|
One of the characteristic features of Parkinson’s is the loss of dopamine neurons in the brain. One experimental approach that is being explored for treating the condition involves investigating biological pathways involved in the early development of dopamine neurons. By re-exposing the dopamine neurons to supportive, growth-encouraging proteins that are present during early development (but absent or reduced in adulthood), researchers hope to be able to rejeuvinate these cells and make them healthier. In a recent post, we discussed one such developmental supportive protein: Glial cell derived neurotrophic factor (Click here to read more about this). In today’s post, we will look at a different type of developmental protein which is having interesting effects in models of Parkinson’s. That protein is called Sonic Hedgehog.
|
The humble fly (Drosophila). Source: Ecolab
No one should ever be allowed to say that fly genetists don’t have a sense of humour.
When it comes to the naming of genes, they have had a great deal of fun. A gene is a section of DNA that can be copied into RNA (which may then provide the instructions for making a protein), and each gene has been given a name. Some names are boring – such as leucine-rich repeat kinase 2 (or LRRK2… Boring!) – while other names are rather amusing.
For example, there is one fly gene called indy, which stands for I‘m Not Dead Yet. Flies with genetic variation in this gene have longer than average lifespans (Click here to read more about this).
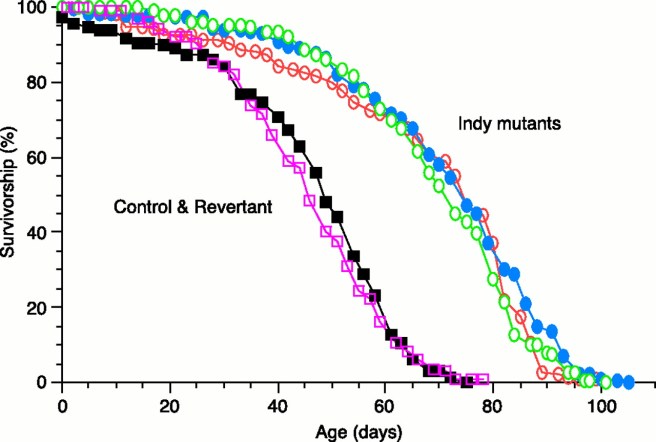 Source: Sciencemag
Source: Sciencemag
Another amusingly named gene is Cheap Date. Flies with a genetic mutation in this gene are very susceptible to alcohol (Click here to read more about this).
Source: Lordsofthedrinks
There is also Ken and Barbie – genetic variations in this gene result in a lack of external genitalia (Click here to read more about this). There are lots of great gene names: “lunatic fringe”, “headcase” and “mothers against decapentaplegia (MAD)”. Like I said, fly genetists have a lot of fun.
But one of the most popular gene names in all of biology is a gene called Sonic Hedgehog.
What is Sonic Hedghog?
First discovered in flies, the Hedgehog signaling pathway controls a wide range of developmental processes and is implicated in a variety of cancers.
In flies, there is only one hedgehog proteins, but in humans there is a family of three (sonic, desert, and indian). The name ‘hedgehog’ comes from the original discovery of the gene in flies (Drosophila) – flies that carry a mutated form of the hedgehog gene will grow up and develop into something that is said to resemble a hedgehog (another example of fly genetists humour).
This extreme effect on development points towards a major role for hedgehog proteins in the development of organisms though, and in mammals, sonic hedgehog is a major player.
How does sonic hedgehog function?
Sonic Hedgehog is a protein that is released by cells, and it functions by binding to a receptor called ‘Patched’. A receptor is like a switch, which is waiting for the right protein to come along and turn it on. Patched is sitting on the outer surface of a cell waiting for Sonic to come along and activate it.
In the absence of Sonic Hedgehog, Patched inhibits a transmembrane protein called ‘Smoothened’ which allows the ‘Suppressor of Fused’ (SUFU) to block any transcriptional activity – the transcribing of DNA into RNA – of particular Sonic Hedgehog associated genes. When Sonic Hedgehog does bind to Patched it blocks ‘Smoothened’, which results in transcriptional activity of Sonic Hedgehog associated genes.
 Sonic hedgehog signalling. Source: Cincancerres
Sonic hedgehog signalling. Source: Cincancerres
In the image above, in panel B there are a number of drugs that are named as inhibitors of this pathway (all of them acting as inhibitors, noting the role Sonic Hedgehog plays in several cancers).
