|
Researchers at Cambridge University published a new report this week that extends on a very interesting line of Parkinson’s research. The studies focus on a compound (and derivatives of that compound) that has been derived from the dogfish shark. The protein – called Squalamine – has an amazing ability to prevent the Parkinson’s-associated protein alpha synuclein from clustering (or aggregating) together. The aggregation of alpha synuclein is considered to be a key component of the biology underlying Parkinson’s, and thus any compound that block/reduce this aggregation is viewed with therapeutic applications in mind. Unfortunately there is a problem with squalamine: it does not cross the blood brain barrier (the protective membrane surrounding the brain). But a derivative of squalamine – called Trodusquemine – does! In today’s post, we will look at what Squalamine and Trodusquemine are, we will review the new research, and look at current clinical research efforts involving these compounds. |
The effects of aggregated Alpha Synuclein protein in a neuron. Source: R&D
We often talk about one particular protein on this website. It is called alpha synuclein. It is one of the most common proteins in the human brain, and it appears to be centrally involved with Parkinson’s.
In the Parkinsonian brain, alpha synuclein clumps (or aggregates) together, which is believed to lead to the appearance of Lewy bodies.
What are Lewy bodies?
Lewy bodies are dense circular clusters of alpha synuclein protein (and other proteins) that are found in specific regions of the brain in people with Parkinson’s (Click here for more on Lewy bodies).
A cartoon of a neuron, with the Lewy body indicated within the cell body. Source: Alzheimer’s news
The aggregated alpha synuclein protein, however, is not limited to just the Lewy bodies. In the affected areas of the Parkinsonian brain, aggregated alpha synuclein can be seen in the branches (or neurites; see black arrow in the image below) of cells. In the image below, alpha synuclein has been stained brown on a section of brain from a person with Parkinson’s.
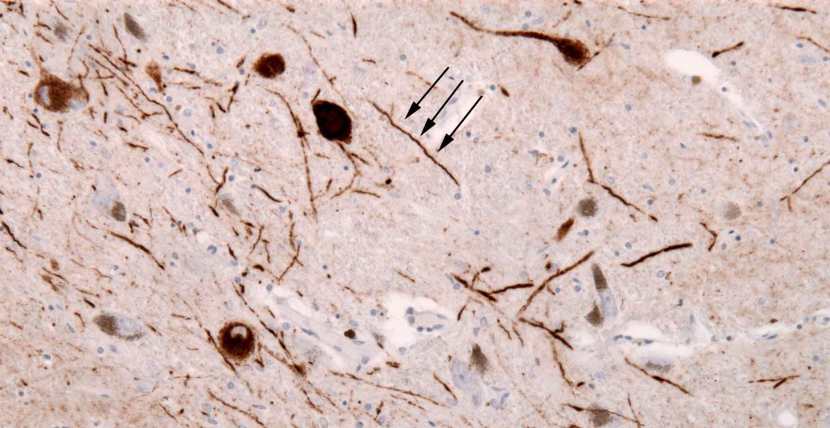
Examples of Lewy neurites (stained in brown; indicated by arrows). Source: Wikimedia
When alpha synuclein protein is produced by a cell, it normally referred as a ‘natively unfolded protein’, in that is does not really have a defined structure. When it is first produced, alpha synuclein will look something like this:
Alpha synuclein. Source: Wikipedia
Like this, alpha synuclein is considered a monomer – which is a single molecule that can bind to other molecules. When it does bind to other alpha synuclein proteins, they form an oligomer (a collection of a certain number of monomers in a specific structure). In Parkinson’s, alpha synuclein also binds (or aggregates) to form what are called ‘fibrils’.

Microscopic images of Monomers, oligomers and fibrils. Source: Brain
And it is believed that the oligomer and fibril forms of alpha synuclein protein give rise to the aggregations of protein that go on to form the Lewy bodies we mentioned above:
Parkinson’s associated alpha synuclein. Source: Nature
Now, given this process, and its association with a neurodegenerative condition like Parkinson’s, a lot of effort is being put into reducing this aggregation of alpha synuclein protein. It is hoped that by limiting this activity, we may be able to slow or stop completely the progression of the condition.
How can we limit protein aggregation?
There are many different ways, and several methods are being currently being clinically tested.
We have recently talked about methods that involve using the immune system to target the aggregated form of alpha synuclein (Click here and here to read about that). Initial results suggest that this approach is safe in humans and we are now waiting to find out if these methods actually work.
