|
Increasing preclinical evidence is being presented that suggests the gastrointestinal system can play a role in models of Parkinson’s. In addition, there is mounting epidemiological data indicating that the gut can have some kind of influence in people with the condition. Recently, a new paper was published which explores the involvement of the vagus nerve. This is the bundle of nerves connecting the gut to the brain. Specifically, the researchers cut the vagus nerve in mice who had the Parkinson’s associated protein alpha synuclein introduced to their guts, and they found that these mice did not develop the characteristics of Parkinson’s, while those mice with intact vagus nerves did. In today’s post, we will discuss this new report, review some of the additional preclinical and epidemiological data, and try to understand what it all means for our understanding of Parkinson’s.
|
Source: Originubud
Today’s post is about the origin of things. Specifically, Parkinson’s.
But we will begins with words: Consider for a moment the title of this post: Viva las vagus.
When most people read of the word ‘Viva‘, they think of it as a call to cheer or applaud somthing (for example: “Vive la France!” or “Viva las Vegas”), but the origin of the word has a slightly different meaning.
Viva is a shortening of the Latin term viva voce, meaning “live voice”. And in this context it refers to an oral examination – typically for an academic qualification. For example, a European PhD examination is referred to as “viva” and it is an oral denfense (sometimes public – eek!) of the thesis.
 A PhD viva examination. Source: Guardian
A PhD viva examination. Source: Guardian
‘Las’ is simply the Spanish word for ‘the’. And the word ‘vagus’ originates from the Latin, meaning ‘wandering, uncertain’.
Thus, the title of today’s post could – in effect – be “an examination of the uncertain”.
In anatomy and medicine the word, Vagus also refers to an important part of our nervous system.
Which is?
The vagus nerve is the longest of the 12 cranial nerves – these are the nerves which emerge directly from the brain and brainstem, in contrast to the spinal cord. These cranial nerves provide motor and sensory information mainly to and from structures within the head and neck. The olfactory nerve, for example, conveys the sense of smell.
The vagus nerve is slightly different from the other 11 cranial nerves in that it connects with organs outside of the head and neck. It literally ‘wanders’ through the body, and makes connections with many important organs, from the heart to the kidneys:
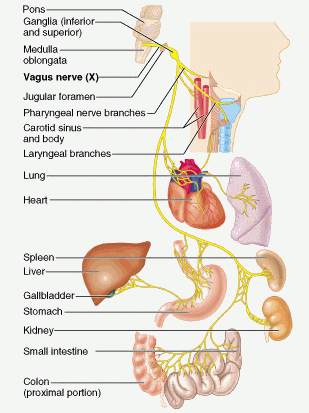 The vagus nerve. Source: Edsinfo
The vagus nerve. Source: Edsinfo
As a result of these connections, the vagus is involved with the control many critical functions of the human body.
And what does any of this have to do with Parkinson’s?
This is Prof Heiko Braak:
 Prof Heiko Braak. Source – Memim.com
Prof Heiko Braak. Source – Memim.com
Many years ago, Prof Braak – a German neuroanatomist – sat down and examined hundreds of postmortem brains from people with Parkinson’s.
He had collected brains from people at different stages of Parkinson’s and was looking for any kind of pattern that might explain where and how the condition starts. His research led to what is referred to as the Braak stages of Parkinson’s – a six step explanation of how the condition spreads up from the brain stem and into the rest of the brain (Click here to read more about this).
 The Braak stages of PD. Source: Nature
The Braak stages of PD. Source: Nature
Braak’s results also led him to propose that Parkinson’s may actually begin in the gut and then spread to the central nervous system (the brain). He based this on the observation that many brains that exhibited the very early stages of Parkinson’s had disease-related pathology in a population of neurons called the dorsal motor nucleus of the vagus nerve.
Yes – that vagus nerve.
This observation led Braak and his colleagues to inspect the vagus nerve and the mesh-like nerve fibres lining the gastrointestinal system (or the gut).
The nerves surrounding the gut are collectively referred to as the enteric nervous system, and they are connected to the brain via vagus nerve (yes again – that vagus nerve).