Is Sonic Hedgehog named after the 1990s video game?
The story behind the name is that a British research scientist named, Robert Riddle (who was working in Professor Cliff Tabin’s lab where Sonic Hedgehog was first discovered – one of three labs that co-discovered it) proposed the name from a comic book that his daughter brought over from the U.K. At the time the videogame was not available in the U.S.
 The evolution of Sonic the hedgehog (& computer games). Source: Youtube
The evolution of Sonic the hedgehog (& computer games). Source: Youtube
The name was not universally popular though, and some members the Human Genome Organization’s Nomenclature Committee (yes, such an organisation does exist – sounds like a hoot!) put Sonic on their top 10 list of gene names that they wanted to change.
A big argument insued, but silliness won the day and the name stuck.
It’s a great name. Why did they want to change the name?
Basically, clinicians were having a hard time explaining to patients that their conditions were associated with genes that had silly names.
I mean, imagine your doctor telling you that you have a very serious medical condition which is associated with an error in your ‘lunatic fringe’ gene!
Or even better: your ‘headcase’ gene!
And it actually got even more silly: A potential inhibitor of the sonic hedgehog signaling that was being developed for cancer, was named “Robotnikinin” – a salute to Sonic the Hedgehog’s nemesis, Dr. Ivo “Eggman” Robotnik.
 Dr. Ivo Robotnik. Source: Wikipedia
Dr. Ivo Robotnik. Source: Wikipedia
Imagine your experimental treatment being named after a Disney character.
Or one of the Simpsons.
One can certainly appreciate where the clinicians were coming from.
I see, but getting back to Sonic Hedgehog, what does it have to do with Parkinson’s?
Sonic Hedgehog is critical for the development of dopamine neurons.
Remind me again, what are dopamine neurons?
Dopamine is a chemical that the brain uses to help pass messages/signals between cells (also known as a neurotransmitter). The bulk of the dopamine generated by your brain is produced by a population of dopamine producing neurons in a region of the midbrain called the substantia nigra.
The loss of these dopamine neurons is one of the cardinal features of the Parkinsonian brain. The substantia nigra (or ‘substance dark’) region is visible (with the human eye) on postmortem sections of brain due to the production of a molecule called neuromelanin in the dopamine neurons. And as you can see in the image below, the Parkinsonian brain has less dark pigmented cells in the substantia nigra region of the midbrain.
 The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinsonian brain (right). Source:Memorangapp
The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinsonian brain (right). Source:Memorangapp
The loss of dopamine in the brain is associated with the appearance of the motor features of Parkinson’s. By the time an individual is diagnosed with the condition, they have lost approximately 40-60% of the dopamine neurons in the substantia nigra region.
Ok, and this protein called sonic hedgehog is important for the development of these dopamine neurons?
Yes.
Back in 1995, researchers first reported that sonic hedgehog was required for the generation of dopamine neurons:
 Title: Induction of midbrain dopaminergic neurons by Sonic hedgehog.
Title: Induction of midbrain dopaminergic neurons by Sonic hedgehog.
Authors: Hynes M, Porter JA, Chiang C, Chang D, Tessier-Lavigne M, Beachy PA, Rosenthal A.
Journal: Neuron. 1995 Jul;15(1):35-44.
PMID: 7619528 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers found that inhibiting Sonic Hedgehog signalling blocked the development of dopamine neurons. They also found that when Sonic Hedgehog protein was added to the solution cells were growing in, it was able to induce the production of dopamine neurons. This finding was hugely exciting for the scientists as they were interested in the idea of growing dopamine neurons in cell culture for the purpose of cell replacement therapy for Parkinson’s (Click here to read more about this). They knew that any factor that increases the development of dopamine neurons would be very useful.
And these results were independently replicated shortly after this report was published (Click here to read more about the second report).
Almost immediately researchers began asking: If sonic hedgehog can give rise to dopamine neurons, can it also protect them? That is to say, are these genes that are critical to the development of dopamine neurons, also able to rescue them from disease or insult?
The first indications that this may be so were reported in 2002:
 Title: Intrastriatal injection of sonic hedgehog reduces behavioral impairment in a rat model of Parkinson’s disease.
