|
As the age of personalised medicine approaches, innovative researchers are rethinking the way we conduct clinical studies. “Rethinking” in radical ways – think: individualised clinical trials! One obvious question is: Can you really conduct a clinical trial involving just one participant? In this post, we will look at some of the ideas and evaluate the strengths and weaknesses these approaches. |
A Nobel prize medal. Source: Motley
In the annals of Nobel prize history, there are a couple winners that stands out for their shear….um, well,…audacity.
One example in particular, was the award given to physician Dr Werner Forssmann. In 1956, Andre Cournand, Dickinson Richards and Forssmann were awarded the Nobel Prize in Physiology or Medicine “for their discoveries concerning heart catheterisation and pathological changes in the circulatory system”. Forssmann was responsible for the first part (heart catheterisation).
Source: Nobelprize
In 1929, at the age of 25, Forssmann performed the first human cardiac catheterisation – that is a procedure that involves inserting a thin, flexible tube directly into the heart via an artery (usually in the arm, leg or neck). It is a very common procedure performed on a daily basis in any hospital today. But in 1929, it was revolutionary. And the audacious aspect of this feat was that Forssmann performed the procedure on himself!
And if you think that is too crazy to be true, please read on.
But be warned: this particular story gets really bonkers.
The historical account of this event begins with Forssmann somehow persuading an operating-room nurse, Gerda Ditzen, to assist him in his crazy experiment. Naturally, she agreed to help of course, but only if he promised (and I kid you not) that he would do the procedure on her rather than on himself (and remember that this procedure had never been conducted before – she was volunteering to be his guinea pig).
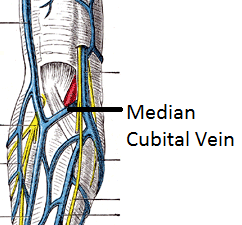
Forssmann naturally agreed to this condition, and off the two of them cheerfully went to the operating room. Having securely restrained Ditzen to the operating table, Forssmann then pretended to locally anaesthetise her arm whilst actually anesthetising his own arm. He next inserted a catheter (a flexible tube) into his own median cubital vein.
The median cubital vein lies just below the elbow. Source: Study.com
Following this, Forssmann somehow managed to one-handedly release Ditzen – who had apparently realised what was happening at this point – and he told her to call the X-ray department. The two of them then excitedly walked down to the X-ray department (it was one floor below where they had started this madcap adventure). Once there, with the help of a fluoroscope (an Xray-like instrument), Forssmann continued to advance the catheter a full 60 cm into his arm. It crossed his chest and entered the right ventricle of his heart. To record this amazing achievement they made an X-Ray before the catheter was removed.
Ladies and gentlemen, the X-ray:
Forssmann’s X-ray image. Source: Jbsr
Bonkers right?
But truth be known, the history of science is full of these sorts of stories.
I have previously discussed the case of researcher Barry Marshall – an Australian physician – who swallowed a petri dish containing cultured microbes just to prove that they cause stomach ulcers. And he was right. He got both stomach ulcers and the Nobel prize in Medicine in 2005 for his efforts (Click here to read a previous post about this).
Barry Marshall (left) and Robin Warren. Source: AustraliaUnlimited
Perhaps we only hear about the success stories (the individuals that actually survived their big gamble), but it has to be said that these individuals are the ones that change the world.
Remember the words of Apple founder Steve Jobs:
“Here’s to the crazy ones, the misfits, the rebels, the troublemakers, the round pegs in the square holes… the ones who see things differently — they’re not fond of rules… You can quote them, disagree with them, glorify or vilify them, but the only thing you can’t do is ignore them because they change things… they push the human race forward, and while some may see them as the crazy ones, we see genius, because the ones who are crazy enough to think that they can change the world, are the ones who do”
— Steve Jobs, 1997
Inspirational stuff. But what has any of this got to do with Parkinson’s disease?
Well, we have a few “misfits and troublemakers” in the world of Parkinson’s disease research who are trying to change the way we do things for the better.
Sara (soon to be Dr) Riggare is one of them:
Sara is an engineer by training, but her focus is very much on Parkinson’s – a condition that she lives with.
