epub ahead of print]
PMID: 30947516 (This report is OPEN ACCESS if you would like to read it)
In this review, the researchers provide a very thorough explanation of how CDNF has its beneficial effect (by reducing endoplasmic reticulum (ER) stress):
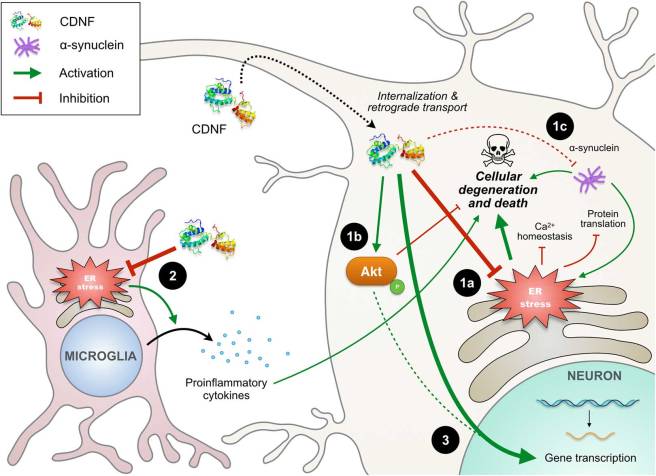 The actions of CDNF. Source: Sage
The actions of CDNF. Source: Sage
The review also provide a very timely review of the previous clinical trials of neurotrophic factors in Parkinson’s.
In the current CDNF trial, the research team at Herantis Pharma are using the same delivery system that was used in the Cure Parkinson’s/Parkinson’s UK supported Bristol GDNF clinical trial, which involved tubes being implanted in the brain with pin point precision thanks to the assistance of robotics (a system developed by Prof Steven Gill and a company called Renishaw). These tubes connected with another tube which ran to a small port which was placed just behind the ear.
 The CDNF delivery system. Source: Medium
The CDNF delivery system. Source: Medium
CDNF is pumped into the brain via the small port place behind the ear once a month for 6 months, and the inital safety data that was presented at the ADPD meeting looked very good (Click here to read more about the trial). The treatment appears to be very well tolerated and safe.
This clinical trial has received funding from the European Union’s (Horizon 2020; #732386). The name of the project is TreatER. The researchers are now conducting an extension study in which they are hoping determine the efficacy of the CDNF treatment in the participants (Click here to read more about this study). They plan to have the results from this study available next year.
3. Sanofi Genzyme’s Venglustat safety results
The biotech company Sanofi Genzyme presented the safety/tolerability data from their Phase II MOVE-PD clinical trial evaluating their drug Venglustat in early stage GBA-associated Parkinson’s (Click here to read more about this study).
Small variations in a specific region of DNA called “GBA” are some of the most common genetic risk factors for developing Parkinson’s (Click here to read a previous SoPD post on this topic). Venglustat is a treatment that is targeted towards people that have GBA-associated Parkinson’s (Click here to read more on this).
The safety/tolerability data presented at the ADPD meeting looked good. There were no difference between the treatment and placebo groups in terms of frequency of complications, and there were no serious adverse events. In addition, the levels of blood and cerebrospinal fluid (the liquid our brains sit in) glucosylceramide synthase – the protein specifically inhibited by Venglustat decreased over 4 weeks (in a dose dependent manner).
This safety/tolerability study was Part 1 of a larger study being conducted by Sanofi Genzyme. And based on these results, the investigators conducting the study are now confident to move on to Part 2 of the clinical trial. Part 2 will determine the efficacy of Venglustat administered daily (compared to placebo) in 200+ people with recently diagnosed GBA-associated Parkinson’s. The study will involve 52 weeks of treatment period, followed by 104 weeks of follow-up assessments.
The results of that study will be available in 2022.
4. The Roche smart phone biomarkers research
Biomarkers was a major theme at the ADPD meeting.
And on the Parkinson’s side of things, Roche was clearly leading the pack with regards to the monitoring of Parkinson’s progression using smartphone technology. Kirsten Taylor, one of the Roche employees involved with this work, presented some very rich data that has been generated by their technology.