Title: Intrastriatal injection of sonic hedgehog reduces behavioral impairment in a rat model of Parkinson’s disease.
Authors: Tsuboi K, Shults CW.
Journal: Exp Neurol. 2002 Jan;173(1):95-104.
PMID: 11771942
In this study, the researchers treated rat dopamine neurons with Sonic Hedgehog before and after exposure to a neurotoxin and found that this administration reduced the behavioural deficits and preserved some of the dopamine neuron branches in the brain.
And this result was also rapidly replicated and extended by independent researchers (Click here to read more about that). There has also been a gene therapy-based replication of this result (Click here to read more about this). Gene therapy involves introducing DNA into cells to produce new proteins (Click here to read a previous SoPD post about this topic). By introducing Sonic Hedgehog DNA into the brain, the researchers were able to improve outcomes in a rodent model of Parkinson’s.
In addition to these findings, it has been reported that Sonic Hedgehog helps to maintain the dopamine system in the adult brain:
 Title: Sonic hedgehog maintains cellular and neurochemical homeostasis in the adult nigrostriatal circuit.
Title: Sonic hedgehog maintains cellular and neurochemical homeostasis in the adult nigrostriatal circuit.
Authors: Gonzalez-Reyes LE, Verbitsky M, Blesa J, Jackson-Lewis V, Paredes D, Tillack K, Phani S, Kramer ER, Przedborski S, Kottmann AH.
Journal: Neuron. 2012 Jul 26;75(2):306-19.
PMID: 22841315 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported that interruption of Sonic Hedgehog signaling in the adult brain caused progressive degeneration of not just dopamine neurons, but also other types of neurons (cholinergic and fast spiking GABAergic neurons). It also resulted in motor deficits in mice that were reminiscent of Parkinson’s.
Has anyone ever developed drugs for targetting Sonic Hedgehog in the brain?
Most of the research on Sonic Hedgehog has been focused on blocking it. This situation is due to Sonic Hedgehog’s role in cancer.
For example, Vimodegib (or Erivedge) is a cancer drug that blocks the activation of the Sonic Hedgehog pathway (by anatagonising Smoothened – the receptor of sonic).
But there has been some compounds that have been identified as activators of the Sonic Hedgehog pathway, and some of these have been tested in models of Parkinson’s. For example, this recent report:
 Title: Activation of Sonic hedgehog signal by Purmorphamine, in a mouse model of Parkinson’s disease, protects dopaminergic neurons and attenuates inflammatory response by mediating PI3K/AKt signaling pathway
Title: Activation of Sonic hedgehog signal by Purmorphamine, in a mouse model of Parkinson’s disease, protects dopaminergic neurons and attenuates inflammatory response by mediating PI3K/AKt signaling pathway
Authors: Shao S, Wang GL, Raymond C, Deng XH, Zhu XL, Wang D, Hong LP.
Journal: Mol Med Rep. 2017 Aug;16(2):1269-1277
PMID: 28627590 (This report is OPEN ACCESS if you would like to read it)
In this study, the investigators reported that Purmorphamine – which is was the first small-molecule activator (or agonist) of the protein Smoothened (Click here to read more about this compound) – exhibited both anti-inflammatory and neuroprotective effects on cells grown in culture and in a mouse model of Parkinson’s.
Has Purmorphamine ever been clinically tested?
Not that I am aware of.
But directly targetting activation of Sonic Hedgehog may be delicate given the role this protein takes in certain cancers.
In addition, it may not be necessary to directly activate Sonic Hedgehog in order to get benefits. We may be able to improve Sonic Hedgehog activity by targetting other proteins, some of which are associated with Parkinson’s.
What do you mean?
Late last year, this report was published:
 Title: A pathway for Parkinson’s Disease LRRK2 kinase to block primary cilia and Sonic hedgehog signaling in the brain.
Title: A pathway for Parkinson’s Disease LRRK2 kinase to block primary cilia and Sonic hedgehog signaling in the brain.
Authors: Dhekne HS, Yanatori I, Gomez RC, Tonelli F, Diez F, Schüle B, Steger M, Alessi DR, Pfeffer SR.
Journal: Elife. 2018 Nov 6;7. pii: e40202.