In addition, there are also efforts to increase the removal of alpha synuclein protein using mechanisms inside the cells (such as increased autophagy (a waste disposal/recycling system). And there are currently drugs being clinically tested for this approach (Click here to read a recent post about one of these sorts of drugs).
And this week, new research was published which identified another approach that could be useful in reducing the amount of alpha synuclein aggregation. But this method is slightly different: this new compound stops the protein from actually aggregating.
Here is the research report:
Title: Multistep Inhibition of α-Synuclein Aggregation and Toxicity in Vitro and in Vivo by Trodusquemine.
Authors: Perni M, Flagmeier P, Limbocker R, Cascella R, Aprile FA, Galvagnion C, Heller GT, Meisl G, Chen SW, Kumita JR, Challa PK, Kirkegaard JB, Cohen SIA, Mannini B, Barbut D, Nollen EAA, Cecchi C, Cremades N, Knowles TPJ, Chiti F, Zasloff M, Vendruscolo M, Dobson CM.
Journal: ACS Chem Biol. 2018 Jun 28. doi: 10.1021/acschembio.8b00466.
PMID: 29953201
In this study, the researchers discovered that a derivative of Squalamine – called Trodusquemine – has some very interesting properties concerning the aggregation of alpha synuclein.
Hang on a minute! What is Squalamine?
Squalamine is a compound with a wide range of antimicrobial activity. It was originally discovered in dogfish sharks.
The spiny dogfish shark. Source: Discovery
An interesting bit of pub-quiz information for you regarding dogfish sharks:
They are extremely robust when it comes to infection.
They really don’t get sick all that often. And this is despite the fact that dogfish sharks have a relatively “primitive” immune system (Click here to read more on this). A research team led by Prof Michael Zasloff (of Georgetown University) discovered that a chemical called ‘Squalamine’ may be one of the reasons for this robustness of health in the dogfish shark.
Prof Michael Zasloff. Source: Nextbigfuture
But more recently, Prof Zasloff and co. discovered something else very interesting about Squalamine: it has a potent effect on alpha synuclein aggregation.
They published the results of their research on this topic last year (2017):

Title: A natural product inhibits the initiation of α-synuclein aggregation and suppresses its toxicity
Authors: Perni M, Galvagnion C, Maltsev A, Meisl G, Müller MB, Challa PK, Kirkegaard JB, Flagmeier P, Cohen SI, Cascella R, Chen SW, Limboker R, Sormanni P, Heller GT, Aprile FA, Cremades N, Cecchi C, Chiti F, Nollen EA, Knowles TP, Vendruscolo M, Bax A, Zasloff M, Dobson CM.
Journal: PNAS 2017 Feb 7;114(6):E1009-E1017.
PMID: 28096355 (this article is OPEN ACCESS if you would like to read it)
In this study, the researchers discovered that squalamine can actually block alpha synuclein from aggregating. They treated human cells (that produce too much alpha synuclein, which ultimately kills them) in culture with squalamine and they observed an almost complete suppression of the toxic effect of alpha synuclein.

Caenorhabditis elegans – cute huh? Source: Nematode
The researchers next looked at the effects of squalamine in a microscopic worm called Caenorhabditis elegans . These tiny creatures are widely used in biology because they can be easily genetically manipulated and their nervous system is very simple and well mapped out (they have just 302 neurons and 56 glial cells!). The particular strain of Caenorhabditis elegans used in this particular study produced enormous amounts of alpha synuclein, which results in muscle paralysis.
By treating the worms with squalamine, the researchers observed a dramatic reduction of alpha synuclein protein aggregating and an almost complete elimination of the muscle paralysis. In addition, they noted a reduction in the cellular damage caused by the aggregation of alpha synuclein.
All in all, a pretty impression result! The researchers suggested that their findings indicate that “squalamine could be a means of therapeutic intervention in Parkinson’s”.
Wow. Where can I get me some of that squalamine stuff?
Yeah,… about that. Before you get too excited, there is one small problem:
Squalamine does not cross the blood brain barrier.
What is the blood brain barrier?
The blood brain barrier is a membrane that surrounds the brain, protecting it from the nasty outside world.
This video provides a basic explanation of the blood brain barrier:
And if squalamine can not cross the blood brain barrier, it can not have any effect on the cells inside the brain.
Not very useful for a neurological condition like Parkinson’s.
Back to the drawing board then?
Well, no. This is where the new research from some Cambridge university scientists is so interesting.
