|
As the amazing Australian Parkinson’s Mission project prepares to kick off, across the creek in my home land of New Zealand, another very interesting clinical trial programme for Parkinson’s is also getting started. The study is being conductetd by a US biotech firm called resTORbio Inc. The drug being tested in the study is called RTB101. It is an orally-administered TORC1 inhibitor, and it represents a new class of drug in the battle against Parkinson’s. In today’s post, we will look at what TORC1 is, how the drug works, the preclinical research supporting the trial, and what this new clinical trial will involve.
|
 Rapa Nui. Source: Chile.Travel
Rapa Nui. Source: Chile.Travel
Today’s post kicks off on an amazing south Pacific island… which is not New Zealand.
In 1965, a rather remarkable story began in one of the most remote inhabited places on Earth – the mysterious island of Rapa Nui (or “Easter Island”).
And when we say ‘remote’, we really do mean remote. Did you know, the nearest inhabited island to Rapa Nui is Pitcairn Island, which is 2,075 kilometres (1,289 mi) away. And Santiago (the capital of Chile) is 2,500 miles away – that’s a four-hour+ flight!!!
Rapa Nui is the very definition of remote. It is as remote as remote gets!
 Does Amazon deliver to the town of Hanga Roa? Source: Atlasandboots
Does Amazon deliver to the town of Hanga Roa? Source: Atlasandboots
Anyways, in 1965 a group of researchers arrived at Rapa Nui with the goal of studying the local inhabitants. They wanted to investigate their heredity, environment, and the common diseases that affected them, before the Chilean government built a new airport which would open the island up to the outside world.
It was during this investigation, that one of the researchers – a University of Montreal microbiologist named Georges Nógrády – noticed something rather odd.
What?
At the time of the study, wild horses on Rapa Nui outnumbered humans (and stone statues).
Wild horses roaming the east coast of Rapa Nui. Source: Farflungtravels
But what was odd about that?
Georges discovered that locals had a very low frequency of tetanus – a bacterial infection of the feet often found in places with horses. He found this low incidence of tetanus particularly strange given that the locals spent most of their time wandering around the island barefoot. So Georges decided to divide the island into 67 regions and he took a soil sample from each for analysis.
In all of the vials collected, Nógrády found tetanus spores in just one vial.
Something in the soil on Rapa Nui was extremely anti-fungal.
In 1969, Georges’ collection of soil samples was given to researchers from the pharmaceutical company Wyeth and they went looking for the source of the anti-fungal activity. After several years of hard work, the scientists found a soil bacteria called Streptomyces hygroscopicus which secreted a compound that was named Rapamycin – after the name of the island – and they published this report in 1975:
 Title: Rapamycin (AY-22, 989), a new antibiotic
Title: Rapamycin (AY-22, 989), a new antibiotic
Authors: Vézina C, Kudelski A, Sehgal SN.
Journal: J Antibiot (Tokyo). 1975 Oct;28(10):721-6.
PMID: 1102508 (This report is OPEN ACCESS if you would like to read it)
It is no understatement to say that this was a major moment in biomedical history. So much so that there is actually a plaque on the island commemorating the discovery of rapamycin:
 Source: DiscoveryMag
Source: DiscoveryMag
Why was the discovery of ‘anti-fungal’ rapamycin so important?!?
Because it didn’t just have anti-fungal properties.
The researchers at Wyeth discovered that rapamycin did a lot more than just kill fungi (and before we continue, I can recommend you read this article about the history of the discovery of Rapamycin – it’s an amazing story!).
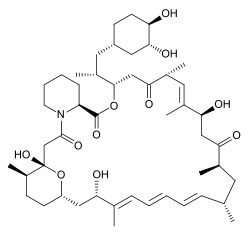 Rapamycin. Source: Wikipedia
Rapamycin. Source: Wikipedia
Rapamycin was also a powerful suppressor of the immune system (reducing the ability of the immune system to do its job) and it demonstrated strong anti-cell proliferative properties. These properties resulted in new immune suppression treatments for organ transplant procedures (Rapamycin itself received clinical approval in 1999 for use in the prevention of organ rejection in kidney transplant patients) as well as novel cancer treatments (Torisel and Afinitor were approved in 2007 and 2009 for treatment of advanced kidney cancer), in addition to a host of other medical applications.