PMID: 30398148 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported an interesting connection between Sonic Hedgehog and Parkinson’s associated LRRK2.
What is LRRK2?
Leucine-rich repeat kinase 2 (or LRRK2 – pronounced ‘lark 2’) – also known as ‘Dardarin‘ (from the Basque word “dardara” which means “trembling”) – is an enzyme that has many functions within a cell – from supporting efforts to move things around inside the cell to helping to keep the power on (involved with mitochondrial function).
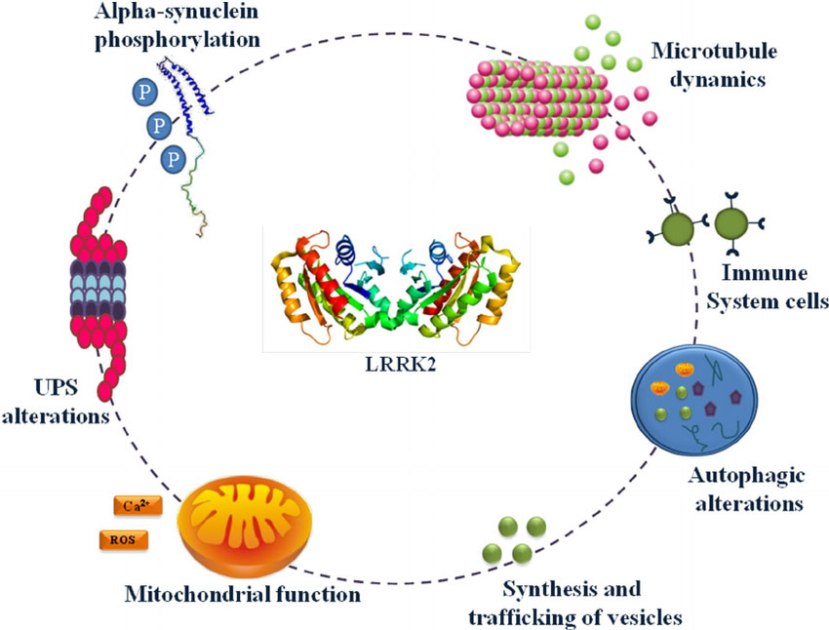
The many jobs of LRRK2. Source: Researchgate
The LRRK2 gene is made up of many different regions. Each of those regions is involved with the different functions of the eventual protein. As you can see in the image below, the regions of the LRRK2 gene have a variety of different functions:
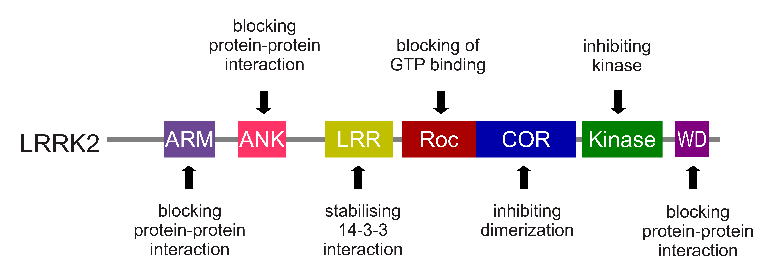
The regions and associated functions of the LRRK2 gene. Source: Intechopen
Genetic errors or variations within the LRRK2 gene are recognised as being some of the most common with regards to increasing ones risk of developing Parkinson’s (LRRK2 variants are present in approximately 1-2% of all cases of Parkinson’s).
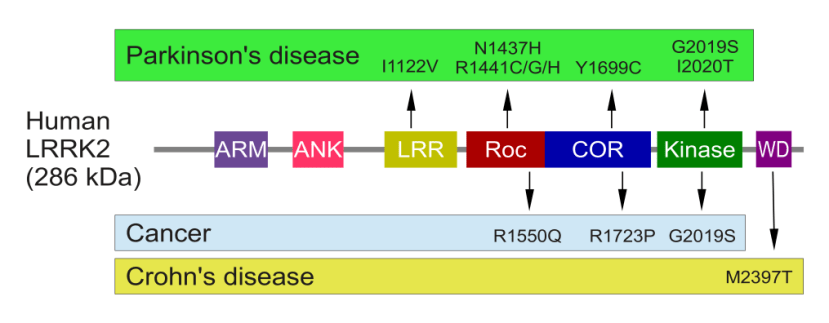
The structure of Lrrk2 and where various mutations lie. Source: Intech
As the image above suggests, mutations in the PARK8 gene are also associated with Crohn’s disease (Click here and here for more on this) – though that mutation is in a different location to those associated with Parkinson’s. And one particularly common Parkinson’s-associated LRRK2 mutation – called G2019S – is also associated with increased risk of certain types of cancer, especially for hormone-related cancer and breast cancer in women – Click here to read more about this. If you have a G2019S mutation, no reason to panic – but it is good to be aware of this association and have regular check ups.