The company has previously conducted a Phase I clinical trial of their smartphone technology, which they have published and made publicly available:
 Title: Evaluation of smartphone‐based testing to generate exploratory outcome measures in a phase 1 Parkinson’s disease clinical trial
Title: Evaluation of smartphone‐based testing to generate exploratory outcome measures in a phase 1 Parkinson’s disease clinical trial
Authors: Lipsmeier F, Taylor KI, Kilchenmann T, Wolf D, Scotland A, Schjodt-Eriksen J, Cheng WY, Fernandez-Garcia I, Siebourg-Polster J, Jin L, Soto J, Verselis L, Boess F, Koller M, Grundman M, Monsch AU, Postuma RB, Ghosh A, Kremer T, Czech C, Gossens C, Lindemann M.
Journal: Mov Disord. 2018 Aug;33(8):1287-1297.
PMID: 29701258 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers at Roche conducted a 6‐month, phase Ib clinical trial involving 44 people with Parkinson’s completing 6 daily motor-associated tests using their smartphone. Their results were compared to an independent, 45‐day study conducted on 35 age‐matched healthy controls who also completed the 6 daily motor active tests.
 Source: PMC
Source: PMC
The daily motor assessment tests included:
-
Sustained phonation – making a continuous “ahh” sound for as long as possible.
-
Balance task – standing still while having the smartphone in the trouser pocket.
-
Gait analysis task – with the smartphone in the trouser pocket, walking 20 yards, turning around, and returning to the starting point.
- Finger‐tapping – with the smartphone on a flat surface alternately tapping two touchscreen buttons as regularly as possible.
-
Resting tremor – seated, holding the phone in the palm of the hand resting on the lap.
-
Postural tremor – seated, holding the phone in their outstretched hand.
Watch this video for an explanation of these tests:
The scores of each of these tests on the smart phone technology were compared with scores from clinical assessments that were made by neurologists during the study (as determined by the Unified Parkinson’s Disease Rating Scale or UPDRS).
And the really interesting aspect of the data was how well some of these tests picked up aspects of Parkinson’s that the neurologists did not!
If you look at panels A, B, C & D in the image below, you will see that the smart phone tests were picking up subtle differences that were indicative of Parkinson’s in tests like resting tremor & finger tapping in participants that were displaying no signs (or a score of 0) in the corresponding clinical assessments conducted by neurologists. It was only in the postural tremor test that the technology did not pick up any subtle changes.
 Source: PMC
Source: PMC
I really like this “detection of subtle differences” aspect of the technology from the standpoint of trying to find better methods of assessing Parkinson’s. By providing more accurate measures of the UPDRS, we begin at a better starting point in such an endeavour.
Roche is now using this method of monitoring Parkinson’s in their ongoing PASADENA study – a Phase II clinical trial of their immunotherapy treatment Prasinezumab (also known as RG7935/PRX002), which is being conducted in collaboration with the biotech firm Prothena Biosciences.
I should add here that the pharmaceutical company Biogen was also presenting smartphone Parkinson’s progression monitoring technology. And it was hugely encouraging to see major pharmaceutical companies investing serious resources in this biomarker technology. Not only from the view point for advancing the field of monitoring Parkinson’s progression (where we desperately need more tools), but also from the perspective of this effort being a sign of commitment for future clinical investigations in Parkinson’s.
5. All of the PPMI related research
There was also biomarker data being presented from the Parkinson’s Progression Markers Initiative (or PPMI).
Started in 2010, the PPMI project is a major observational clinical study which aims to identify biomarkers of Parkinson’s progression. A biomarker is an objectively measurable physical characteristic associated with a condition.
The project is doing this by assessing an international collection of people with Parkinson’s, which includes building the largest collection of clinical, imaging and biologic specimens ever created in the Parkinson’s community (Click here to read a previous SoPD post on this topic).
The project is being conducted in the United States, Europe, Israel, and Australia and it is sponsored by The Michael J. Fox Foundation.
In addition, an overview of the next step in the PPMI project or the “Path to Prevention” was presented. This new project will focus on establishing biomarkers for very early stage (or prodromal) Parkinson’s.
An amazing amount of biomarker-related data was presented at the meeting.
So you liked the ADPD meeting?
Loved it.
Exhausting as it was (there were a bunch of CPT meeting organised around the event), it was an extremely stimulating meeting.
I do, however, have two observations for consideration (not critiques, just observations).
Observation #1 – the lack of a patient voice
There were very few patients at the conference on Alzheimer’s and Parkinson’s. I saw one or two members of the Parkinson’s community in the audience at several sessions, but the absence of the affected community was a striking feature of the meeting.