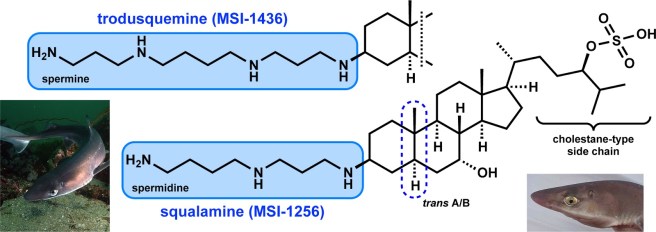
They have looked at a derivative of squalamine, which is called Trodusquemine, which DOES cross the blood brain barrier. And they have discovered that Trodusquemine displays the same positive benefits of squalamine… plus a few more!
You have my attention. Please tell me more. What is Trodusquemine?
Like squalamine, trodusquemine is an aminosterol – especially any of a group of such antibiotics obtained from sharks. It is primarily viewed as an inhibitor of a protein called PTP1B.
PTP1B is a negative regulator of the insulin signaling pathway – that is, PTP1B reduces the levels of insulin in the blood stream. Given this function, PTP1B is widely considered a promising therapeutic target for the treatment of type 2 diabetes (Click here and here to read more about this)
Trodusquemine has a very similar chemical structure to squalamine:
Source: Modernsteroid
And as I suggested above, there is evidence that trodusquemine can cross the blood brain barrier (Click here and here to read more about this).
Ok, so what does the new research published this week suggest?
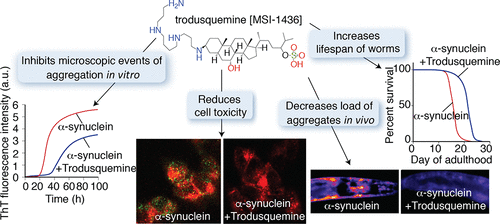
Given that trodusquemine can enter the brain, the investigators wanted to determine if this protein had similar properties to squalamine. Firstly, the researchers investigated the effect of trodusquemine treatment on synuclein aggregation under a series of specifically designed conditions that make it possible to characterise each step in the processes of aggregation (for example, the binding of monomers, the formation of oligomers, and clustering of fibrils).
The results of this analysis suggested that trodusquemine – like squalamine – can disrupt the initial stage of protein aggregation (by displacing monomeric alpha synuclein from the cell membrane). BUT unlike squalamine, trodusquemine can also suppress the interaction of alpha synuclein oligomers and fibrils (the later stages of the aggregation process).
As a results of this protein aggregation inhibiting activity, the researchers found that trodusquemine suppresses the toxicity of alpha synuclein oligomers in human cells grown in culture. And when they conducted experiments on the alpha synuclein-producing Caenorhabditis elegans (the tiny worms mentioned above), the investigators found that trodusquemine could inhibit the formation of the alpha synuclein aggregates and increase the fitness and longevity of the worms.
Source: Pubs
The only missing component from this study was the use of trodusquemine is larger animals (eg. mice), but we can be sure that the researchers are currently conducting those experiments.
The researchers concluded that “trodusquemine appears to be able to displace alpha synuclein and its oligomers from the membrane, inhibiting both the lipid-induced initiation of the aggregation process and the ability of the oligomers to disrupt the integrity of membranes“. And they also noted “the present results suggest that this compound has the potential to be an important therapeutic candidate for Parkinson’s and related disorders“.
Cool. Is Trodusquemine being clinically tested?
It have been clinically tested in humans.
There have been three clinical trials of trodusquemine (and one terminated trial – the study was recently stopped because no one would sponsor it).
The first study was a safety and tolerance assessment in normal healthy individuals (Click here to read more about this study), while the other two were focused on testing trodusquemine in diabetes and obesity (Click here and here to read more about these trials).
Unfortunately, none of the results of these trials have ever been published. And unpublished clinical trial results has become a real problem. One analysis of data collected between January 2006 to November 2014, found that 45% of the 25,927 assessed clinical trials had not had their results published (Click here to read more about this). How are we to learn anything if results do not get published?!?
The trodusquemine (also known as MSI-1436) clinical trials were conducted by a biotech firm called Genaera Corporation (previously known as Magainin Pharmaceuticals), which as subsequently gone out of business. While the company never published the results of these studies, they did present the Phase I safety results at the 2007 North American Association for the Study of Obesity Annual Meeting in New Orleans (Click here to read more about this) and then Phase Ib results from treatment of individuals with type 2 diabetes and obesity (Click here to read more about this).
In both of those presentations, trodusquemine was reported to be well tolerated, and there were no serious adverse events.
Great. Trodusquemine is safe in humans. When can we start a clinical trial?
Well, we firstly need to independently replicate the initial cell culture results and also test trodusquemine in other models of Parkinson’s (mice and primates).
