|
Each year King’s College London holds the Edmond J. Safra Memorial Lecture. It is a public event – exploring cutting-edge research on Parkinson’s – held in honour of the late philanthropist and financier, Mr Edmond J Safra, . I was lucky enough to attend this year’s event (entitled A vision of tomorrow: How can technology improve diagnosis and treatment for Parkinson’s patients?). It highlighted the fantastic research being carried out by Professor Marios Politis and his team. During the Q&A session of the event though, a question was asked from the audience regarding what the evolutionary advantage of Parkinson’s might be. The question drew a polite chuckle from the audience. But the question wasn’t actually as silly as some might think. In today’s post we look at some evidence suggesting an evolutionary advantage involving Parkinson’s. |
King’s College London Chapel. Source: Schoolapply
Despite the impressive name, King’s College London is not one of the grand old universities of England.
Named after its patron King George IV (1762-1830), the university was only founded in 1829 (compare this with 1096 for Oxford and 1209 for Cambridge; even silly little universities like Harvard date back further – 1636). The university is spread over five separate campuses, geographically spread across London. But if you ever get the chance to visit the main Strand campus, ask for the chapel and take a moment to have a look – it is very impressive (the image above really doesn’t do it justice).
As I mentioned in the intro, each year King’s College London holds the Edmond J. Safra Memorial Lecture. It is an event that is open to the public and it involves a discussion regarding innovative new research on Parkinson’s. The evening is held in honour of the late Mr Edmond J Safra.
Edmond J. Safra. Source: Edmondjsafrafoundation
This year, Professor Marios Politis and members of his research group were presenting lectures on “How can technology improve diagnosis and treatment for Parkinson’s”. The lectures were very interesting, but the reason I am writing about it here is because during the question and answer session at the end of the lectures, the following question was asked:
“What’s the evolutionary advantage of Parkinson’s?”
Given the debilitating features of the condition, the audience was naturally amused by the question. And there was most likely several people present who would have thought the idea of any evolutionary advantage to Parkinson’s a ridiculous concept.
But it’s not.
And there is actually research to suggest that something evolutionary could be happening with Parkinson’s.
?!?!? What do you mean?
Back in 2014, this research report was published:
Title: Abnormal visual gain control in a Parkinson’s disease model.
Authors: Afsari F, Christensen KV, Smith GP, Hentzer M, Nippe OM, Elliott CJ, Wade AR.
Journal: Hum Mol Genet. 2014 Sep 1;23(17):4465-78.
PMID: 24718285 (This article is OPEN ACCESS if you would like to read it)
In this report, the researchers wanted to investigate visual ability in flies that had been genetically engineered so that their dopamine neurons carried the human LRRK2-G2019S genetic variant (we have previously discussed this Parkinson’s associated risk factor – click here to read that post). When the researchers looked at contrast sensitivity in the eyes of these flies, they found something really interesting:
The presence of human LRRK2-G2019S in the dopamine neurons resulted the flies having faster and stronger visual responses in the young stages of life, but this effect was lost and eventually deteriorated as the flies aged.
Impressively, by treating the flies with a LRRK2 inhibitor (BMPPB-32), the researchers restored normal contrast sensitivity in both the young and aged LRRK2-G2019S flies (we have previously discussed the development of LRRK2 inhibitors for Parkinson’s – click here and here to read those posts). This finding indicated that the early hypersensitivity was due to the abnormal (kinase domain) activity of the mutant version of LRRK2 protein.
The investigators were so intrigued with their finding that they decided to investigate whether other Parkinson’s-associated genes could cause similar effects. They published this report a year later:
Title: Classification of Parkinson’s Disease Genotypes in Drosophila Using Spatiotemporal Profiling of Vision.
Authors: West RJ, Elliott CJ, Wade AR.
Journal: Sci Rep. 2015 Nov 24;5:16933.