She experienced her first symptoms as a teenager in 1984, but her condition was not correctly diagnosed until nearly 20 years later. Taking the bull by the horns, in 2010 she decided to combine her personal experiences of the condition with her engineering skills to try to improve things for herself and others within the community. This effort began with a masters degree in health informatics at the Karolinska Institute in Stockholm (Sweden), and it has subsequently led to a PhD on the topic of “Personal observations as a tool for improvement in chronic disease”.
Sara is an actively engaged with the Parkinson’s disease community and her website – www.riggare.se – is must reading. For a long time, she has been very keen on the idea of self-tracking:
Frustrated by the lack of medical monitoring – just 1 hour with her neurologist per year out of the 8,765 hours there are in a year – Sara has taken matters into her own hands.

A schematic illustrating the limited monitoring of Parkinson’s. Source: Riggare
So why is she a misfit?
While Sara is not as much of a misfit as Dr Forssmann, she is keen to change a few things in the world of Parkinson’s research. Recently Sara has turned her attention to the topic of clinical trials. And she has collaborated with her friend, Jasmine Sturr (a fellow Parkinson’s sufferer) in this effort.
Having developed Parkinson’s when she was just 14 years old, Jasmine makes a perfect ‘partner in crime’ for Sara – Jasmine really is a misfit: she makes up her own L-dopa!
Watch this video to learn more about her:
Their main concern with the current model of clinical trials is that they are too long and too expensive to run. Affected individuals have to wait over a decade for a new drug to pass through the clinical trial process, and often possibly effective drugs can fail the clinical process because the population that the drug is being tested on are too heterogeneous (meaning different or mixed).
This is a worry for a lot of folks in the Parkinson’s community. But Sara and Jasmine are not the types to sit around complaining about it. They have collaborated with another Parkinson’s sufferer, Dr Kent Unruh (who is a researcher at the University of Washington) to explore new ideas in clinical testing.
Here is a video of Dr Unruh explaining some of his own work:
This week, the three of them published this research report:
Title: Patient-driven N-of-1 in Parkinson’s Disease – Lessons learned from a Placebo-controlled study of the effect of Nicotine on Dyskinesia
Authors: Riggare S, Unruh KT, Sturr J, Domingos J, Stamford JA, Svenningsson P, Hägglund M.
Journal: Methods Inf Med 2017; 56(99):e123-e128.
PMID: 29064509 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers aimed to investigate the use of patient-driven individualised studies in the area of Parkinson’s self management. They used the example of controlling levodopa-induced dyskinesias (or LIDs) with nicotine to test this idea. And the entire experiment was conducted between 10:00 and 11:00 am on the 23rd September (2016) in the Oregon Convention Centre during the 4th World Parkinson Congress, and the study involved just one participant (thus the “N-of-1” title, N indicating the ‘number’ of participants in the study).
Source: WPC.
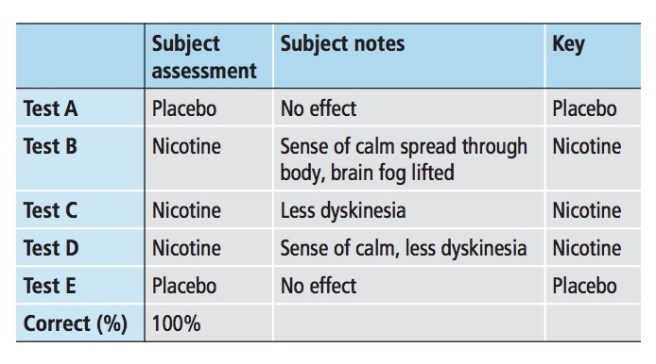
Two identical e-cigarettes (KangerTech mini starter kit) were purchased along with two bottles of e-juice of identical flavour. The difference was that one e-cigarette contained nicotine (3 mg/ ml) and the other did not. Sara was the sole participant in the study and she took additional L-dopa one hour before they started the experiment to increase the appearance of dyskinesia.
Sara was blind to which e-cigarette contained the nicotine and after taking 3 to 4 puffs from the e-cigarette that was handed to her, she would write down notes of her observations of the effect that she experienced on her dyskinesias. She did the test 5 times (blindly being handed an e-cigarette each time) and these were the results:
Source: Methods
Nicotine has been shown to reduce L-dopa induced dyskinesias in both mice and primates (Click here and here to read more about this). A clinical study has also been conducted, but the results have never been published (Click here to read more about this). Sara found that she could easily determine when she was inhaling nicotine by the response that she experienced with her dyskinesias.