But what really fascinated the researchers was the HOW.
How was Rapamycin doing all of these things? How was a compound that was produced by a soil bacteria be having all of these effects?
And after many years of further research they discovered that these properties resulted from rapamycin’s interactions with a protein called mTOR.
What is mTOR?
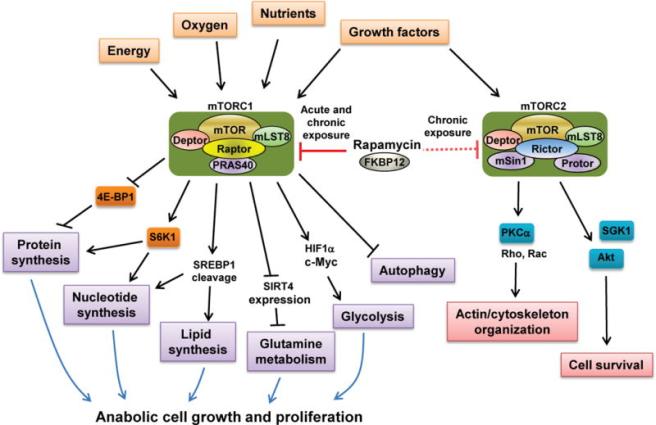
Mechanistic target of rapamycin (or mTOR) is a protein that serves as a central regulator of cell metabolism, growth, and survival. It integrates signals from both inside (intracellular) and outside (extracellular) the cell.
It performs its various activities via two protein complexes (that is a combination of proteins that must bind together in order to do their function).
These two complexes are known as mTOR Complex 1 (TORC1) and mTOR Complex 2 (TORC2).
 TORC1 and TORC2. Source: dev.biologists
TORC1 and TORC2. Source: dev.biologists
What do TORC1 and TORC2 do?
mTORC1 has four components plus mTOR and it is a positively regulator of cell growth and proliferation. It encourages the biosynthesis of new proteins and lipids, and limits recycling processes (such as autophagy)
What is autophagy?
Autophagy (from the Ancient Greek αὐτόφαγος autóphagos, meaning “self-devouring”) is an absolutely essential function in a cell. Without autophagy, old proteins would pile up making the cell sick and eventually causing it to die. Through the process of autophagy, the cell can break down the old protein, clearing the way for fresh new proteins to do their job.

The process of autophagy. Source: Wormbook
Waste material inside a cell is collected in membranes that form sacs (called vesicles). These vesicles then bind to another sac (called a lysosome) which contains enzymes that will breakdown and degrade the waste material – the same way enzymes in your washing powder break down muck on your dirty clothes). The degraded waste material can then be recycled or disposed of by spitting it out of the cell.
While activating TORC1 reduces autophagy, inhibiting TORC1 activity results in an increase in autophagy – the waste disposal and recycling process (remember this last part – inhibiting TORC1 activity results in an increase in autophagy – the waste disposal and recycling process).
Reducing TORC1 activity has also been shown to prolong lifespan, enhance immune function, enhancing memory, and delaying the onset of age-related conditions in multiple animal models (Click here for a review of this topic).
And what about TORC2?
On a very basic level, TORC1 and TORC2 are like yin & yang.
While inhibiting TORC1 is associated with lots of good stuff, TORC2 inhibition has been associated with lots of bad stuff (eg. decreased lifespan, worsening memory, etc – Click here for more on this).
This major difference has lead a lot of biotech/pharmaceutical companies seeking to develop drugs that inhibit TORC1, but do NOT disrupt TORC2 activity.
But hang on a second, what about rapamycin? How does it interact with mTOR, TORC1 and TORC2?
Rapamycin is an inhibitor of mTOR – it has a mTOR binding region (see image below), but it affects TORC1 and TORC2 differently.
