|
# # # # Over the last 20 years, researchers have identified a number of genetic variations that can confer an increased risk of developing Parkinson’s. Tiny alterations in regions of DNA (called genes) – which provide the instructions for making a protein – can increase one’s chances of Parkinson’s. A better understanding of the biological pathways associated with these genetic risk factors is opening up vast new areas of research. Recently researchers have been exploring the biology behind one particular genetic risk factor – involving a gene called TMEM175 – and they have discovered something quite unexpected: While one genetic variation in the TMEM175 gene increases the risk of Parkinson’s, another variation reduces it. In today’s post, we will explore the biology of TMEM175, review what the results of the new research indicate, and consider why these findings might be interesting in terms of potential future therapeutic targets. # # # # |
 Wadlow (back left). Source: Telegraph
Wadlow (back left). Source: Telegraph
Robert Pershing Wadlow was always in the back of school photos.
Born February 22nd 1918, Wadlow’s birth certificate indicated that he was “normal height and weight“, but from that point onwards, there was nothing normal about his rate of growth.
By the time, Robert was 8 years old, he was taller than his father (he was 6 foot/183cm). And eight years later when he turned 16, Robert was 8 foot 1 (2.47 m)… and he was still growing.
Here is a picture of him with his family at 19 years of age:
 Source: Businessinsider
Source: Businessinsider
Robert was the tallest person in recorded history, and at the time of his death – at the tragically young age of 22 – Robert was almost 9 feet tall (8 ft 11; 2.72 m)… and still growing.
His incredible growth was caused by a condition called hyperplasia of his pituitary gland. This condition that results in an overactive pituitary gland which causes an abnormally high level of the human growth hormone to be produced.
 Source: Britannica
Source: Britannica
Human growth hormone (or somatotropin) is a peptide hormone that belongs to a much larger group of molecules that are referred to as growth factors.
In general terms, growth factors are small molecule that plays an important and fundamental role in biology. They stimulate cell proliferation, wound healing, and occasionally cellular differentiation.
And Robert’s story is an example of how powerful the effect these tiny molecules can have.
Growth factors are secreted from one cell and they float around in the extracellular world until they interact with another cell and initiate survival- and growth-related processes.
 Source: Wikimedia
Source: Wikimedia
We have often discussed growth factors on this website in the past, with posts of growth factors like GDNF (Click here to read a SoPD about this) and CDNF (Click here to read a SoPD post on this). These discussions have largely focused on how growth factors could have neuroprotective and regenerative potential for Parkinson’s, stimulating survival and growth of cells.
Recently, however, new research has been published that demonstrates how some of these growth factors could be influencing an entirely different aspect of cellular biology that is connected to Parkinson’s: lysosomal function.
What is lysosomal function?
Lysosomes are small bags that are full of digestive enzymes that help to break down material inside of cells. Sometimes that material is newly imported from outside of the cell, while other times it may be old proteins that need to be disposed of.
Lysosomes provide the digestive enzymes for the job of breaking down the material.

How lysosomes work. Source: Prezi
We recently discussed lysosomes in more depth (Click here to read that SoPD post) – they are a critical component of normal biological function inside of cells.
But wait a minute. How do growth factors floating around outside of cells affect lysosomes inside of cells?
It’s a good question. One that was addressed in this very recent research report:
 Title: A growth-factor-activated lysosomal K(+) channel regulates Parkinson’s pathology.
Title: A growth-factor-activated lysosomal K(+) channel regulates Parkinson’s pathology.
Authors: Wie J, Liu Z, Song H, Tropea TF, Yang L, Wang H, Liang Y, Cang C, Aranda K, Lohmann J, Yang J, Lu B, Chen-Plotkin AS, Luk KC, Ren D.
Journal: Nature. 2021 Jan 27. Online ahead of print.
PMID: 33505021
In this study, the researchers were interested in the molecules that help to regulate lysosomes in cells.
Through a series of experiments, they made the surprising discovery that growth factors like brain-derived neurotrophic factor (BDNF) or nerve growth factor (NGF) were able to activate a potassium channel on lysosomes.
They called this channel lysoKGF (Lysosome – Potassium (K) – growth factor (GF)).
Hang on a second. What is a potassium channel and why do lysosomes have them? I thought they were just “bags of enzymes”.
It’s another good question.
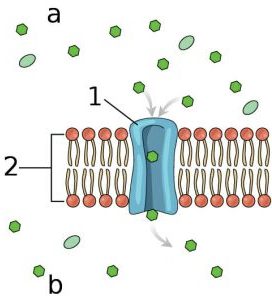
In order to do their job, most lysosomal enzymes need a highly acidic environment. The level of acidity can be regulated by certain channels in the membrane (the outer surface) of lysosomes.