But that will take time?
Yes, but fear not, squalamine-based research is already being conducted in the clinic.
A Boston-based biotech firm called Enterin Inc. is currently testing a synthetic version of squalamine in Parkinson’s.
The company is running the RASMET study which is evaluating safety and tolerability of ENT-01 – the synthetic version of squalamine – as a treatment for constipation in Parkinson’s. According to the company, ENT-01 is not absorbed into the bloodstream. Rather the compound acts locally in the gut, acting on the enteric nerve cells in the lining of the gut wall. It is hoped that this treatment will stimulate gut motility and alter neural signaling from the gut to the brain.
The trial involves 50 people with Parkinson’s (who have had constipation for over 6 months), and it is being conducted in two stages: Phase 1 involved 10 participants and it sought to assess the safety, tolerability, and pharmacokinetics of single escalating doses of ENT-01 over a 30-60 day period. This dose-escalation study involved a pre-treatment, 2-week run in period of assessment and it was followed by a 2-week wash-out period with further assessments. This study has now been completed, though the results have not been released. Given that the company has announced that they have completed the recruitment for the Phase 2a part of the trial (Click here to read the press release), we can make the assumption that the safety profile of the drug is acceptable.
The RASMET study is now conducting the Phase 2a trial, which has enrolled 40 people with Parkinson’s. This study will be composed of 4 periods:
- A 2-week run-in period
- A 3-5 week escalating dose period to identify a prokinetic dose in the initial set of 10 patients
- A 1-week period of randomised dosing (placebo versus the previously identified pro-kinetic dose)
- A 2-week wash-out period.
What is a prokinetic dose?
A prokinetic (also known as a gastroprokinetic or gastrokinetic agent) is a class of drug which enhances gastrointestinal motility by increasing the frequency (or strength) of contractions in the small intestine, but without actually disrupting their rhythm.
In the RASMET study, pharmacodynamics (or the effect of the drug on the body) will be assessed along with safety and tolerability. Relative outcomes will be compared within each patient and across groups. Frequency of bowel movements and other non-motor symptoms of Parkinson’s will be collected over the course of both phases.
The primary outcome measure of the study will be the number of participants with treatment-related adverse events. This is a measure of the safety of the drug.
The secondary outcome measure will be the frequency of bowel movements.
We are hoping to see the results of this trial before the end of the year. And positive safety outcomes from these studies will help in getting trodusquemine into clinical trials.
So what does it all mean?
Last year (2017), researchers reported that a novel experimental Parkinson’s treatment (called Squalamine) displayed amazing properties – being able to inhibit the aggregation of a Parkinson’s-associated protein. But there was just one problem: the compound could not enter the brain.
So the researchers went back to the lab bench and devised a new version of the compound – called trodusquemine – which can enter the brain AND has additional beneficial properties with regards to the aggregation of proteins.
One minor concern with regards to trodusquemine is that previous studies in models of obesity and diabetes suggest that weight loss could be issue with taking this compound forward to the clinic (Click here to read more about this). This should not prevent further investigations of this compound in models of Parkinson’s (and hopefully clinical studies), but it is something to be aware of – especially given that weight loss can be an issue for some folks with PD.
I have often said on this website that the pace of Parkinson’s research is rather incredible at the moment. And the Squalamine story is a great example of this: In February 2017, the first results of this compound in models of Parkinson’s were published, 4 months later a clinical trial was started, and now just 12 months – while we are eagerly waiting for the results of said clinical trial – we have a derivative of squalamine that appears to be even better than the original compound. After 200 years, things are certainly heating up!
And rest assured, we will be on the look out for any new information regarding both the RASMET trial and trodusquemine.
The banner for today’s post was sourced from Wikimedia









another interesting piece by Simon
LikeLike
Thanks PFB, glad you liked it!
Kind regards,
Simon
LikeLike
What is the relationship of shark cartilage and Squalamine?
LikeLike
Does anyone know if squalamine is orally available? All of the research I see on it they are administering it IV or SubQ. Does it make it through the digestive tract intact?
LikeLike
“… (and one terminated trial – the study was recently stopped because no one would sponsor it).”
AAARGH !!!!
LikeLike
There was another limited trial using trodusquemine sponsored by the British Heart Foundation that reports a single dose of it reversed atherosclerosis. I can’t find any evidence of further trials in humans (they used mice), but this was fairly recent. Further searching reveals that it is also being trialled as an appetite suppressant as of 2019/20. And there is some suggestion it is also being looked at for heart failure issues.
LikeLike