PMID: 26597171 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers compared flies with genetic mutations associated with early-onset forms of Parkinson’s (PINK1 and DJ-1) with the later-onset Parkinson’s-associated LRRK2. They also compared these results with normal control flies. What they found was that flies with the genetic mutations associated with early-onset forms of Parkinson’s also had elevated levels of visual responses in the young stages of life (a 2x increase in response amplitudes compared with controls).
So the effect was not unique to just LRRK2.
The investigators speculated that the increased level of neuronal activity would probably result in increased energy requirements for the cell (more activity = burning more energy). This need for energy would put pressure on the mitochondria (the power stations of the cell) to supply more energy (in the form of ATP). Increased ATP production, however, would result in the increased production of oxidative stress species, and if this is not carefully controlled for, it could potentially lead to cell death.
They tested this idea in a follow up study that was published recently:
Title: Abnormal visual gain control and excitotoxicity in early-onset Parkinson’s disease Drosophila models.
Authors: Himmelberg MM, West RJH, Elliott CJH, Wade AR.
Journal: J Neurophysiol. 2018 Mar 1;119(3):957-970.
PMID: 29142100 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers took their flies with genetic mutations associated with early-onset forms of Parkinson’s and they generated additional neuronal stress by exposing those flies to randomly pulsating light for 7 days. This increased the level of activity for the cells would put extra pressure on the energy requirements of the cells. And the investigators found that this exposure to the pulsating lights resulted in a profound loss of vision in all of the flies with genetic mutations associated with early-onset forms of Parkinson’s.
This result supports the idea of a ‘excitotoxicity model’ of Parkinson’s in some of these genetic forms of the condition. It also supports the idea that these mutations may be linked to more sensitive neuronal signalling in the early life (or perhaps prodromal stages) of Parkinson’s, which may cause the appearance of Parkinson’s symptoms later in life. And there are there also largely unsubstantiated suggestions that people with genetic variants in the Parkinson’s-associated GBA gene may have early advantages as well (Click here to read more about this). Such advantages could partly explain why some of these genetic mutations (which increase risk of developing Parkinson’s later in life) have lasted so long across man’s history (for example, the LRRK2-G2019S variant has existed for over 2000 years and has two distinct origins – click here to read more about this).
And the investigators from the research report reviewed above also suggested that their findings could explain why we see a higher incidence of Parkinson’s in people with higher socioeconomic status jobs (Click here to read more about this).
What do you mean “we see a higher incidence of Parkinson’s in people with higher socioeconomic status jobs”?
So last year this research report was published:

Title: Mortality from Amyotrophic Lateral Sclerosis and Parkinson’s Disease Among Different Occupation Groups – United States, 1985-2011.
Authors: Beard JD, Steege AL, Ju J, Lu J, Luckhaupt SE, Schubauer-Berigan MK.
Journal: MMWR Morb Mortal Wkly Rep. 2017 Jul 14;66(27):718-722.
PMID: 28704346 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers used data from the CDC’s National Institute for Occupational Safety and Health (NIOSH) database called the National Occupational Mortality Surveillance (NOMS). NOMS is a US-based program that monitors work-related acute and chronic disease mortality among workers in 30 U.S. states. The database contains information associated with approximately 12.1 million deaths.
In total, there were 26,917 deaths associated with Amyotrophic Lateral Sclerosis and 115,262 deaths associated with Parkinson’s. The occupations of the deceased were then grouped into 26 categories based on similarities of job duties and ordered roughly from high socioeconomic status (e.g., legal, finance, management) to lower socioeconomic roles (e.g., construction, transportation and material moving).
After accounting for age, sex, and race, the investigators found that occupations associated with higher socioeconomic status (such as computer and mathematical; Legal; Life, physical, and social sciences) were particularly elevated in people that passed away with Amyotrophic Lateral Sclerosis and Parkinson’s (Click here to see the full list).
And curiously, 11 occupation categories were significantly not associated with Parkinson’s. These included:
- Building and grounds cleaning and maintenance
- Protective service
- Food preparation and serving
- Transportation and material moving
- Installation, maintenance, and repair
And one in particular – called ‘extraction‘ [involving mining or oil and gas drilling] – was particularly not associated with Parkinson’s.