The G2019S mutation (the name designates its location on the gene) is the most common LRRK2 mutation. In some populations of people it can be found in 40% of people with Parkinson’s (Click here to read more about this). But what is interesting about this mutation is that it gives rise to a LRRK2 enzyme that is hyperactive.
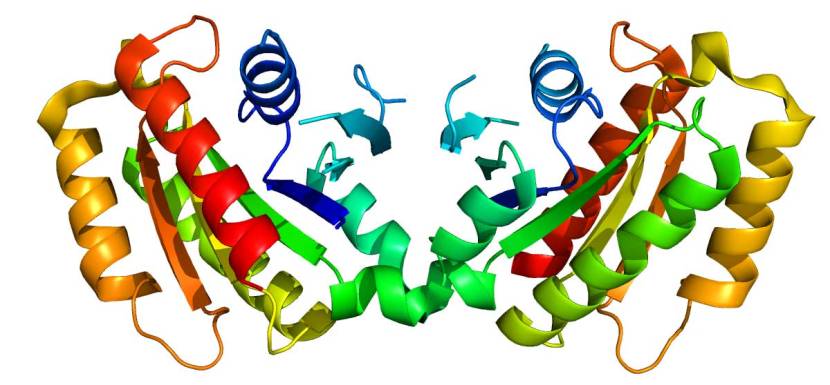
The structure of LRRK2 protein. Source: Wikipedia
As a protein, LRRK2 interacts with many different types of other proteins, and you can imagine that in a finely balanced environment like the cells that a mutant ‘hyperactive’ form of LRRK2 is going to cause problems. The consequences of this constantly active form of LRRK2 protein is believed to be influential in the cell death in LRRK2-associated Parkinson’s.
Thus, biotech companies have been looking for compounds that help to inhibit or modulate LRRK2 activity.
Interesting. So what did the researchers report about LRRK2 and Sonic Hedgehog?
The researchers reported that adult dopamine neurons secret Sonic Hedgehog, which in turn causes a different cell type (the Cholinergic neurons in the striatum) to release the neuroprotective protein GDNF (or glial cell line-derived neurotrophic factor – Click here to read more about this). This forms a feed-back survival loop: the dopamine neurons provide cholinergic neurons with Sonic Hedgehog, and the cholinergic neurons provide dopamine neurons with GDNF.
Cholinergic neurons require Sonic Hedgehog in order to survive (Click here to read more about this), and GDNF is a recognised neuroprotective factor for dopamine neurons.
But, the investigators found that the hyperactive mutant forms of the LRRK2 protein reduces the ability of cholinergic neurons to detect Sonic Hedgehog, which in turn reduces the amount of GDNF being released. A reduced ability of cholinergic neurons to sense Sonic Hedgehog in this neuro-protective feed-back loop, could leave the dopamine neurons more vulnerable to stress or shock, as they have less GDNF being released to keep them fit and well.
Thus, rather than directly activating Sonic Hedgehog, an alternative approach could be inhibiting or modulating LRRK2 levels.
Is anyone clinically testing this idea?
The testing of LRRK2 inhibition? Yes. The activation of Sonic Hedgehog? Not that I am aware of.
And the inhibition of LRRK2 could soon become a crowded field.
In December 2018, Denali Therapeutics announced the initiation of the next step in their clinical testing of their LRRK2 inhibitor, DNL-201 (Click here to read the press release).
This study is a 28-day, randomized, placebo controlled Phase 1b clinical trial in people with mild to moderate Parkinson’s, with and without genetic LRRK2 mutations (Click here to read more about the details of the study). The purpose of the study is to evaluate safety, tolerability, pharmacokinetics (the effect of body on the drug) and pharmacodynamics (the effect of drug on the body) of DNL-201. In addition, the researchers will be evaluating target and pathway engagement – using different biomarkers – following multiple oral doses of DNL201 (or placebo). There will also be an assessment of certain clinical endpoints.