On the ADPD website, it states:
“AD/PDTM is a unique conference that provides a world class platform for clinical investigators and basic scientists to present, discuss and trade expertise on a broad range of themes and topics”
And that is fine – if that is the goal of the meeting.
But without a patient presence, it felt a bit like a football conference without any football players. The sports agents, managers, shoe & football makers, and referees all showed up, but the players are not invited.
And as I said, this is not a critique, just an observation.
I also think that having more of patient presence is not something to be taken lightly. Involving them would depend on the goal of the meeting, and it would require careful consideration and management (particularly of expectations/frustrations when negative clinical data is presented).
But the patient voice can bring tremendous added value to such meetings.
My friend Ben Stecher – relentless Parkinson’s advocate and busy blogger – was the only member of the Parkinson’s community who actually took part in the meeting (and please correct me if I am wrong here).
He and Prof Bas Bloem presented a very good, Roche-sponsored session called “Now you see me: The voice of people with Parkinson’s in clinical trials“.
Here is a video of this presentation (recommended viewing!):
Also, check out this recent video of a lecture Ben gave at McGill University in Canada).
More of an emphasis on patients could provide the meeting with a breath of fresh air and provide young basic science researchers with an opportunity to learn about the lived experience of Parkinson’s.
It is just a thought.
Observation #2 – the public domain
I have become a bit of a twitter fiend (@scienceofPD).
It is a great method of sharing information, and I like to ‘tweet’ photos and info about presentations as they are being given. For example, this was one of many tweets sent from the APDP meeting:
I fully respect unpublished data, and do not post anything that I think might upset the person presenting the data. Rather I prefer to link readers with what is already publicly available (previously published reports, etc) and with websites regarding interesting tools/resources that may be discussed in the presentation.
But here’s the thing: Presenting data at an international conference is putting that information in the public domain.
Patents have not been awarded because data was disclosed at conferences – ergo, it is very much the public domain.
All of that said, in a couple of sessions at the ADPD meeting, the chairperson of the session or the ushers were instructing people not to take photos of the slides that were being presented on the big screen. In this modern age of technology, such requests were analogous to standing on the beach and verbally trying to stop the tide from coming in (many in the audience just ignoring the requests).
Where asked, I respected the request, but I have to say as a policy, it really grated me.
(Ok, maybe this is more of a critique than an observation, but it is being shared with the goal of trying to improve things)
The whole idea of a conference is to come together share data, propose ideas, and brainstorm horizons.
Otherwise, what on Earth is the point of holding a conference?
And in the modern age of technology, why limit ourselves to sharing ideas, data, etc with just the participants of the meeting???
 Source: ECU
Source: ECU
There is a pervasive culture of paranoia in biomedical research which infects all levels of it, and displays itself at some of these events. Seriously, in one session, a chairperson stood up at the start and said “if any photos are taken, I’ll stop this session immediately“.
It was shocking and such a sad/pathetic contrast to the IPDGC meeting I had just attended before the ADPD meeting which proactively encouraged a culture the sharing of information/data (Click here to read the SoPD post on that inspirational meeting). I understand that people’s livelihoods are on the line with some of this disclosure of data, and by sharing it they run the risk of “being scooped” with their next publication which affects their career and grant funding opportunities (Click here for a previous SoPD post about academic life).
But something has to change – such paranoia is not good for the ‘common good’.
Perhaps a patient presence at such meetings could help?
It’s just a thought.
Ok, does this mean you didn’t like the ADPD meeting?
No, as I said, I loved it. My rant above was just a shared observation. Folks can do with it whatever they like.
This was my first time at the ADPD meeting (the 14th time the meeting has been held), and for a curious mind, the event was a total overdose of information and a wonderful opportunity to meet and talk with researchers from all over the world working about all sorts of interesting areas of Parkinson’s. And I am hugely excited by many of the take aways from the meeting (particularly the biomarker research that is now being included in clinical trials). There are going to be a wave of future SoPD posts based on many of the presentations and discussions that were had at the ADPD meeting.
So what does it all mean?
The 2019 ADPD meeting was a marathon effort for organisers and participants alike. Five days of 8am till 8pm research presentations, poster presentations, meetings and associated activities (I didn’t get to see much of Lisbon, but it looked very nice – I will be heading back there at some point for a visit).