Channels are protein structures in membranes that allow certain elements to pass through them into the interior. Many of these channels are very selective in what they allow to pass (for example, there are calcium channels and sodium channels which only allow calcium and sodium to pass, respectively).
 Source: Biologydictionary
Source: Biologydictionary
The chemical element potassium is the most abundant ion to interact with membranes in cells, and the channels in the lysosome membrane allow potassium to pass in and out. This transit helps to adjust the acidity of the environment within lysosomes, allowing for better enzymatic function.
Ok, so potassium channels help to regulate acidity inside lysosomes, but how is this associated with Parkinson’s?
Several years ago, the researchers behind the report we are reviewing today published this study:
 Title: TMEM175 Is an Organelle K(+) Channel Regulating Lysosomal Function.
Title: TMEM175 Is an Organelle K(+) Channel Regulating Lysosomal Function.
Authors: Cang C, Aranda K, Seo YJ, Gasnier B, Ren D.
Journal: Cell. 2015 Aug 27;162(5):1101-12.
PMID: 26317472 (This report is OPEN ACCESS if you would like to read it)
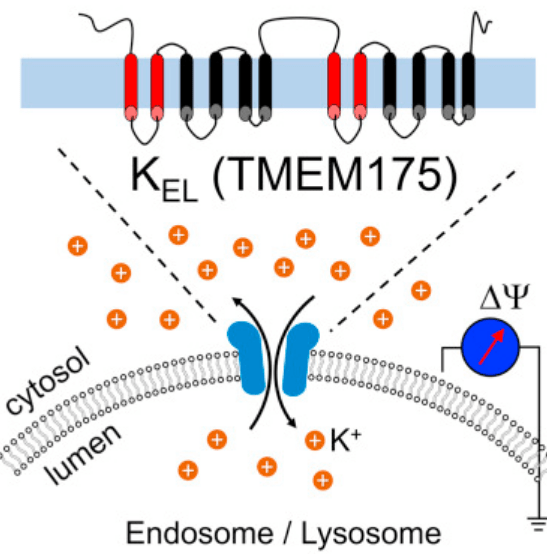
In this study, the investigators discovered a major potassium-selective channel, which is made up of a protein called TMEM175, which at the time was a protein of unknown function.
 Source: Cell
Source: Cell
By disrupting and manipulating TMEM175, the researchers found that this protein plays a major role in potassium flow in lysosomes, and contributes significantly to levels of acidity inside of lysosomes. They reported that lysosomes lacking TMEM175 exhibit no potassium flow and displayed marked instability in acidity.
This discovery was particularly interesting for the Parkinson’s research community because the year before this study was published, another report indicated that genetic variations in and around the TMEM175 gene (the section of DNA that provides the instructions for making TMEM175 protein) are a risk factor for developing Parkinson’s.
This is the report in question:
 Title: Large-scale meta-analysis of genome-wide association data identifies six new risk loci for Parkinson’s disease.
Title: Large-scale meta-analysis of genome-wide association data identifies six new risk loci for Parkinson’s disease.
Authors: Nalls MA, Pankratz N, Lill CM, Do CB,…(there are a lot of researchers were in the list of authors)…, Gasser T, Bertram L, Eriksson N, Foroud T, Singleton AB.
Journal: Nat Genet. 2014 Sep;46(9):989-93.
PMID: 25064009 (This report is OPEN ACCESS if you would like to read it)
In this report, it was discovered that there was a highly significant genetic risk factor for Parkinson’s on chromosome 4. The region in question covers several genes (including TMEM175 – see blue arrow in image below).
 The location of TMEM175 (blue arrow). Source: PMC
The location of TMEM175 (blue arrow). Source: PMC
And this finding has supported by a second large genetics study published a few years later (Click here to read that second study). The two genetics studies found that the region of DNA where TMEM175 sits contains the third most significant association with Parkinson’s.
Thus, this region is of serious interest for the research community.
|
# RECAP #1: Lysosomes are small bags that are full of digestive enzymes that help to break down material inside of cells. Recently researchers have discovered a number of genetic variations in genes involved in lysosomal function that are associated with an increased risk of Parkinson’s. One of these lysosomal genes in called TMEM175 . # |
Interesting. What do we know about the biological function of TMEM175 and how it relates to Parkinson’s?
In 2017, an independent group of researchers published one of the first report exploring the function of TMEM175 in the context of Parkinson’s:
 Title: TMEM175 deficiency impairs lysosomal and mitochondrial function and increases α-synuclein aggregation.