The 30 participants enrolled in the study will be randomized to receive either a low dose of DNL201, a high dose of DNL201, or placebo. Both the participants and investigators will be blind to who is receiving which treatment, and the results of the study should be available before the end of 2019 (Click here to read a previous SoPD post about this).
The pharmaceutical company GlaxoSmithKline is also developing a LRRK2 inhibitor programme.
The company signed a 4 years partnership with the DNA analysis company 23andMe in 2018. As part of that deal, GSK is contributing its LRRK2 inhibitor program, and they will be hoping to use 23andMe’s database of people who know their LRRK2 genetic status. Such access may help their Parkinson’s clinical program enroll participants quicker in a potential clinical trial of their LRRK2 inhibitor (Click here to read more about this).
 In addition, GSK has initiated a LRRK2 observation clinical study at King’s College in London. This study will involve 75 participants (25 with LRRK2-associated Parkinson’s, 25 with non-LRRK2 Parkinson’s, and 25 healthy control subjects), and the researchers will be assessing a wide range of biomarkers over 12 months (Click here to read more about this). And click here to read an article by GSK on their efforts in this area.
In addition, GSK has initiated a LRRK2 observation clinical study at King’s College in London. This study will involve 75 participants (25 with LRRK2-associated Parkinson’s, 25 with non-LRRK2 Parkinson’s, and 25 healthy control subjects), and the researchers will be assessing a wide range of biomarkers over 12 months (Click here to read more about this). And click here to read an article by GSK on their efforts in this area.
And still on the LRRK2 front, there are other new entrants in the area of LRRK2 inhibitors, for example Cerevel Therapeutics.
 This is a new biotech firm that was started last year by Bain Capital and the Pharmaceutical company Pfizer (Click here to read more about this). Cerevel has taken on many of the neuroscience treatments that Pfizer was clinically testing until it shut down their neuroscience division in early 2018 (Click here to read more about this). In addition to those clinically tested assets, Cerevel have also quietly added ‘LRRK2 inhibitor’ to their preclinical ‘lead development’ area of research.
This is a new biotech firm that was started last year by Bain Capital and the Pharmaceutical company Pfizer (Click here to read more about this). Cerevel has taken on many of the neuroscience treatments that Pfizer was clinically testing until it shut down their neuroscience division in early 2018 (Click here to read more about this). In addition to those clinically tested assets, Cerevel have also quietly added ‘LRRK2 inhibitor’ to their preclinical ‘lead development’ area of research.
Another biotech firm Servier has recently entered the LRRK2 inhibition area.
In March of this year Seriver announced a strategic R&D partnership with the biopharma company Oncodesign with the goal of developing LRRK2 inhibitors (Click here to read more about this).
Lots of activity in this area.
So what does it all mean?
One of the exciting things about maintaining this website – and about Parkinson’s research at present in general – is that there are soooo many facets to explore. There are an incredible number of avenues of investigation at the moment. Even when considering proteins involved in the development of dopamine neurons, Sonic Hedgehog is just one of many being experimentally tested (GDNF is one example which we have discussed above and Nurr1 is another example – click here to read a previous SoPD post on this).
It is also fascinating how these developmental biological pathways overlap amongst themselves, but also with Parkinson’s associated proteins. In today’s post, we have looked at how Sonic Hedgehog and LRRK2 overlap in a manner that could make dopamine neurons in the brain more vulnerable. And while researchers seek activators of Sonic Hedgehog activity, biotech firms are investigating how to inhibit LRRK2 activity in an effort to slow the progression of Parkinson’s.
All very exciting stuff.
But this has been a long post, and if you’ll excuse me I need to give my “lunatic finge” and “head case” genes a break and get some sleep.
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
In addition, many of the companies mentioned in this post are publicly traded companies. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. None of the companies have requested that this material be produced, nor has the author had any contact with any of the companies or associated parties. This post has been produced for educational purposes only.
The banner for today’s post was sourced from gamewpp





interestingly written!
LikeLike
Thanks DKDC – glad you liked it!
LikeLike