And this finding keeps popping up in other reports:

Title: Occupational complexity and risk of Parkinson’s disease.
Authors: Valdés EG, Andel R, Sieurin J, Feldman AL, Edwards JD, Långström N, Gatz M, Wirdefeldt K.
Journal: PLoS One. 2014 Sep 8;9(9):e106676.
PMID: 25198429 (This article is OPEN ACCESS if you would like to read it)
In this study, the investigators used the Swedish Twin Registry that included 28,778 twins (born between 1886 and 1950). They identified 433 cases of Parkinson’s disease. When they assessed occupations, they found that high occupational complexity (with data and people) was associated with increased risk of Parkinson’s, particularly in men.
And there are previous research reports supporting the idea of more complex occupations being associated with an increased risk of Parkinson’s (Click here and here to see two examples). So perhaps there is some evidence that people at risk of Parkinson’s have intellectual advantages in younger life while they are being educated and trained for a career in a high socioeconomic occupation.
Thus, maybe there is an ‘evolutionary advantage’ of a sort to Parkinson’s.
Unfortunately somebody forgot to tell mother nature that we are all living longer.
What does it all mean?
Charles Darwin. Source: rmg
Charles Darwin didn’t teach us ‘evolution’.
Jean-Baptiste Lamarck can lay claim to that.
Nor did Darwin come up with “Survival of the fittest”. That classic belongs to Herbert Spencer.
No, what Darwin taught us is the importance of context.
It is only when the context of an organisms situation changes that the full outcomes of certain traits become apparent, and either more or less desirable. So, while some of these Parkinson’s-associated genetic mutations could be resulting in faster and stronger neuronal signalling in the first half of a human life span – which could be advantageous – as we have gradually started to live longer (and the context has changed), these traits have become less desirable. During the first centuries BC and the first centuries AD, the average life span was about 35 years, which meant that these the downside to these genetic variants which increase one’s risk of developing Parkinson’s were largely irrelevant.
It will be interesting to see if other research groups can replicate and expand on the genetic findings discussed in this post – perhaps in other parts of the brain or body. In addition, it would be fascinating if this research could be applied to the human situation in some way (I am thinking of induced pluripotent stem cells being generated from people with Parkinson’s-associated genetic mutations, differentiated into dopamine neurons and their electrical activity tracked over time).
But I hope that you will agree having read this post that the question asked at the Edmond J. Safra Memorial Lecture this year, regarding the ‘evolutionary advantage of Parkinson’s’ was not such a silly idea.
And hopefully it will encourage more ‘silly’ questions to be asked.
ADDENDUM: 30th March, 2018
It would appear that this idea of genetic mutations potentially inferring some early life cognitive benefits is not restricted to Parkinson’s. Apparently there is evidence that genetic variants associated with other neurodegenerative conditions also have early beneficial effects.
Huntington’s disease is a neurodegenerative condition which results in individuals losing all inhibition of movement, giving rise to appearance of chorea – jerky, random, and uncontrollable movements (similar to dykinesias).
Huntington’s disease is caused by an increase in a region of DNA inside the Huntingtin gene. This regions is made up of CAG repeats. In normal healthy humans, we usually have up to 30 repeats of CAG. If you have more than 40 CAG repeat, you are definitely going to develop Huntington’s disease.