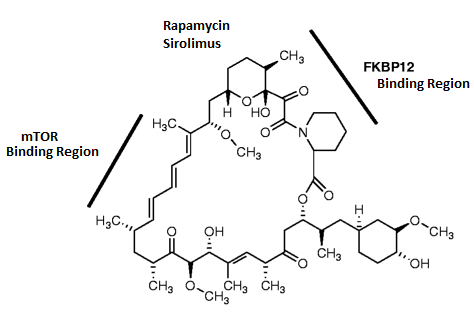 Rapamycin binding regions. Source: Agscientific
Rapamycin binding regions. Source: Agscientific
TORC1 is very sensitive to rapamycin, and becomes inhibited in its presence. TORC2, on the other hand, is considered to be resistant to rapamycin (except under conditions of long term exposure).
 Source: PMC
Source: PMC
Ok, so rapamycin inhibits mTOR/TORC1 which enhances autophagy/waste recycling. But what does any of this have to do with Parkinson’s?
Rather a lot of research has been conducted on mTOR & autophagy in the context of Parkinson’s.
Firstly, there have been postmortem examinations of the Parkinsonian brain suggesting issues with mTOR activity in PD. For example, this study:
 Title: Selective molecular alterations in the autophagy pathway in patients with Lewy body disease and in models of alpha-synucleinopathy.
Title: Selective molecular alterations in the autophagy pathway in patients with Lewy body disease and in models of alpha-synucleinopathy.
Authors: Crews L, Spencer B, Desplats P, Patrick C, Paulino A, Rockenstein E, Hansen L, Adame A, Galasko D, Masliah E.
Journal: PLoS One. 2010 Feb 19;5(2):e9313.
PMID: 20174468 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers found that levels of mTOR were higher in the brains of individuals who passed away with Lewy body-associated conditions (such as Parkinson’s or Lewy body dementia) than the brains of unaffected control subjects or even brains from people with Alzheimer’s.
The investigators also reported similar high levels of mTOR in the brains of mice that had been engineered to produce high levels of the Parkinson’s associated protein, alpha synuclein. This protein builds up in the brain in Parkinson’s and is believed to be associated with the cell death that is seen in the condition. The researchers in this study treated these mice with rapamycin, which reduced the accumulation of alpha synuclein in these mice and protected them from “associated neurodegenerative alterations”.
Other research groups have also demonstrated the ability of rapamycin to rescue alpha synuclein-based models of Parkinson’s (Click here and here to read examples). And rapamycin has also been shown to have beneficial effects in other neurotoxin-based models of Parkinson’s.
For example, this report:
 Title: Rapamycin protects against neuron death in in vitro and in vivo models of Parkinson’s disease.
Title: Rapamycin protects against neuron death in in vitro and in vivo models of Parkinson’s disease.
Authors: Malagelada C, Jin ZH, Jackson-Lewis V, Przedborski S, Greene LA.
Journal: J Neurosci. 2010 Jan 20;30(3):1166-75.
PMID: 20089925 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers found that rapamycin (“rapa”in the image below) was able to protect cells in various neurotoxin-based models of Parkinson’s.
 A condensed nucleus in a cell suggests the cell is dying. Source: PMC
A condensed nucleus in a cell suggests the cell is dying. Source: PMC
Rapamycin achieved this effect by only inhibiting certain mTOR activities, because when the investigators compared rapamycin to a more complete mTOR inhibitor (Torin1, which blocks both TORC1 and TORC2 activities), only rapamycin was able to protect the cells exposed to the neurotoxins.
And similar neuroprotective results from rapamycin treatment in neurotoxic models of Parkinson’s have also been published by other research groups (click here, here, and here to read some examples).
In addition to these alpha synuclein and neurotoxin models, rapamycin has been reported to have beneficial effects in models of genetic forms of Parkinson’s (specific genetic variations in DNA account for 10-20% of cases of Parkinson’s – Click here to read more about the genetics of PD)
Such as this report:
 Title: Rapamycin activation of 4E-BP prevents parkinsonian dopaminergic neuron loss.
Title: Rapamycin activation of 4E-BP prevents parkinsonian dopaminergic neuron loss.
Authors: Tain LS, Mortiboys H, Tao RN, Ziviani E, Bandmann O, Whitworth AJ.
Journal: Nat Neurosci. 2009 Sep;12(9):1129-35.