 The vagus/vagal nerve connection with the enteric nervous system. Source: Nature
The vagus/vagal nerve connection with the enteric nervous system. Source: Nature
Braak and his colleagues found large deposits of the Parkinson’s-associated protein called alpha synuclein (Click here to learn more about this protein) nerves surrounding the gut and the vagus nerve. These deposits were present even at very early stages of the condition, which supported his theory that maybe the disease was starting (or ‘originating’) in the gut.
This ‘gut to brain’ theory was supported by the fact that people with Parkinson’s often complaining of gastrointestinal problems (eg. constipation) and some of these issues have been suggested to predate the onset of other Parkinson’s symptoms.
In addtion, the theory received a further boost from research suggesting that severing the vagus nerve from the gut reduces the risk of developing Parkinson’s:
 Title: Vagotomy and Subsequent Risk of Parkinson’s Disease.
Title: Vagotomy and Subsequent Risk of Parkinson’s Disease.
Authors: Svensson E, Horváth-Puhó E, Thomsen RW, Djurhuus JC, Pedersen L, Borghammer P, Sørensen HT.
Journal: Annals of Neurology, 2015, May 29. doi: 10.1002/ana.24448.
PMID: 26031848
In this study, a group of Danish researchers looked at the medical reports of all the people in Denmark that received a vagotomy between 1975 and 1995.
So what’s a vagotomy?
A vagotomy is a surgical procedure in which the vagus nerve are cut. It is typically done to help treat stomach ulcers. A vagotomy can be ‘truncal‘ (in which the main nerve is cut) or ‘superselective’ (in which specific branches of the nerve are cut, which the main nerve is left in tact).
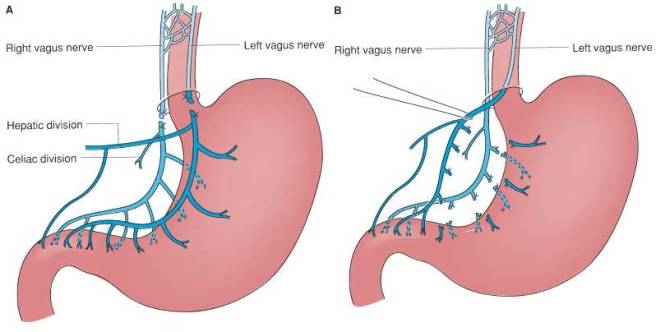 A ‘truncal’ vagotomy (left) & a ‘superselective’ vagotomy (right). Source: Score
A ‘truncal’ vagotomy (left) & a ‘superselective’ vagotomy (right). Source: Score
And what did the Danish scientists find?
The Danish researcher found that between 1975 and 1995, 5339 individuals had a truncal vagotomy and 5870 had superselective vagotomy. Using the Danish National registry (which which stores everyone’s medical information), they then looked for how many of these individuals went on to be diagnosed with Parkinson’s. They compared these vagotomy subjects with more than 60,000 randomly-selected, age-matched controls.
The investigators found that subjects who had a superselective vagotomy had the same chance of developing Parkinson’s as anyone else in the general public.
But when they looked at the number of people in the truncal vagotomy group who were later diagnosed with Parkinson’s, the risk had dropped by 35%. Further, when they followed up the ‘truncal group’ 20 years later, checking to see who had been diagnosed with Parkinson’s in 2012, they found that they rate was half that of both the superselective group and the control group (see table below – HR=0.53).
 Source: Annals
Source: Annals
The researchers concluded that a truncal vagotomy reduces the risk of developing Parkinson’s.
Wow! Should I put a vagotomy on my list of things to do today?
Uh,…no.
I’m not sure I would do that.
Why not?
A different group of Danish researchers analysed a larger pool of Danish medical data and did not find that vagotomy reduces the risk of developing Parkinson’s. They found trends towards reduced risk, but the statistics did not reach significance (Click here to learn more about that).
In addition, since the original Danish report was published some Swedish researchers conducted a similar analysis of Swedish medical data, and they also found that vagotomy was not associated with lower risk of developing Parkinson’s:
 Title: Vagotomy and Parkinson disease: A Swedish register-based matched-cohort study
Title: Vagotomy and Parkinson disease: A Swedish register-based matched-cohort study
Authors: Liu B, Fang F, Pedersen NL, Tillander A, Ludvigsson JF, Ekbom A, Svenningsson P, Chen H, Wirdefeldt K.