But surely she could tell from the physiological effect of nicotine on her body when she was puffing the nicotine e-cigarette so is this really a proper experiment?
It is a fair comment, especially considering that Sara is not a smoker, and the nicotine probably had a very notable physiological effect on her. The researchers acknowledged in their discussion that the n=1 design has limitations that prevent any definite conclusion from being drawn. But the researchers were conducting the experiment not just to investigate the effects of nicotine on dyskinesias. They were also assessing the issue associated with conducting N=1 studies. And they found that the main challenges of patient-driven N=1 studies were:
- the planning of the study
- recruiting a suitable research team
- making sure the data collection is optimal
- analysis and publication of the results
And the researchers concluded that the main contribution of the study was “highlighting the work done by patients on a daily basis for understanding their conditions and conducting self-tracking experiments. More work is needed to further develop methods around patient-driven N-of-1 studies for PD”.
Mmmm. Not sure I’m buying it. What other implications could there be for this sort of research?
A fantastic potential output of this kind of study would be people with Parkinson’s all over the world beginning to report the results of their own N-of-1 studies on a wide variety of variables that could affect their condition (a good example of this is the mannitol group set up by Clinicrowd). Thus, rather than there simply being the occasional anecdotal case of a person responding to something in particular, there could potentially be an enormous database of information built up by individuals conducting N-of-1 studies on themselves. This kind of endeavour could also help highlight particular subtypes of the condition, as responders and non-responders share their experiences and backgrounds.
And before readers pass a negative judgement on this idea, understand that the implications are important.
Cannabis being grown for medicinal use. Source: BusinessWire
Previously we have discussed the use of medical cannabis to help manage Parkinson’s symptoms (Click here to read that post), and the conclusion of that post was that the findings of large clinical studies had not really found supporting evidence for the idea. I am now regularly contacted by individuals who claim that cannabis certainly works for them. These individuals may be a subset of people with Parkinson’s that can be considered responders to cannabis. But by highlighting the negative results of a large clinical study in the published research reports, are we in fact ignoring the individuals for whom a particular variable may work?
Are we limiting ourselves to medicines that work for the majority?
If so, why? It’s kind of stupid when you think about it.
Thus, there is a place for N=1 research.
But there would need to be careful guidance provided for conducting such studies in isolation. And this is apparent from previous attempts at N-of-1 studies.
What previous N-of-1 research has been conducted on Parkinson’s disease?
Quite a lot actually.
Here is one example:

Title: A proof-of-concept, randomized, placebo-controlled, multiple cross-overs (n-of-1) study of naftazone in Parkinson’s disease.
Authors: Rascol O, Ferreira J, Nègre-Pages L, Perez-Lloret S, Lacomblez L, Galitzky M, Lemarié JC, Corvol JC, Brotchie JM, Bossi L.
Journal: Fundam Clin Pharmacol. 2012 Aug;26(4):557-64.
PMID: 21585523
Naftazone (also known as 1-2-naphtoquinone-2-semicarbazone) is a treatment for varicose veins and venous insufficiency (a condition where the flow of blood through the veins is inadequate, causing blood to pool in the legs). Naftazone has also been shown to reduce the glutamate (a chemical involved in neural activity). A previous study of Naftazone in Parkinsonian primates suggested that the drug had anti-parkinsonian and anti-dyskinetic properties (Click here to read more about this – download the PDF and search for 310 on page S96; “FP0011 extends the duration of the anti-parkinsonian actions of L-DOPA…”). In this study, the researchers wanted to determine if Naftazone could also have anti-parkinsonian and/or anti-dyskinetic properties in humans.
In a proof-of-concept, randomised, double-blind, placebo-controlled, multiple-cross-over n-of-1 study in patients with Parkinson’s with dyskinesias, the researchers wanted to “assess if the response of the first few PD patients exposed to naftazone would be compatible with animal observations regarding efficacy, dosage and safety profile. Such information was thought to be useful before embarking into a longer, larger and resources-consuming full Phase II development program”. (Smart thinking!)