PMID: 19684592 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported that rapamycin rescued mitochondrial defects in cells collected from people with PARKIN-associated Parkinson’s (Click here to read a previous SoPD post about PARKIN and the related mitochondrial issues). In fact, the investigators found that when flies with Parkinson’s-associated PARKIN or PINK1 genetic mutants were raised and aged on rapamycin supplemented food, they exhibited little or no dopamine neuron degeneration.
And it is not just PARKIN/PINK1-associated Parkinson’s that could benefit from mTOR-modulation, there is also evidence supporting this approach in GBA-associated Parkinson’s (Click here to read a previous SoPD post about GBA-associated Parkinson’s):
 Title: mTORC2 Promotes Tumorigenesis via Lipid Synthesis
Title: mTORC2 Promotes Tumorigenesis via Lipid Synthesis
Authors: Guri Y, Colombi M, Dazert E, Hindupur SK, Roszik J, Moes S, Jenoe P, Heim MH, Riezman I, Riezman H, Hall MN.
Journal: Cancer Cell. 2017 Dec 11;32(6):807-823.e12.
PMID: 29232555 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers found that mTOR activity promotes lipid synthesis, particularly glucosylceramide. Although this particular study was conducted in the context of cancer, the findings are interesting for two reasons:
- Accumulation of glucosylceramide is associated with GBA-associated Parkinson’s (Click here to read more about this).
- The investigators found that mTOR inhibition reduced glucosylceramide levels.
And it has been previously reported that rapamycin treatment can rescued models GBA-associated Parkinson’s (Click here to read more about this).
Thus, there is ample preclinical evidence indicating that rapamycin/mTOR modulation could be interesting to assess in the context of Parkinson’s.
And not just Parkinson’s.
There is evidence for TORC1 inhibition potentially having beneficial effects in many conditions associated with intracellular protein aggregation. Most recently, it was reported that a protein called dynasore inhibits TORC1 activity and this helps to reduce levels of proteins associated with the neurodegenerative condition of Huntington’s disease.
Dinosaur?
No Dynasore.
Here is the report:
 Title: Dynasore Suppresses mTORC1 Activity and Induces Autophagy to Regulate the Clearance of Protein Aggregates in Neurodegenerative Diseases.
Title: Dynasore Suppresses mTORC1 Activity and Induces Autophagy to Regulate the Clearance of Protein Aggregates in Neurodegenerative Diseases.
Authors: Chen Y, Xu S, Wang N, Ma Q, Peng P, Yu Y, Zhang L, Ying Z, Wang H.
Journal: Neurotox Res. 2019 Mar 29 [Epub ahead of print]
PMID: 30924108
In this study, the researchers found that by treating cells with a protein called dynasore they inhibited the activity of TORC1, which resulted in an increase in autophagy. When they caused cells to produce high levels of the toxic Htt protein (associated with Huntington’s disease) and then treated the cells with dynasore, they found that TORC1 levels were reduced and autophagy activity increased, which enabled these cells clear the build up of this neurodegeneration-associated protein.
Remind me again, what is Huntington’s disease?
Huntington’s disease is a neurodegenerative condition which results in individuals losing all inhibition of movement, giving rise to appearance of chorea – jerky, random, and uncontrollable movements (similar to dykinesias).
Huntington’s disease is caused by an increase in a region of DNA inside the Huntingtin (Htt) gene. This regions is made up of CAG repeats. In normal healthy humans, we usually have up to 30 repeats of CAG. If you have more than 40 CAG repeat, you are definitely going to develop Huntington’s disease.
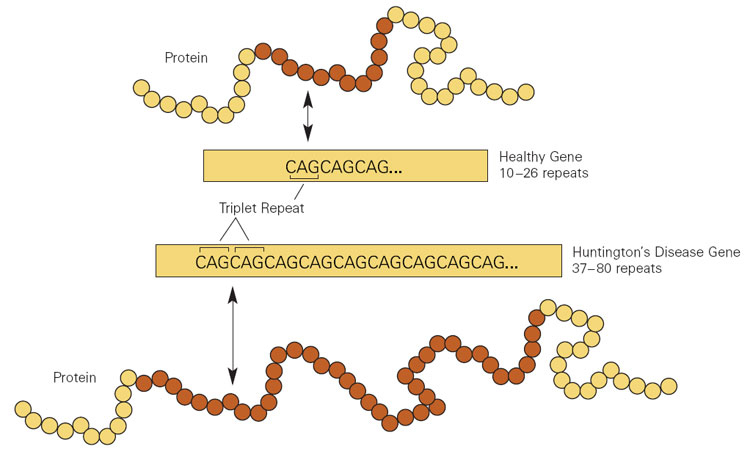
Expansion of CAGs in the Huntingtin gene. Source: NIST
The expansion of the CAGs in the Huntingtin gene results in a mutant Huntingtin protein, which is believed to be associated with the problems observed in people suffering from Huntinton’s disease.