Journal: Neurology. 2017, 23;88(21):1996-2002.
PMID: 28446653 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers collected information regarding 8,279 individuals born in Sweden between 1880 and 1970 who underwent vagotomy between 1964 and 2010 (3,245 truncal and 5,029 selective). For each vagotomized individual, they collected medical information for 40 control subjects matched for sex and year of birth (at the date of surgery).
The vagotomy group presented only a 4% reduction in cases of Parkinson’s (hazard ratio (or HR) of 0.96; ranging between 0.78-1.17).
Truncal vagotomy was associated with a lower risk more than five years after the surgery, but that result was not statistically significant (HR of 0.78; ranging between 0.55-1.09). The researchers suggested that the findings needs to be verified in larger samples. Given that their results are not statistically significant, the investigators could only suggest that there may be a trend towards truncal vagotomy lowering the risk of Parkinson’s.
What are the differences between the studies?
The Danish researcher analysed medical records between 1975 and 1995 from 5339 individuals had a truncal vagotomy and 5870 had superselective vagotomy. The Sweds on the other hand, looked over a longer period of time (1964 – 2010) but at a smaller sample size for the truncal group (3,245 truncal and 5,029 selective). Perhaps if the truncal group in the Swedish study was higher, the trend may have become significant.
But either way, some people who had vagotomies still developed Parkinson’s, suggesting that the condition may be starting elsewhere in the body OR it was initiated in the gut and a later vagotomy had no impact on the course of the condition.
All of this needs to be further investigated.
So I shouldn’t rush out and ask my doctor for a vagotomy?
No, that would not be advised (although I’d love to be a fly on the wall for that conversation!).
While the results of these studies are interesting, we really need a much larger study for definitive conclusions to be made. You see, in the Danish study the number of people that received a truncal vagotomy (total = 5339) who then went on develop Parkinson’s 20 years later was only 10 (compared with 29 in the superselective group). And while that may seem like a big difference between those two numbers, the numbers are still too low to be truly conclusive. We really need the numbers to be in the hundreds to be able to make any serious conclusions.
Plus, it is important to determine whether this result can be replicated in other countries. Or is it simply a Scandinavian trend?
It is also interesting to note that vagotomy does not appear to influence risk of developing dementia (in Taiwan – click here to read more about this).
Has vagotomy ever been investigated in models of Parkinson’s?
Yes, it has.
In fact, very recently this report was published:
 Title: Transneuronal Propagation of Pathologic α-Synuclein from the Gut to the Brain Models Parkinson’s Disease
Title: Transneuronal Propagation of Pathologic α-Synuclein from the Gut to the Brain Models Parkinson’s Disease
Authors: Kim S, Kwon SH, Kam TI, Panicker N, Karuppagounder SS, Lee S, Lee JH, Kim WR, Kook M, Foss CA, Shen C, Lee H, Kulkarni S, Pasricha PJ, Lee G, Pomper MG, Dawson VL, Dawson TM, Ko HS
Journal: Neuron, 2019 Jun 17. pii: S0896-6273(19)30488-X.
PMID: 31255487
In this study, the researchers began their study by injecting preformed mouse alpha synuclein fibrils into the nerves surrounding the gut of mice and then waiting to see if the protein could be transported to the brain.
Wait a minute. What are alpha synuclein fibrils?
Alpha synuclein sounds like a distant galaxy, but it is one of the most common proteins in our brains. It makes up about 1% of all the protein in a neuron. When alpha synuclein protein is produced by a cell, it normally referred as a ‘natively unfolded protein’, in that is does not really have a defined structure.
When it is first produced, alpha synuclein will look something like this:

Alpha synuclein. Source: Wikipedia
In this form, alpha synuclein is considered a monomer – which is a single molecule that can bind to other molecules. When it does bind to other alpha synuclein proteins, they form an oligomer (a collection of a certain number of monomers in a specific structure). It is believed that alpha synuclein has certain functions as a monomer, but may also have specific tasks as an oligomer.
In Parkinson’s, alpha synuclein will also misfold and aggregate together to form fibrils.