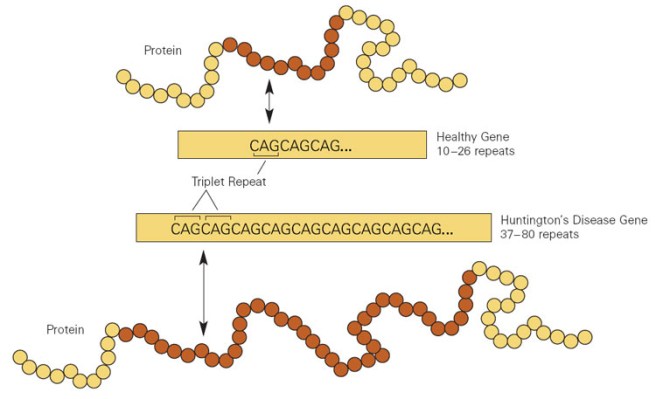
Expansion of CAGs in the Huntingtin gene. Source: NIST
Now, the Huntingtin gene goes back a long way in evolution – it is a very old gene. But there is an interesting trend across evolution: the number of CAG repeats increasd as species got more sophisticated. For example, Dictyostelium discoideum, an amoeba, has the huntingtin gene, but it contains no CAG repeats. The huntingtin gene in Sea urchins have two repeats, the huntingtin gene in zebrafish have four, mice have seven, dogs = ten, monkeys = 15, humans = 27 (on average).
Interesting trend, right?
And there is actually evidence of the number of CAG repeats in the Huntingtin gene having an impact on cognition and motor skills in humans.
Prof Peg Nopoulos (University of Iowa) and her team of researchers assessed the cognitive and motor skills of 80 children aged from six to 18, and comparing those test results and brain scans with the children’s CAG counts. They found a strong association between the number of repeats and how the children performed on each test. The more CAG repeats the child had in their Huntingtin gene, the higher their intelligence score. They also reported a correlation between the number of CAG repeats in the children and the volume of the basal ganglia in their brain – the basal ganglia is a region that is badly affected by Huntingtin’s disease (Source: Economist).
In addition, there was a brain imaging study in adults which found a similar result:
 Title: Variation within the Huntington’s disease gene influences normal brain structure
Title: Variation within the Huntington’s disease gene influences normal brain structure
Authors: Mühlau M, Winkelmann J, Rujescu D, Giegling I, Koutsouleris N, Gaser C, Arsic M, Weindl A, Reiser M, Meisenzahl EM.
Journal: PLoS One. 2012;7(1):e29809.
PMID: 22235343 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers recruited 278 normal healthy subjects, then determined the number of CAG repeats thay each had before imaging their brains (MRI). The investigators found that the putamen (a region of the basal ganglia) increased in size with an increase in the number of CAG repeats.
So perhaps genetic variants associated with other neurodegenerative conditions in addition to Parkinson’s can also have beneficial effects. It’s a really interesting idea. Worthy of further investigation.
The banner for today’s post was sourced from flickr






fun topic!
LikeLike
Fascinating topic! Glad you liked it
LikeLike
I read somewhere that analytic cognitive style, as opposed to habituation, was found more commonly in PD. Maybe that’s an evolutionary adaptation? Or maybe I’m misunderstanding the concept!
LikeLike
Just a thought Simon .. ( you said you would be interested in any wild theories about the connection between cleft chins and Parkinson’s !) … I came across several references to the fact that people with cleft chins tend to be sensitive, emotional and attention- seeking . And in fact a surprising number of celebrities have cleft chins ( and also Parkinsons? Billy Connolly eg . Robin Williams, Bob Hoskins, Neil Diamond ( less obvious but still show on some photos) Could this type of personality be a reflection of higher neuronal sensitivity in early life?
It would also be interesting to see what proportion of people with Parkinson’s would describe themselves as having an HSP personality . ….
LikeLike
Hi Mel,
Thanks for your comment. Interesting idea – maybe contact Dr Alastair Noyce at the PredictPD project (https://www.predictpd.com/) and ask him. He would be able to deal with this question this better than me (he is part of the team that did the chin cleft work – for readers not familiar with what we are talking about here: https://scienceofparkinsons.com/2018/03/04/associations/). And when Alastair gives you the answer, please share it here 🙂
Kind regards,
Simon
LikeLike
Very interesting read. Made me wonder around the impact of stress, and its brain chemistry. Does it lead to excitotoxicty, and therefore could it be a trigger or cause. It’s widely talked about as making symptoms worse of course.