Title: TMEM175 deficiency impairs lysosomal and mitochondrial function and increases α-synuclein aggregation.
Authors: Jinn S, Drolet RE, Cramer PE, Wong AH, Toolan DM, Gretzula CA, Voleti B, Vassileva G, Disa J, Tadin-Strapps M, Stone DJ.
Journal: Proc Natl Acad Sci U S A. 2017 Feb 28;114(9):2389-2394.
PMID: 28193887 (This report is OPEN ACCESS if you would like to read it)
In this study, the investigators reported that eliminating (or reducing) TMEM175 protein in cells disrupted lysosomal function. This effect resulted in increased neuronal vulnerability to stressors, such as the aggregated form of Parkinson’s-associated alpha synuclein protein (Click here to read a previous SoPD post about this).
Their findings suggested not only that loss of TMEM175 screwed up the ability of cells to dispose of waste, but that this also made the cells more sensitive to stress.
These same researchers published a second report in 2019 that expanded on these results:
Title: Functionalization of the TMEM175 p.M393T variant as a risk factor for Parkinson disease.
Authors: Jinn S, Blauwendraat C, Toolan D, Gretzula CA, Drolet RE, Smith S, Nalls MA, Marcus J, Singleton AB, Stone DJ.
Journal: Hum Mol Genet. 2019 Oct 1;28(19):3244-3254.
PMID: 31261387 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers dug deeper into the genetics TMEM175 and identified one genetic variant (called p.M393T) in the TMEM175 gene, which was 20 orders of magnitude more significantly associated with Parkinson’s than any other variant in the area.
The investigators then explored this particular genetic variant in models of Parkinson’s and found that while increasing levels of normal TMEM175 protein was able to reduce levels of alpha synuclein aggregation (via normal lysosomal activity), increasing levels of TMEM175 protein carrying the p.M393T variant resulted in no change in alpha synuclein aggregation.
Cells that produced the TMEM175 protein carrying the p.M393T variant were found to have reduced regulation of lysosomal acidity and reduced lysosomal activity. And these findings were supported by another publication from a separate group of researchers:
 Title: Genetic, Structural, and Functional Evidence Link TMEM175 to Synucleinopathies.
Title: Genetic, Structural, and Functional Evidence Link TMEM175 to Synucleinopathies.
Authors: Krohn L, Öztürk TN, Vanderperre B, Ouled Amar Bencheikh B, Ruskey JA, Laurent SB, Spiegelman D, Postuma RB, Arnulf I, Hu MTM, Dauvilliers Y, Högl B, Stefani A, Monaca CC, Plazzi G, Antelmi E, Ferini-Strambi L, Heidbreder A, Rudakou U, Cochen De Cock V, Young P, Wolf P, Oliva P, Zhang XK, Greenbaum L, Liong C, Gagnon JF, Desautels A, Hassin-Baer S, Montplaisir JY, Dupré N, Rouleau GA, Fon EA, Trempe JF, Lamoureux G, Alcalay RN, Gan-Or Z.
Journal: Ann Neurol. 2020 Jan;87(1):139-153.
PMID: 31658403
In this study the researchers identified two genetic variations in the TMEM175 gene that were associated with Parkinson’s: the p.M393T variation mentioned above, but also p.Q65P. Interestingly, p.M393T was also found to be associated with REM sleep behaviour disorder – a prodromal condition for PD.
When the investigators explored the function of the M393T variation, they found that it was associated with reduced GCase activity (the lysosomal enzyme that is affected in GBA-associated Parkinson’s – click here to read a previous SoPD post about this).
All of this research suggested that TMEM175 may be an important player in the biology underlying some cases of Parkinson’s.
|
# # RECAP #2: TMEM175 is a protein that is involved with a potassium channel in the membranes of lysosomes. This protein helps to regulate the acidity of lysosomes. Variations the DNA encoding TMEM175 not only disrupt lysosomal function, but are also associated with an increased risk for developing Parkinson’s. # # |
Ok, so this Parkinson’s associated protein forms a channel on lysosomes to regulate the acidity inside. But (again) how do growth factors outside the cell, affect lysosomal function?
The researchers who conducted the study we are reviewing today found that their lysosomal potassium channel (that they called LysoKGF) consists primarily of two proteins: TMEM175 and AKT
What is AKT?
AKT (also called Protein kinase B) is a protein that plays an important role in many cellular processes (from cell proliferation and cell death, to transcription, and cell migration). It is an intermediate protein in biological cascades that regulate these processes.
 Akt protein structure. Source: Wikipedia
Akt protein structure. Source: Wikipedia
AKT is also a major target of growth factor receptors.