Microscopic images of monomers, oligomers and fibrils. Source: Brain
And it is believed that the oligomer and fibril forms of alpha synuclein protein that aggregate together and become toxic. They are also believed to then go on to form what we call Lewy bodies.
 Parkinson’s associated alpha synuclein. Source: Nature
Parkinson’s associated alpha synuclein. Source: Nature
A Lewy body is referred to as a cellular inclusion, as they are almost always found inside the cell body. They are a characterisitic feature of the Parkinsonian brain.
 A photo of a Lewy body inside of a neuron. Source: Neuropathology-web
A photo of a Lewy body inside of a neuron. Source: Neuropathology-web
Ok. Got it. Fibrils are a toxic form of the alpha synuclein protein. So what happened when the researchers injected alpha synuclein fibrils into nerves surrounding the mouse gut? What happened in vagus?
A small amount of preformed alpha synuclein fibrils (2.5ul) was injected into the muscle layers of the gut, which are innervated by the branches of the vagus nerve.
One month after that injection, alpha synuclein could be found in the dorsal motor nucleus of the vagus nerve in the brainstem. By 3 months post injection, alpha synclein was starting to accumulate in the substantia nigra (the region of the brain where dopamine neurons reside; an area of the brain severely affected by Parkinson’s). At 7 months, the researchers noted the loss of dopamine neurons in the substantia nigra and motor problems were apparent. And by 10 months after injection, wider regions of the brain exhibited alpha synuclein accumulation and further loss of dopamine neurons was reported. Additional motor/behaviour issues also became apparent, such as a loss of sense of smell and nest building deficits.
As a control experiment, the researchers injected alpha synuclein monomers into the muscle of the gut and reported that at no point was alpha synuclein accumulation observed in the brain.
Did they try a vagotomy on the mice?
They did.
They gave a seperate group of mice truncal vagotomies and conducted the same experiment on them. They found that 7 months after injection and vagotomy there was no alpha synuclein accumulation in the substantia nigra (and no loss of dopamine neurons).
Interestingly, the researchers also repeated the experiment in mice with no alpha synuclein (they have been genetically engineered not to produce alpha synuclein). In the absense of alpha synuclein in the mice, the researchers reported that they failed to see any accumulation of protein at 7 months after injecting alpha synuclein in the muscles of the gut (this is in intact mice; no vagotomy). This finding suggested to the scientists that alpha synuclein must be present in the cells for the accumulation to occur – a finding which is similar to research by Scottish researchers in embryonic stem cells (Click here to read more about that research and click here to read a SoPD post about this).
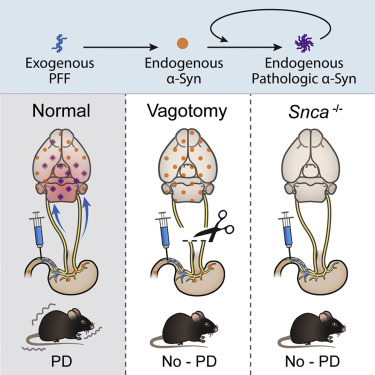 Source: Cell
Source: Cell
The researchers concluded that this new model of Parkinson’s implicates the gut-brain connection in the development of the condition and also supports the Braak hypothesis of Parkinson’s originating in the gut.
And this report is supported by previously published research demonstrating very similar results (Click here to read that research), as well as by more recent research which was published shortly after the vagotomy study was published.
Specifically, this study:
 Title: Evidence for bidirectional and trans-synaptic parasympathetic and sympathetic propagation of alpha-synuclein in rats
Title: Evidence for bidirectional and trans-synaptic parasympathetic and sympathetic propagation of alpha-synuclein in rats
Authors: Van Den Berge N, Ferreira N, Gram H, Mikkelsen TW, Alstrup AKO, Casadei N, Tsung-Pin P, Riess O, Nyengaard JR, Tamgüney G, Jensen PH, Borghammer P.
Journal: Acta Neuropathol. 2019 Jun 26. [Epub ahead of print]
PMID: 31254094 (This report is OPEN ACCESS if you would like to read it)
In this study, Danish researchers directly injected preformed human alpha synuclein fibrils into the gut of rats and observed alpha synclein deposits gradually accumulate in the brain (4 months post injection). Crucial to this study is that some of the rats involved in the study had been genetically engineered to produce human alpha synuclein in the cells of their body. Normal rats injected in the gut with preformed human alpha synuclein fibrils did not develop alpha synuclein deposits.