LikeLike
Hi Yopd,
Thanks for your comment and interesting question. I guess if the ‘excitotoxicity model’ of Parkinson’s is correct and the mitochondria are being stretched, then I suppose that stress could be detrimental and put further pressure on a fragile situation. It could explain the identical twins situations, where they both have genetic risk factors for Parkinson’s, but only one develops the condition. In particular, I am thinking of the case of the Gernsheimer twins (https://scienceofparkinsons.com/2016/07/27/identical-twins-and-parkinsons-disease/). Both have a GBA mutation, but in the late 1980’s one of them lost a son in tragic circumstances. The brothers speculate that the stress/suffering associated with that particular event may have been a catalyst for the Parkinson’s that followed. Food for thought about leading a less stressful style of life.
Kind regards,
Simon
LikeLike
Hi. I met a lovely lady last year who had been diagnosed with Parkinson’s a couple of years previously following chemotherapy. She was convinced that the stress of her battle with cancer had triggered the Parkinson’s and told me she had met several others with the same experience. Personally I know of several PWP whose first obvious symptoms followed a stressful experience and/ or a longer term stress ( myself included).
LikeLiked by 1 person
Hi Mel,
That is really interesting! I previously looked at stress in PD some time ago (https://scienceofparkinsons.com/2017/04/26/stress-and-parkinsons-disease/). Perhaps a revisit is required.
Kind regards,
Simon
LikeLike
Very interesting and certainly counter-intuitive. What about Lewy Bodies? Could they have an evolutionary advantage if they are there to gather up unprocessed bits of membrane, a-synuclein fibrils and dead mitochondria?
Could a-synuclein aggregation have evolved to reduce vesicle formation and subsequent dopamine release from over-excited dopaminergic neurons?
LikeLiked by 2 people
Hi Kevin,
Glad you found the post interesting and thanks for the great question – it’s actually a really intriguing idea. Lewy bodies as a rate limiting measure. I like it! But then I guess we’d have to explain the 50% of Alzheimer’s cases and 10-20% of normal healthy aged brains that also have Lewy bodies.
Elsewhere on the interweb, Prof Patrik Brundin (Van Andel Institute) has read your question and proposed another alternative:
“What if increased alpha-synuclein levels (that increase risk for PD) are also associated with improved defences against viral infections – the precise mechanism not known, but evidence is emerging that alpha-synuclein is upregulated during viral infections.
https://www.ncbi.nlm.nih.gov/pubmed/27631132”
(I’m sure he won’t mind me sharing)
I also really like Prof Brundin’s idea. I have long suspected that the aggregation we see is due to viral or microbial issues (https://scienceofparkinsons.com/2017/06/06/flu-jabs-and-parkinsons-disease/ & https://scienceofparkinsons.com/2016/05/29/a-change-of-dogma-for-alzheimers-disease/). There a lot of research building up now supporting this idea (just yesterday another research article appeared on pubmed supporting this idea: https://www.ncbi.nlm.nih.gov/pubmed/29572816). Most proteins have numerous functions, and it could be that beta amyloid and alpha synuclein have anti-microbial/viral properties. And any infection would put a great deal of stress upon already stretched cells. Imagine a high performance cell (due to a genetic variant that infers better performance) demanding extra energy from over-worked mitochondria. What would happen if that cell is suddenly infected by a virus – what does the already hyperactive cell do? Push everything off to one side (forming a Lewy body), while it deals with the viral intruder (who will also demand energy from the mitochondria, further increasing the production of oxidative species). Then imagine that scenario on a seasonal basis in a 50-60 year old person.
This is all wild speculation here, and I’m sure that the complex systems of the cell have equally complex redundancies, but I am intrigued with the idea outlined in this post. And I should add that this whole ‘evolutionary concept’ is largely Prof Chris Elliot’s (University of York) idea – he has conducted all of the fly research mentioned above.
Kind regards,
Simon
LikeLike
Hmm, it is well known in the patient/caregiver community that infections such as UTIs seem to lead to dramatic temporary worsening of PD symptoms, especially cognitive.