When the growth factors bind to their respective receptors, they activate AKT which leads to it playing a role in further downstream biological activities.
 Source: Researchgate
Source: Researchgate
And one of those activities is being part of the LysoKGF potassium channel on lysosomes.
The researchers found that manipulating levels of AKT affected potassium channel flow on lysosomes. They reported that AKT was necessary for normal TMEM175 function and this is how growth factors are influencing lysosomal activity.
So growth factors activate AKT, and that protein works with TMEM175 to form the LysoKGF potassium channel on lysosomes?
That is what this new research reports.
Interestingly, the researchers next looked at what happens to the LysoKGF potassium channel on lysosomes when TMEM175 protein has the p.M393T variation. They found that this results in a loss-of-function of the channel, which causes a reduction in lysosomal function.
Interesting. How many people have this p.M393T genetic variation?
So this is where things get rather intriguing.
You see, the study suggests approximately 17% of the general population possess the p.M393T variation. This makes it rather common!
It would be interesting to assess how many folks with “idiopathic” (or spontaneous) Parkinson’s also carry this variant.
That is interesting!
And what was even more interesting than that is what the researchers did next: they looked at another genetic variation (called p.Q65P).
p.Q65P is present in about 7% of the general population and it causes gain-of-function of LysoKGF potassium channels on lysosomes when cells are under stress. This genetic variation actually results in enhanced channel activity, and is associated with a reduced risk of developing Parkinson’s (by about 20%).
So one variant in the TMEM175 gene is associated with an increased risk of Parkinson’s, while another is associated with a reduced risk of the condition.
Wow!
“Wow!” indeed. But wait there’s more:
Having established the basic biology of the LysoKGF potassium channels, the researchers next wanted to explore their function in models of Parkinson’s.
They found that mice with reduced levels of TMEM175 protein exhibited an increased build up of the Parkinson’s-associated protein alpha synuclein. These mice also displayed deficits in behavioural tasks that require coordination. In addition, the researchers found that these mice showed evidence of dopamine neuron loss (these are the neurons that are badly affected in Parkinson’s).
The researchers also wanted to conduct the same set of experiments on the other variant so they engineered mice that produced the TMEM175 protein with the Q65P variant. In these mice, the investigators found that p.Q65P provides additional protection to neurons against stressors like oxidation and nutrient removal.
That’s in mice, but what about in humans? Has anyone looked at this in people with Parkinson’s?
To finish off their study, the researchers collected data from the Michael J Fox foundation supported Parkinson’s Progression Markers Initiative (or PPMI).
 The PPMI is studying volunteers with and without Parkinson’s for up to five years. It is a massive study attempting to better understand biological changes occurring in Parkinson’s over time.
The PPMI is studying volunteers with and without Parkinson’s for up to five years. It is a massive study attempting to better understand biological changes occurring in Parkinson’s over time.
The researchers collected data from 373 PPMI participants (who have available clinical and genetic data), and they found that people with Parkinson’s who carry the p.M393T genetic variants declined more rapidly than those without the variant.
The researchers concluded their study by writing “our studies uncover a pathway by which extracellular growth factors regulate intracellular organelle function, and establish a targetable mechanism by which common variants of TMEM175 confer risk for Parkinson’s“.
|
# # # RECAP #3: TMEM175 forms part of LysoKGF – a potassium channel on lysosomes that helps to regulate lysosomal function. New research has found that a genetic variant in the TMEM175 gene that occurs in 17% of the population can reduce the function of LysoKGF and increases the risk of developing Parkinson’s. A second genetic variation in the TMEM175 gene actually has the opposite effect – enhancing the channel activity and reducing the risk of Parkinson’s. # # # |
Are any biotech companies exploring TMEM175?
Yes.
Despite the research on TMEM175 only being 3-4 years old, there is already a biotech company called Caraway Therapeutics going after this area therapeutically.
Originally called Rheostat Therapeutics, this company focused on improving lysosomal function. Caraway has multiple active programs working on small molecules targeting cellular waste disposal, but their most advanced drug is targeting TMEM175.
And in late 2020, Caraway Therapeutics announced that they had received a research grant from the Michael J Fox Foundation to further develop their TMEM175-activators (Source), so Parkinson’s is an area of interest for the company.
It is also interesting to note that most of the researchers listed as authors on the Jinn et al (2017) and Jinn et al (2019) studies mentioned above are employees of the pharmaceutical company Merck.
![]() A large company like Merck would certainly have the resources and expertise to conduct the large screening experiments required to identify potential TMEM175-activators for Parkinson’s.