But this was not the most interesting finding of this particular study.
You see, given the wide range of organs that the vagus nerve makes connections with, the researchers decided to look elsewhere in the body (not just the brain). And what did they find?
They found alpha synuclein deposits in many different regions, including the heart:
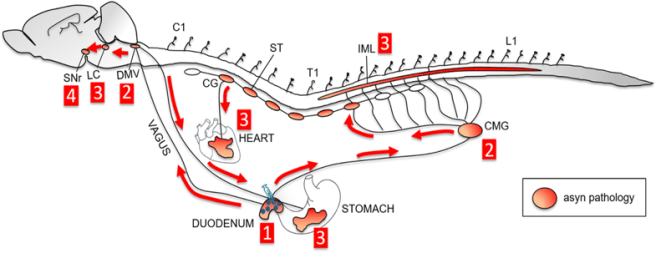 Source: Springer
Source: Springer
This finding suggested to the scientists that accumulating alpha synuclein deposits is not simply spreading up the vagus nerve to the dorsal motor nucleus of the vagus, but also spreading to other organs connected to the vagus nerve (anterogradely).
And this finding is supported by previous research from Prof Haiko Braak demonstrating alpha synuclein in the nerve fibres surrounding the heart (Click here to read more about this).
Um, this is making me seriously think about getting a vagotomy. Why should I not?
It is important to understand that a vagotomy can have very negative side-effects, such as vomiting and diarrhoea (Click here to read more on this). It is an invasive surgery with serious consequences.
In addition, there is scant evidence that a vagotomy will actually slow/stop Parkinson’s once the condition has been diagnosed.
And finally, we now have evidence that some cases of Parkinson’s do not have evidence of alpha synuclein deposits. Thus, a vagotomy seems rather extreme if one’s Parkinson’s does not involve alpha synuclein.
So if I shouldn’t get a vagotomy, what should I do?
Keep an eye on the Enterin clinical trials.
The what?
There is an experimental drug called ENT-01 which prevents alpha synuclein from clustering and forming the toxic form of the protein (Click here to learn more about this protein).
ENT-01 is currently being clinically tested a company called Enterin Inc.
 In 2017-2018, the company conducted the RASMET study, which was a Phase I safety clinical trial of ENT-01. ENT-01 is a derivative of a compound called squalamine (Click here to read a SoPD post on this topic).
In 2017-2018, the company conducted the RASMET study, which was a Phase I safety clinical trial of ENT-01. ENT-01 is a derivative of a compound called squalamine (Click here to read a SoPD post on this topic).
The issue with ENT-01 is that it does not cross the blood brain barrier – the membrane that surrounds the brain, protecting it from the nasty outside world. Thus, Enterin are focusing their clinical trial on Parkinson’s-associated constipation – can this drug reduce alpha synuclein aggregation in the gut and alleviate complaints like constipation.
We are still waiting to hear the results of the RASMET study (Click here for the details of the study), but the company announced the completion of enrollment in a Phase II ‘KARMET’ clinical study of ENT-01 in March of last year (Click here to read the press release). This kind of progress suggests that the safety profile of the drug was acceptable for continuing on with the project. The Phase II study is scheduled to finish in late 2019 (Click here to read more about the trial). Hopefully we should get some news in 2019 regarding this set of clinical trials.
But such a treatment may be a better way of limiting the amount of alpha synuclein making its way from the gut to the brain.
And if the ENT-01 proves unsuccessful for whatever reason, then there are a growing collection of immunotherapy approaches being clinically tested that should robustly remove aggregating alpha synuclein (Click here to read a previous SoPD post on this topic).
Certainly better than having a vagotomy!
So what does it all mean?
Late last year, research was published which suggested that removal of the appendix reduces one’s risk of developing Parkinson’s (if one lives in rural areas – click here to read more about this). More recently, data presented at a scientific research conference suggested that having your appendix removed makes it three times more likely to be diagnosed with Parkinson’s at some point in your life (Click here to read about this – seriously, when did press releases before peer-review become a thing?!?).