LikeLike
Hi Simon, thanks for the reply. Starting with the Lewy Bodies, it could be argued that their presence in Alzheimer’s and normal aged brains is support for the contention that Lewy Bodies are a consequence not a cause. If they are there to hoover up cell debris in cells that are compromised in some way, for example through mitochondrial stress, why shouldn’t that happen in circumstances other than Parkinson’s? Perhaps the stimulus for LB formation isn’t always related to aSN? It’s then more easy to appreciate the potential evolutionary advantages.
Could Lewy Bodies be an associated phenomenon that varies from Parky to Parky, in much the same way as the classical prodromal symptoms of anosmia, RBD and constipation? Presuming that Lewy Bodies are only discovered after death, the “symptom” is more likely to be present, as most of the people examined will be in advanced stages. Perhaps we get a disproportionate view of the prevalence of LBs compared to phenotypic symptoms that are more clearly apparent during life.
The potential anti-microbial role for aSN is fascinating. Thinking about its normal role – clustering synaptic vesicles at nerve terminals – plus how viruses could transfer and infect other cells, could an interruption in the normal role of aSN be a mechanism to prevent viral transmission from cell to cell, giving time for microglia to kill the infected cells? Again, an evolutionary advantage.
It would be interesting to find out the influence of viral particles on aSN aggregation using RT-QuIC.
I love this wild speculation!!
Kevin
LikeLike
I am struck that the occupations NOT associated with PD in the MMWR report are often ones that involve work at night or in dark environments. This might include “production” in that factory work often has night shifts. The occupations that ARE associated with PD are not only higher SES but involve a lot of interaction with people and other brain taxing activities. On the other hand, there is an inference that the lower SES jobs may have risk factors that lead to death sooner than PD. Fascinating!
LikeLike
Hi Anne,
Thanks for your comment – glad you found the post interesting. There may be something to the idea of ‘night shifts’ – for instance, just today a manuscript on this topic was uploaded to the preprint manuscript website BioRxiv (https://www.biorxiv.org/content/early/2018/03/26/288241). The finding of the study was that a tendency towards being a “Morning person” – a state driven by the circadian clock – increases the risk of developing Parkinson’s. So there does appear to be some kind of light/dark thing at play in Parkinson’s.
And your comment regarding occupations with more social interaction could also be associated with easier spread of viruses (see my response to one of the comments above).
It is a really interesting topic for discussion.
Kind regards,
Simon
LikeLike
Hi Simon. I sincerely hope some of the people I have talked to over the last few years read your article. Several times in the past when I have mentioned that the vast majority of Parky people I have encountered seem to be above average intellectually, or when compared professionally seem to come from the employment categories involving a more cerebral communicative set of skills, I have been contradicted. Indeed, one person took me to task for supposedly implying that Parky people were superior to the unafflicted masses! It is (obviously) hard to strut around feeling smug and superior when you cannot strut or even hold your head up. The problem is that some people out there see or hear everything as containing a hidden agenda, a political mindset informing their judgement.
Scientists are looking for the truth (or should be doing) and sometimes their own pet hypotheses become a casualty of the truth and they have to start again. When it comes down to it there is no possible chance of of a breakthrough being achieved if the base line we are using is not correct.
Anyway, I can point to these studies when any nay-sayer says PD people are no different to the rest: Oh, how I wish that were true!
LikeLike
Hi Lionel,
I hope all is well – glad you liked the post. I would refer you back to Darwin on that ‘Scientists are looking for the truth’ line. Truth is a matter of context as well. I have also often heard it said that people with PD are of higher intelligence, but I’ve always wondered how to test this idea. IQ testing after diagnosis? Retrospective collection of IQ scores? And what exactly is IQ testing?
I’ve also often heard it said that people who write about PD are of higher intelligence. Given the number of typos and spelling errors on this rubbish site, I guess there’s no evidence supporting that idea here 🙂
Kind regards,
Simon
LikeLike
I wonder if there could also be a connection with athletic abilities in younger life. Just anecdotally pwp that I’ve met seem proud of earlier skills.
LikeLike