 Source: Wikipedia
Source: Wikipedia
The meeting was mainly an Alzheimer’s affair, but there was still a great deal of Parkinson’s research being presented – I have to admit I felt sorry for the poor AD researchers, as the amount of PD research presented already felt overwhelming! I am not sure how the AD researchers could have manage the volume of AD research.
This short post does not do justice to the amount of Parkinson’s research presented, but over the next few months I’ll be drip feeding nuggets of gold that were presented at the meeting.
ADPD is one of those gifts that keeps on giving.
EDITOR’S NOTE – The author of this post is an employee of the Cure Parkinson’s Trust which has supported the AiM-PD clinical trial. Neither the AiM-PD team nor CPT has asked for this post to be written. This post has been provided by the author solely for the purpose of sharing what the author considers very interesting information.
In addition, some of the companies mentioned in this post are publicly traded companies. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. None of the companies have requested that this material be produced, nor has the author had any contact with the companies beyond speaking with researcher employees about study data. This post has been produced for educational purposes only.
The banner for today’s post was sourced from ADPD














Simon,
Thanks for your report.
Was there much cross-fertilization between the AD and PD sides?
And, in general, should there be?
John
LikeLike
Hi John,
Glad you liked the post. Yes, there was cross-fertilization. On a personal level, curiousity drags me along to a lot of AD presentations (one in particular by a biotech company called Cortexzyme (https://www.cortexyme.com/) may well get it’s own post here in coming weeks if I have time. That company has a very interesting view on Alzheimer’s). But in general, the cross fertilization can not be helped as there is a lot of research being discussed around conditions like Dementia with Lewy bodies, which kind of bridge the two fields and requires comparative analysis with AD and PD. This was particularly evident in the neuropathological sessions.
Should there be? I am in two minds about this. On the one hand, I like the “don’t read anyone else’s research and just go discover stuff for yourself” idea (reading too much can bias our thinking and lead us down the paths others forged), but I also like to think that the AD and PD fields can learn a great deal from each other (for example, maybe that Cortexyme stuff).
What do you think?
Kind regards,
Simon
LikeLike
Fascinating blog, Simon. Thanks.
Of particular interest for me:
– the Ambroxol trial data (as I was on the trial and rather felt that my experience on the drug was met by some clinicians with extreme scepticism. I feel like a reverse Spike Milligan – “I told you I was well”)
– the thing about the smartphone app. As a serial volunteer, I’ve filled in countless questionnaires describing my sleep and long to say “here, look at the data from my Fitbit”. Definitely an underused resource. And if there’s any chance of getting away from that bloody Montreal assessment thing (face, velvet, church, daisy, red)!
There’s stuff about the Lisbon earthquake in Candide, I seem to remember from A level days. Ah yes, just looked it up. Apparently, you can prevent earthquakes by burning heretics. So that’s all good.
LikeLike
Hi Jellywoman,
You’re welcome. Glad you liked the post.
I would be curious to hear more about your experience of being in a clinical trial (particularly for the sake of those who have never made that choice).
Regarding fitbits, etc. – we are currently working on an idea for collecting all of that un-collected data, and we will hopefully have something on that soon.
And moving away from the learned memory tests is a must. I have been skeptical about the smartphone approach for a long time (“too much of an intrusion in people’s lives, requiring too much effort, adherence will drop off too quickly,…etc”). But I have to be honest: I was very impressed by the richness of some of the data some of the research groups were presenting.
Kind regards,
Simon
LikeLike
Simon I applaud your stance on the “Observation #2 – the public domain” part. I couldn’t agree more!!! Also, thanks for sharing your take on the meeting, cheers!
LikeLike
Thanks Diego, glad you liked the post and agree with the public domain rant.
Kind regards,
Simon
LikeLike
There are some patients who take ambroxol – import it from Germany
https://www.smartpatients.com/conversations/41861?sb_post_id=679017#sbposts-679017
Not recommending or warning against this. I have no idea
LikeLike
Regarding the syndrome known as PD: how much clinical sub-group analysis is done in therapeutic trials?
Regarding the involvement of patients in conferences: how could it be made more useful? I’m a retired rheumatologist and my wife has PD, but many people would not identify her problems as PD.
LikeLike