My point here is that we must be careful with how we interpret data and what actions we take based on that data. An appendectomy (removal of the appendix) and vagotomy are serious, invasive procedures. And until we have a clearer idea of what impact the gut may or may not be having on Parkinson’s, we should not be considering such drastic measures as potential therapeutic interventions.
These are very exciting times for gut research in neurodegeneration (not just Parkinson’s) – not a week goes by when some new peice of Parkinson’s gut research is being published. It is marvellous that we are acknowledging the importance of the organ and its inhabitants on our basic wellbeing and functioning.
 Source: HuffingtonPost
Source: HuffingtonPost
In addition, there was a number of gut-targetting therapies being developed for conditions like Parkinson’s, such as the Entrin drug that is being clinically tested. If indeed the gut is found to be influential in Parkinson’s (in some individuals), these approaches may provide a powerful new method of treatment.
As I say, keep an eye on the Enterin clinical trial.
Addendum – 27/8/2019
So the results of the Enterin multicenter, dose-escalating RASMET study to evaluate the safety, tolerability & efficacy of orally administered ENT-01 for the treatment of constipation in Parkinson’s have been published… and they look promising (Click here to read them and click here to read the press release).
This was an open label trial, but 80%+ participants achieved the primary efficacy endpoint (with the mean number of complete spontaneous bowel movement per week increasing from 1.2 at baseline to 3.6 during fixed dosing. This is very encouraging.
How the drug was having its effect is to be determined – the systemic absorption was <0.3%, meaning that very little of the drug was being absorbed into the body. The researcher suggest that ENT-01 was acting locally on the enteric nerve fibres. If I have time in coming weeks, I’ll try to write a post about these results. It will be interesting to see how the ongoing clinical trial of ENT-01 fares.
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by a research scientist, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. Please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from ifarmusiqueconcretecompilation


Love the title of this post! And a superb write-up as usual. (long-time lurker here!)
I wonder what you think of the usage of these pre-formed fibrils? It seems to me they were initially developed as a technical aide – to quickly kick-start protein aggregation that was otherwise painfully slow or absent in animal models. But now it seems to have become so commonly used and we are building so much basic science on the basis of phenotypes seen from injecting (often large amounts) of these artificial protein aggregates.
And if I remember correctly, they didn’t actually test for any actual synuclein aggregation in the brain in this model. Would have been nice to see, right?
LikeLike
Hi Vinoy,
Thanks for your interesting comment – glad you liked the post/title. I have to be careful what I say here, but I am a bit weary about the ‘in vogue’ use of preformed fibrils as well. Herd mentality is a live and well. I wonder in 100 years time when AI has solved all the problems of the world and we have a thorough understanding of Parkinson’s, where will preformed fibrils stand in the history books? It is late and I better stop there.
Keep lurking.
Kind regards,
Simon
LikeLike
Thank you for making the science accessible to the educated non-scientist. Your visuals and conversational style make it easier to read and more personally relevant
LikeLike
Hi Macgreene,
Thanks for your kind comment – much appreciated. And you are very welcome. Glad you like the content, please let me know if there is any particular topic you would like explored.
Kind regards,
Simon
LikeLiked by 1 person
Great article again thank you for making the research so accessible. Enterin sounds very exciting.
LikeLike
Hi Richelle,
Thanks for your kind comment – glad you liked the post.
Kind regards,
Simon
LikeLike
Hi Simon,
You wrote : “The issue with ENT-01 is that it does not cross the blood brain barrier …”
I noticed that Enterin are also running a trial of orally-administered ENT-01 for Parkinson’s disease dementia (NCT03938922). Given the BBB issue that you have highlighted, what are your thoughts?
https://clinicaltrials.gov/ct2/show/NCT03938922?term=NCT03938922&rank=1
Warm Regards,
Jeff
LikeLike
I think I may have found something relevant, in a recent paper by Michael Zasloff et al.
“Overproduction of αS in the enteric nervous system (ENS) and its chronic trafficking to the CNS may damage nerves and lead to Parkinson’s disease. Targeting the formation of αS aggregates in the ENS may therefore slow the progression of the disease.”
Gastrointestinal Immunity and Alpha-Synuclein (September 2019):
https://content.iospress.com/articles/journal-of-parkinsons-disease/jpd191702
LikeLike