In order to do this, each participant in the study received naftazone and placebo in 4 consecutive “cross-over sessions” which were run in a random order. A cross-over session involves blindly changing from the treatment to a placebo or vice versa. Each cross-over session was composed of two phases of 2 weeks each (2 weeks of naftazone or 2 weeks of placebo before switching to the other treatment).
Seven individuals were recruited for the study. They had moderately/severe L-dopa induced dyskinesias (on average more than 25% of the waking day). To assess participants responses the investigators used three different approaches (self -reported diaries, a questionnaire (the Likert scale), and clinician based assessment (UPDRS)). The results showed encouraging improving quality of ON-time in a small number of the participants – a result which may not have been observed in a larger study.
The researchers concluded that “these pilot findings are consistent with preclinical data in primates and support the hypothesis that naftazone may have anti-parkinsonian and anti-dyskinetic effects in humans that deserve further clinical investigation“. (In 2005, Faust Pharmaceuticals initiated a Phase-II clinical trial of FP 0011 (naftazone) in Parkinson’s disease in France – I can not find any information relating to the results of that trial).
Here is another example of a N=1 study in Parkinson’s:
Title: Simvastatin decreases levodopa-induced dyskinesia in monkeys, but not in a randomized, placebo-controlled, multiple cross-over (“n-of-1”) exploratory trial of simvastatin against levodopa-induced dyskinesia in Parkinson’s disease patients.
Authors: Tison F, Nègre-Pagès L, Meissner WG, Dupouy S, Li Q, Thiolat ML, Thiollier T, Galitzky M, Ory-Magne F, Milhet A, Marquine L, Spampinato U, Rascol O, Bezard E.
Journal: Parkinsonism Relat Disord. 2013 Apr;19(4):416-21.
PMID: 23283428
The cholesterol lowering drug Simvastatin has previously been shown to reduce dyskinesias in rodent models of Parkinson’s (Click here and here to read more on this). In this study, the researchers wanted to follow up this finding in both primates and humans. They firstly looked at the effect of Simvastatin on dyskinesias in 6 Parkinsonian monkeys and found that (at very high doses – over 3 mg/kg), Simvastatin did reduce dyskinesia.
Given the high dose the investigators decided to conduct a “n-of-1”, randomised, placebo-controlled, three cross-over trial in 10 people who had Parkinson’s disease with troublesome dyskinesias. This exploratory study, however, revealed no effect at doses considered safe for human (40 mg/day) suggesting the necessity of higher doses, which would not be compatible with a safe prolonged administration.
This small study was probably a better way of testing the drug rather than conducting a large, expensive clinical trial.
A third example of a N-of-1 study in Parkinson’s:

Title: Espresso Coffee for the Treatment of Somnolence in Parkinson’s Disease: Results of n-of-1 Trials.
Authors: Ferreira JJ, Mestre T, Guedes LC, Coelho M, Rosa MM, Santos AT, Barra M, Sampaio C, Rascol O.
Journal: Front Neurol. 2016 Mar 8;7:27.
PMID: 27014181 (This article is OPEN ACCESS if you would like to read it)
Daytime sleepiness or drowsiness (also called somnolence) is a frequent problem in Parkinson’s. In this study, the researchers wanted to evaluate the efficacy of espresso-coffee in the treatment of daytime sleepiness in Parkinson’s. They conducted multiple n-of-1 clinical trials comparing regular espresso coffee to decaffeinated coffee in participants with Parkinson’s that had moderate to severe daytime somnolence (as defined by an Epworth Sleepiness Scale score >9). The study involved three crossovers (two treatment periods separated by two days of no coffee drinking).
Four participants were included in the studies and two of them experienced beneficial effects on their levels of sleepiness during exposure to espresso coffee. The investigators concluded that “multiple single patient trials are feasible in Parkinson’s” and their results suggested that “espresso-coffee may have a beneficial effect on daytime somnolence in some patients“. But they were cautious to point out that “these results cannot be generalised beyond the patients included in these trials“.
A subsequent large clinical trial of caffeine on excessive daytime somnolence in Parkinson’s has recently been completed, but the results are still not published (Click here to read more about this trial).
None of these studies have really provided strong results. So why conduct them?
The point is that they are exploratory.