A large company like Merck would certainly have the resources and expertise to conduct the large screening experiments required to identify potential TMEM175-activators for Parkinson’s.
And given that Parkinson’s may not be the only condition that TMEM175-activators could be useful for, one would think that Merck would certainly be interested.
What do you mean?
TMEM175 appears to be involved with other conditions as well as Parkinson’s.
For example, this week an interesting new report was published highlighting TMEM175 in the context of Lewy body dementia.
Here is the report here:
 Title: Genome sequencing analysis identifies new loci associated with Lewy body dementia and provides insights into its genetic architecture.
Title: Genome sequencing analysis identifies new loci associated with Lewy body dementia and provides insights into its genetic architecture.
Authors: Chia R, Sabir MS, Bandres-Ciga S, Saez-Atienzar S,… (there is a VERY long list of authors in this study – to save space, I’m cutting a few out)…., Chiò A, Ross OA, Gibbs JR, Dalgard CL, Traynor BJ, Scholz SW.
Journal: Nature Genetics 2021 Feb 15. Online ahead of print.
PMID: 33589841 (A preprint manuscript of this report was made OPEN ACCESS if you would like to read it)
In this study, the researchers were interested in determining the genetic risk factors of Lewy body dementia, with the goal of better understanding the underlying biology of the condition.
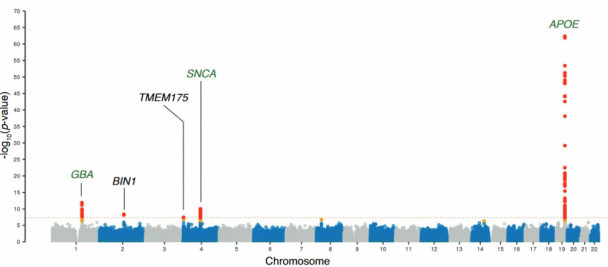
They collected DNA sequencing data from 2,591 individuals with Lewy body dementia and 4,027 unaffected control cases. There analysis found 5 genetic regions where variations were associated with increased risk of Lewy body dementia.
Those 5 regions were GBA, BIN1, SNCA-AS1, APOE, and….. drum roll please, TMEM175.
 Source: BioRxiv
Source: BioRxiv
The investigators highlight TMEM175 in the discussion of their report, noting that the “identification of TMEM175 underscores the role of lysosomal dysfunction in the pathogenesis of Lewy body diseases“.
Many of the reports highlighted in today’s post have pointed towards TMEM175 as a “targetable mechanism”. Hopefully we’ll see more research on this protein in the near future.
So what does it all mean?
I have great sympathy for Robert Wadlow.
I got a bit carried away with puberty during my younger years – reaching 6 foot by the age of 13 (my family doctor actually sent me off for a brain scan to check my pituitary gland and I fell asleep inside the imaging machine). But I remember always being (happily) relegated to the back row of class photos, and always standing out at school assemblies (one year I returned from summer holidays with two black eyes and a broken nose – long story – only for the head teacher to point me out in assembly with the words “So good to have you all back, fit & well, and… dear god, what happened to you?!?“).
Growing up we give little thought to the growth factors that influence much of our biological functioning, nor to the continuous lysosomal activity that kept us healthy. It is only as we age that we note the declining impact of growth factors and the reduced lysosomal function that is associated with numerous conditions.
A new report highlights interesting data that brings together growth factors and lysosomal function, and presents a novel mechanism of action for a Parkinson’s associated protein. It has also provided data pointing towards a genetic variation that may reduce the risk of developing Parkinson’s.
Here at the SoPD HQ, we are keen to see more research on the “targetable” TMEM175 protein and LysoKGF. We would also like to see independent replication of the genetic variant analysis across larger datasets to verify the “reduce risk” finding and explore deeper the p.M393T variation across the PD (and non-PD) populations. Clinical trials of TMEM175 activators are still some ways off in the medium term future, but it will be intriguing to see how this particular area of Parkinson’s research evolves.
There is certainly the potential for lots of growth.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
The banner for today’s post was sourced from pinterest



Hello Simon,
Once again good article.
Thank you.
Have you seen this recent published article https://www.nature.com/articles/s41591-021-01257-1?
Very interesting especially a better outcome when using autologous cells as opposed to allogenic.
This is still a primate model. Unfortunately it is behind a paywall for me. But the article about the research mentioned: ” …the researchers injected millions of dopamine-producing neurons and supporting cells into each monkey’s brain in an area called the striatum, which is depleted of dopamine as a consequence of the ravaging effects of Parkinson’s in neurons.”
The question is also what kind of supporting cells did they add?
LikeLike