Interesting. So the take-home message is TORC1 inhibition increases autophagy/waste disposal which helps to clear the accumulation of proteins associated with neurodegenerative conditions?
Exactly.
So where can I get me some rapamycin?
Yeah, before you think about doing that, please understand that clinicians and researchers are extremely weary about long term use of rapamycin for any condition.
As we discussed above, TORC2 is considered to be resistant to rapamycin (except under conditions of long term exposure). The great worry about using rapamycin over the long term is that it will start to affect TORC2 as well as TORC1 activity, and that would not be a good thing.
There has been tremendous excitement generated around mTOR modulation as a means of extending life span (this has only been demonstrated in mice, not in humans), but the long term use of rapamycin has several undesired side effects.
Two in particular:
- Suppression of the immune system (maybe you’ll live longer, but get the flu more often)
- An increase in metabolic defects (maybe you’ll live longer, but you could develop hyperglycemia, insulin resistance or type 2 diabetes)
As a result of these issues, there has been a great deal of research by biotech/pharmaceutical companies seeking alternatives to rapamycin. Specifically, they have been working on drugs that ONLY inhibit TORC1.
Which brings us to resTORbio Inc.
 This Boston (USA) based biotech firm has developed an experimental compound called RTB101, which is a potent TORC1 inhibitor. The company has been clinically testing this drug on respiratory tract infections in the elderly, but encouraged and supported by the Silverstein Foundation, they are now testing the therapeutic potential for RTB101 in Parkinson’s.
This Boston (USA) based biotech firm has developed an experimental compound called RTB101, which is a potent TORC1 inhibitor. The company has been clinically testing this drug on respiratory tract infections in the elderly, but encouraged and supported by the Silverstein Foundation, they are now testing the therapeutic potential for RTB101 in Parkinson’s.
Which brings us to this new clinical trial in New Zealand.
What will the study involve?
The new Phase Ib/IIa study will evaluating the safety and tolerability of resTORbio’s TORC1 inhibitor RTB101 in 45 people with recently diagnosed Parkinson’s (who are already on standard care – Click here to read the press release).
The participants will be randomly assigned to one of five groups:
• Group 1: RTB101 300 mg capsule given orally once per week for up to 4 weeks.
• Group 2: Sirolimus 2 mg tablet given orally once per week for up to 4 weeks.
• Group 3: RTB101 300 mg capsule + sirolimus 2 mg tablet given orally once per week for up to 4 weeks.
• Group 4: RTB101 300 mg capsule + sirolimus 4 mg tablet given orally once per week for up to 4 weeks.
• Group 5: RTB101 300 mg capsule + sirolimus 6 mg tablet given orally once per week for up to 4 weeks.
The study will including people with and without GBA genetic variants. The primary endpoint of the study will be safety and tolerability, and the secondary endpoints will be amount of drug in blood and cerebrospinal fluid (the liquid that our brains sit in). There are also some exploratory endpoints, which will include blood and cerebrospinal fluid biomarkers, as well as some clinical assessments.
The results of the trial are expected in 2020 (click here to read more about this trial).
What is sirolimus?
Sirolimus is rapamycin (Click here for the wikipedia page and click here to read more about this drug).
And why are they testing it in combination with RTB101?