The goal of conducting them is not to try to determine efficacy of a particular treatment, they are being used to explore the parameters for a potential future trial. Before conducting large, expensive clinical studies, why not conduct small n-of-1 studies to see if specific individuals should be targeted for a particular clinical trial.
But there are aspects of these small studies that are an important part of where the future of the clinical trials process is heading.
So where is the future of clinical trials heading?
Have you ever heard of the Critical Path Initiative?
Source: CPI
Launched in March 2004, the initiative was introduced by the Food and Drug Administration (FDA) in the USA to modify the way we move drugs from the lab to the clinic. Specifically, the goal was to deal with the high rates of attrition that we have seen in the clinical phases of testing.
Acknowledging the increasing difficulty and unpredictability of novel treatments development, the initiative proposed that collective action was required to modernise the tools we are using in the clinical trial process. And this includes harnessing information technology to evaluate/predict the safety and effectiveness of medical products.
Historically, obtaining clinical trial results that were “reliable and valid” required fixed study designs. Randomly allocating a group of affected individuals to two or more groups and blindly treating them with treatments or a placebo. The studies required the conditions to remain exactly the same until the end of the trial period when the results could be determined.
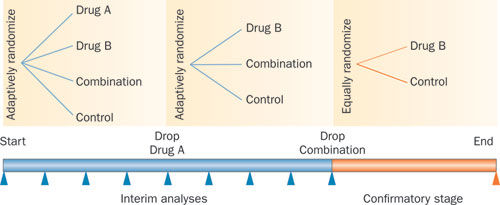
This idea is now being re-considered and has given rise to the idea of “Adaptive clinical trials“.
The aim of an adaptive clinical trial is to offer more “flexibility to investigators to find the optimal clinical benefit without affecting the study’s validity” (Wikipedia). An adaptive trial is dynamic by design; the structure anticipates one or more changes in the trial’s direction or scope as the study is conducted. And those changes will be based on information generated from within the study (not from external sources) and must not alter the scientific integrity of the research. In addition, there should be a focused effort to determine which patient populations can benefit most from a particular drug.
Think of it as a ‘learn as we go’ approach, as opposed to the old fashioned ‘let’s wait and see what the results say at the end of the trial’ model.
An example of an adaptive clinical trial Source: Nature
One of the main differences between traditional randomised controlled trials and adaptive clinical trial is frequent interim analyses – better and earlier use of the data being collected in the study. This actually means letting the data guide the course of the study. Thus rather than waiting years for the trial to end and results to be determined, a more proactive approach is taken. And if used correctly, adaptive clinical trials could save both costs and time (and potentially lives) by eliminating the number of study failures.
A good example of one of the proposed changes is non-adverse patient reactions. Traditionally, only adverse reactions have been recorded during a clinical trial, and non-adverse patient reactions have not been considered until a trial is completed. Why not collect and use this information during the trial?
Sounds interesting. Have there been any examples of adaptive clinical trials?
Yes, there have. In fact, it has been calculated that approximately 20% of Phase III trials utilise some elements of adaptive design (Source).
The main example of adaptive clinical trials that folks discuss is the I-SPY clinical trials – which are focused on cancer.
Source: ispytrials
I-SPY is an adaptive trial design that has enabled two experimental breast cancer drugs to deliver promising results after just six months of clinical testing. Critically, researchers were assessing the results during the trial and they found that the cancer had been eradicated in more than half of one group of patients.
This video explains a lot about the I-SPY trial program:
The basic structure of the I-SPY program can be broken into 3 components:
- I-SPY 1 trial is similar to a standard phase I clinical trial in that it is designed to determine drug safety. The I-SPY program, however, is also designed to decrease the amount of time required to collect drug safety data. The I-SPY program networks the clinical sites, which allows for accelerated expansion of enrolment, through the sharing of earlier feedback between clinical sites. And this information has an important role in the preparation of the I-SPY 2 and I-SPY 3 components.
- The I-SPY 2 trial is focused on treating patients with stage 2-3 breast cancer at the time of primary diagnosis and similar to a standard clinical Phase II trial design the objective is to determine safety and efficacy of a new treatment. However, by incorporating many innovative features, the I-SPY 2 trial decreases the time, the cost, and the number of patients required to efficiently test the new drugs. An important feature of I-SPY 2 trial is that it can simultaneously evaluate candidates developed by multiple companies – escalating or eliminating drugs based on results as the study continues. This approach significant reducing costs with regards to setting up and running individual Phase 3 trials. As of January 2016, I-SPY 2 was comparing 11 novel treatments against the ‘standard therapy’.
- Similar to all Phase III clinical trials, I-SPY 3 is ultimately designed to confirm the efficacy of new treatments. However, the goal of the I-SPY 3 trial will be to accelerate the phase III testing of agents through a variety of mechanisms, such as targeted eligibility criteria and again continuous interim data analysis.
Source: ispytrials
Has this approach ever been used for Parkinson’s disease?
No, but there are efforts underway to use this approach for Alzheimer’s disease (Click here to read more).
Parkinson’s disease would probably be a better fit for this model of clinical testing, however, as there are a wider variety of drug classes being proposed for Parkinson’s disease (both from novel therapeutics and the re-purposing of clinically available drugs).
And it would certainly be very interesting to see more effort focused on setting up this style of clinical testing of drugs for Parkinson’s disease rather than merely continuing down the slow and expensive process we have at the moment.
What does it all mean?
This post was all about re-thinking old ideas in the hope of speeding up and improving success rates.
I was once asked in a job interview which individual I think of when I think of the word ‘success’ (I hate these sorts of idiotic questions).
I answered Helen Keller.
Maybe some people would say Steve Jobs or perhaps Elon Musk, but I suspect they are measuring success by the outcome. An alternative metric is starting point. Steve Jobs grew up in Silicon valley, his starting point was rather convenient. Helen couldn’t see or hear anything from 19 months of age – her starting point was somewhat less than convenient, but her life story and more importantly her legacy are amazing.
The point is: she was always trying new things, learning and adapting as she went along, and it is wondrous what she achieved with just sheer concentrated willpower.
This post was intended to highlight patient-driven research efforts, but it gradually morphed into something slightly different. It is hopefully food for thought in many different areas, but the goal of it is to add to the discussion on how we can speed up and increase the rate of success in the clinical trial setting. In addition to developing new therapies for Parkinson’s, we must begin planning for the future of clinical testing of those treatments, and I think that this is one discussion in which the member’s of the Parkinson’s-affected community (by lay and researcher) can have a powerful influence.
I would be interested to know what you think about these ideas.
The banner for today’s post was sourced from Healthblawg













Interesting. I suppose these N1 studies need to be rigorous and follow most of the scientific guidelines. Just one person saying espresso helps them isn’t enough without the care in setting it up. Seems like a non-scientist like me wouldn’t have too much more than a bit of anecdotal experience to offer? There is no kit you can buy with directions on how to add to the research on a given substance? (I didn’t read this really carefully and am in need of sleep)
LikeLike
Hi DKDC,
I think there would need to be guidelines regarding how to run N=1 studies in isolation. A bit of co-ordinated organisation and some Youtube videos explaining how to do things. I’m not sure we are there yet, but it could be something that the community could look at investing some time in. And it could be relatively effortless, like providing Fit-bit results after a change in medication sort of thing. Unfortunately I’m too busy to take on such an idea, but it would be useful if someone interested did.
Am in need of sleep as well.
Kind regards,
Simon
LikeLike
This recent commentary in Nature may be of interest to readers: http://www.nature.com/nature/journal/v550/n7676_supp_out/full/550S122a.html
LikeLike
Fabulous article! Now to setup my own n=1 study!!
Simon, I read your article on Niacin and the Pink1 mutation, but have you considered looking into Nicotinamide Riboside? I realize there is nothing related to Parkinson’s yet, but I think the benefits of NAD+ precursors shown in animal models to date are “tantalizing” for sure and would most definitely apply to treatment of Parkinson’s as well. Plus I think the preliminary evidence hints that NR would be a superior NAD+ precursor as compared to niacin and nicotinamide.