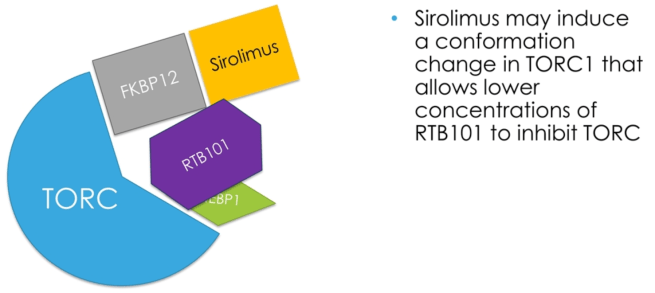
The reason the researchers are using Sirolimus is because they are keen to learn if it changes the shape of the TORC1 protein complex, which would allow a lower dose of RTB101 to be used over time. Can using both drugs in combo allow for lower doses of both, as opposed to high doses of them being used by themselves.
 Source: SEC
Source: SEC
Has RTB101 ever been tested in humans before?
Yes it has.
There have been several studies. Most recently RTB101 was tested in a Phase IIb clinical trial for reducing the incidence of respiratory tract infections (Click here to find out more about that trial). The results of that trial were announced by resTORbio last July (Click here to read the press release). And there was additional analysis of data from the study announced in October (Click here to read more about this).
The study found that all doses of RTB101 were safe and well-tolerated, and that 10 mg of RTB101 once daily had a comparable safety profile to the placebo control treatment. Note that this was a daily treatment regime, whereas the NZ trial will involve just a weekly dosing.
Collectively data from two Phase II clinical trials of RTB101 (testing the drug in more than 900 elderly subjects) suggest that this drug is well tolerated and resulted in a 30.6% reduction in the incidence of laboratory-confirmed respiratory tract infections.
Sounds good!
Interesting. So summing up? What does it all mean?
No, we’re not done yet.
There is another reason why TORC1 inhibition is interesting in the context of Parkinson’s: Dyskinesias
 Title: Inhibition of mTOR signaling in Parkinson’s disease prevents L-DOPA-induced dyskinesia.
Title: Inhibition of mTOR signaling in Parkinson’s disease prevents L-DOPA-induced dyskinesia.
Authors: Santini E, Heiman M, Greengard P, Valjent E, Fisone G.
Journal: Sci Signal. 2009 Jul 21;2(80):ra36.
PMID: 19622833
In this study, the researchers found that the development of L-DOPA-induced dyskinesia in a mouse model of Parkinson’s led to dopamine D1 receptor-mediated activation of TORC1. They also demonstrated that treating the mice with rapamycin prevented the development of dyskinesia without affecting the therapeutic properties of L-DOPA.
And this research has been independently replicated in rats (Click here to read more about this), plus there is evidence to suggest that genetic variations in genes associated with the mTOR pathway could leave some individuals susceptible to developing L-DOPA-induced dyskinesias (Click here to read more about this).
Thus, even if RTB101 fails to demonstrate any long-term disease modifying properties in Parkinson’s in future studies, it could still have the potential to improve quality of life for individuals with Parkinson’s who are suffering from dyskinesias.
So what does it all mean?
As a proud kiwi I am always keen to highlight new Parkinson’s related research being generated in the homeland (for example, the kiwi ketogenic diet in Parkinson’s study – Click here for that SoPD post). I am also very pleased to see clinical trials focused on disease modification in Parkinson’s being conducted in New Zealand (for example the Living Cell Technology clinical trials – Click here to read the SoPD post on that topic).
And this new study is of particular interest to me – I am very curious to see the outcome of this clinical trial programme.
As we have discussed before (Click here for that post), any “curative” therapy for Parkinson’s will require a disease-halting mechanism. And one potential “indirect” method of slowing/stopping the progression of Parkinson’s is boosting the waste disposal system of the cell. By ‘chewing up and spitting out’ the accumulating alpha synuclein and associated proteins, it is hoped that cells will be able to function better and be more healthy. This would predictably slow down the progression of the condition. Thus, given that RTB101 enhancing autophagy (waste disposal) activity in cells, I like the premise behind this new trial.
That said modulating autophagy is a difficult balancing act. But it will be interesting to see if just a slight increase could have beneficial effects for folks with Parkinson’s.
Silver fern koru. Source: Wikimedia
In New Zealand, the koru is a spiral symbol based on the appearance of a new unfurling silver fern frond. It has very important symbolism for the native people of Aotearoa – the Maori – and it has taken a permanent place in the modern society. It represents new life, growth, and strength. Nice symbolism for this new study.