http://www.sciencedirect.com/science/article/pii/S155041311630482X
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3632303/
http://www.sciencedirect.com/science/article/pii/S0969996110002767
http://www.annualreviews.org/doi/10.1146/annurev.nutr.28.061807.155443
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5062546/
As I mentioned previously my own regimen includes trehalose, nicotinamide riboside, and pterostilbene. My theory is trehalose for inducing autophagy, and the other two for cellular repair plus some additional boost to autophagy. I realize the amount of trehalose reaching my bloodstream is bound to be tiny, but then that’s why I took it upon myself to try sniffing a tiny amount (about .3 g) (don’t worry I’m being watched carefully by an ENT doctor) 😀 My hope is that since the damage caused by the Ataxin-1 protein in my brain takes a lifetime to appear that perhaps decades of a tiny amount of trehalose will be meaningful. Of course the study that showed some fall off in result with longer treatment is troublesome, but when dealing with a disease with no available treatment some hope is better than none, at least in my book.
Thanks again for such an incredibly insightful website!
LikeLike
interesting – can you buy all these from the interwebs?
LikeLike
Hi dk – I wasn’t sure if you were referring to my comment or the article above, but if it was to my comment then the answer is yes. trehalose, niagen, and pterostilbene are all available online. They are sold as health supplements. Of course, I’m certain I don’t need to tell you this, but I’ll put it out there just to cover my bases: one should never try a supplement based on an online comment without first doing his own research and talking to his doctor 😀 Having said that I admit I am a HUGE believer in this combination of supplements being combined with a healthy diet and exercise as having enormous potential to help. I am an active poster on many websites associated with ataxia, HD, and PD and now have about 2 dozen people that have reported back to me after trying various combinations of the above, and some people are reporting results similar to what I have witnessed in myself and my father and my aunt (they too have SCA1). I firmly believe there exists today a “cocktail” treatment for many, many neurodegenerative ailments. People just need to understand their own underlying pathology and then seek out the “cocktail” with the most evidence based research for their own situation.
LikeLike
Hi Joe,
Glad you liked the post and thanks for the interesting comment. I have had a couple of readers ask about Nicotinamide Riboside and the reason for not looking at it yet is (as you say) there is so little Parkinson’s research on the topic. I am reluctant to stray too far from the peer-reviewed results for PD. And there are enough other topics to keep me busy until some nicotinamide riboside/PD research does show up (I suspect some will be coming soon).
I would like to see the trehalose ‘falling off’ result independently replicated before we start to worry about it. And I would be interested to hear about your experience with pterostilbene.
Kind regards,
Simon
LikeLike
Thanks, Simon. Very interesting, as usual.
I, too, have also been conducting my own n=1 out-of-clinic trial, but with the NR that Joe has been using (above) and NAC (yes, I’ve informed my neurologist and general practitioner, who rather pooh-poohed the idea). In addition, I’ve been walking 2 miles daily, and have shifted toward a mediterranean diet (based on the work of Dr. Mischle, also discovered from Simon’s sleuthing on SoPD).
The biggest problem with n=1 trials is that they’re only really effective for short-time-constant effects. If an effect takes time to build up, it can be masked by the underlying relentless deterioration that pd is known for. The only way to know if the deterioration has slowed (assuming it doesn’t amount to a reversal) is to compare deterioration rates among a group, in which n>>1.
The other big problem is that, based on what Ive read, I think we’re dealing with multiple diseases which present with varying subsets of symptoms we lump into the convenient category of Parkinson’s disease. Some have tremors, some have freezing, some gait disturbance. Most will eventually progress toward all of the above. Some will not. Most have damage to the Substantia Nigra giving rise to dopamine depletion, while others are suspected of lacking serotonin. Some have genetic modifications leading to oxidative stress on neurons, while most do not (as far as we know). Most of us have constipation, sleep disorders, somnolence, etc., though many do not.
This plethora of presentations, and wildly divergent apparent causes for the ‘same’ disease is bound to confound large clinical trials in which all of us are lumped together at random. Instead, we need much more focussed trials of treatment regimens aimed at known causes of specific PD pathways. What good does it do to test a large non-specific group of people with PD in attempting to resolve an issue with a particular mutation (like Pink1 and Parkin)? The result of such a test would show the effect as small, or non-significant, though if tested against only people with known relevant mutations may prove highly beneficial.
So, I applaud you, Simon, for bringing this to light, and opening up a very important discussion.
Thanks for all your work, keeping us informed (and optimistic).
LikeLike