Haere haumaru RTB101.
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
In addition, the company resTORbio Inc which is mentioned in this post is a publicly traded company. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. resTORbio Inc has not requested that this material be produced, nor has the author had any contact with the company. This post has been produced for educational purposes only.
The banner for today’s post was sourced from restorbio.




Kiwis are so cool! Awesome post.
LikeLiked by 1 person
Thanks Diana – I’m slightly biased but I’d probably agree on both points 🙂
LikeLike
OMG! …creeps all over the body…
LikeLike
Hi Scatmen, not sure what you mean, but thanks for the comment 🙂
Kind regards,
Simon
LikeLiked by 1 person
Dear Simon, an emotional creep all over the body while reading 🙂 I mean that it is so so so astonishing and interesting… layer-BY-layer you dig the research science like a magician! …. 🙂 so OMG! THANK YOU Simon 🙂
LikeLike
Thanks Simon. Very Interesting. I note that there are now a few ‘respiratory’ medications (RTB101, NAC, Ambroxol) that may be useful in Parkinson’s treatment. As a person with ideopathic COPD as well as PD, I pay attention to such things. I wonder sometimes if there’s a causal link between COPD and PD; I’ve searched on line, but the only thing I find is that PD sometimes causes COPD because of accidental inhalation of food as a consequence of dysphagia. Do you think there could be a causality working the other way? For example, hypoxia predominately picking off the dopaminergic neurons of the SNpc? Kind of stands the “free radical/oxidant” theories on their heads, doesn’t it? Not breathing is the ultimate antioxidant! (in more ways than one)
LikeLiked by 1 person
yes I agree (RTB101, NAC, Ambroxol) …. something big is coming here…
LikeLike
Ahah! https://parkinsonsnewstoday.com/2019/04/30/lack-of-tissue-oxygenation-sleep-apnea-linked-parkinsons-disease/
Looks like there might be confirmation!
LikeLike
Interesting stuff. So, once again, a drug that is being used for anti-aging (rapamycin) could be the answer to PD, and other diseases that generally attack us in our later years. Check out the list of interventions this guy is prescribing to his patients for longevity enhancement and then see how often many of them have been mentioned here at SoPD as possible PD therapies.
One small point: Rapamycin seems to have a very low risk of side effects when taken intermittently (once every 1-3 weeks) at low doses (3-6mg). I can wait for the RTB101 results, but I’m starting sirolimus soon, (after my yearly physical and complete blood work). I’ll check it again in 3-6 months. I think a big problem is the lack of objective measurements for physiological age.
Simon, you are aware (better than most) of how powerful the placibo effect can be in PD patients. I will say this about Fisetin (the newest senolytic being trumpeted). After following the protocol being used in the Mayo Clinic trials, my brain fog and short term memory issues seemed to disappear. Then, about a week later, I felt horrible, as if I had a flu or bad cold. After a day or two, my energy level was way up. My wife (who is 20 years my junior) likewise tested Fisetin, and she had similar experiences, so if it’s a placibo, I’ll take more of those, please!
Planning a self-trial of Dasatinib and Quercetin soon.
Here’s to living healthy for as long as we can and avoiding those nasty diseases like AD/PD/etc. At 65, I’m working in IT, and doing just fine, thanks, competing and working with guys up to 35 years younger than I am!
LikeLiked by 1 person
Gavril, can you tell me whether you took the entire 20mg/kg daily dose of Fisetin all at once, or spread out through the day in multiple doses? The description of the Mayo clinical trials does not indicate one or the other method of dosing.
LikeLike
300 mg at once, not spread out during the day.
LikeLike
Thank you Simon as always for another fabulous piece.
Question: Is there any indication of which you are aware of the possible pricing of RTB101?
Joe in NY
LikeLike
A more general question that this article brings to mind is: how much of PD is caused by neurons that have been irretrievably lost, and how much is due to neurons whose functioning has been impaired by the illness, and which might therefore be revived?
Because certainly if we can TORC up the garbage disposal within neurons, that might tend to prevent neurons from being lost. But one might also wonder about how much capacity is recoverable by improving the functioning of neurons that have not quite bit it but are simply unable to function well.
Just wondering if you had any insight into this.
LikeLike