|
Recently a study was published in which the researchers had used a large dataset from the the United States Medicare system. The dataset held medical prescriptions for beneficiaries aged 60–90 years. In their analysis, the researchers found that several diagnoses commonly treated with immunosuppressant medications were inversely associated with Parkinson’s – suggesting that perhaps the immunosuppressants may be reducing the risk of developing PD. When they looked closer at the immunosuppressants, the investigators found that of the six categories of immunosuppressants, two were clearly associated with a lower risk of PD. In today’s post, we will discuss what immunosuppression means, we will review the data, and we will consider some of the issues associated with immunosuppressants. |
George Hitchings and Gertrude Elion. Source: Achievement
After her grandfather died of stomach cancer and her fiance died of inflammation of the heart, Gertrude Elion dedicated herself to a future in medical research.
But despite a passionate love for laboratory research and having an excellent academic record, she was unable to get a graduate fellowship (or even an assistantship) due to the gender discrimination that existed at the time.
In the late 1930s, she enrolled in secretarial school with the goal of saving enough money to continue her education and achieve her goal.
After a year and a half of temporary secretarial and teaching positions, having saved up enough money, Elion enrolled as Master’s student in chemistry at New York University. She worked part-time as a receptionist and later as a substitute teacher to pay for her expenses. And she spent nights and weekends in the laboratory doing her research. She completed her degree in 1941.
George & Gertrude in action. Source: Wikimedia
In 1944, Gertrude was hired by George Hitchings who was working at what is now the pharmaceutical company GlaxsoSmithKline. It was the beginning of an amazing collaboration! And even though she never achieved a PhD, the work that she and Hitchings did together – research that won them the 1988 Nobel Prize in Physiology or Medicine – not only changed the way we design new drugs, but also gave the world its first drugs for immunosuppression.
What is immunosuppression?
Immunosuppression is the process of reducing of the activation or efficacy of the immune system.
Doctors regularly use medication that dampens down the immune system to (for example) allow a transplanted organ to function without being rejected by the body or (for example) reduce the effect of a hyperactive immune response in the case of autoimmune diseases.
How do these drugs do this?
They can reduce the immune response in several different ways. These can include:
- Drugs that block the division and expansion of blood cells (eg. inhibitors of nucleotide synthesis)
- Drugs that target specific cellular pathways involved in the immune response (eg. immunophilin-binding drugs)
- Antibodies that target specific cells in the blood, reducing their capacity to do their job (eg. T-cells, B-cells or both)
- Drugs that reduce immune response by targeting the messengers of the immune response (eg. anti-TNF antibodies)
For those interested in learning more about these approaches to immuno suppression, watch this video:
Interesting. But what does any of this have to do with Parkinson’s?
Last year an interesting research report was published:
Title: A predictive model to identify Parkinson disease from administrative claims data.
Authors: Searles Nielsen S, Warden MN, Camacho-Soto A, Willis AW, Wright BA, Racette BA.
Journal: Neurology. 2017 Oct 3;89(14):1448-1456.
PMID: 28864676
In this study, the researchers outlined a novel method for predicting factors that are associated with Parkinson’s. The study involved collecting information about the beneficiaries (aged 66-90 years) on the Medicare database who had Parkinson’s in 2009. They found 89,790 cases, and they matched those to 118,095 control cases.
Source: Dailyfly
Next, using only demographic data in the notes of each case and Medicare claims data collected from 2004-2009, the investigators started looking for any predictive factors. That is, any common features across the cases that was associated with the group that would go on to be diagnosed with Parkinson’s.
When the investigators began analysing all of the data, they found that Parkinson’s was associated with increased age, constipation, Rapid Eye Movement (REM) sleep behavior disorder (or RBD), and anosmia/hyposmia (loss of the ability to smell things); it was also more common in men than in women and more common in non-Hispanic white than non-Hispanic nonwhite cases. On top of this, Parkinson’s was inversely associated with tobacco smoking.
Smoking – not recommended. Source: Stanford
These connections were all expected and their appearance in the results basically validated the approach that these researchers were taking.
When the researchers started looking for predictive factors in the case notes during the years prior to a diagnosis of PD, they found many of the expected medical issues such as:
- Tremor and abnormalities of posture
- Psychiatric or cognitive dysfunction
- Gastrointestinal/urinary dysfunction
- Hypotension
- Sleep disturbance (including RBD)
- Fatigue
- Peripheral pain
- Restless legs syndrome
- Increasing incidence of falls
- The ordering of blood tests for syphilis (???), vitamin deficiencies, and thyroid conditions
- Weight loss
- Type II diabetes (without any complications)
Diabetic testing insulin levels. Source: Gigaom
All of these were positive predictors of Parkinson’s.
Then the research shifted their attention to the negative associations – case notes that were associated with a reduced incidence of Parkinson’s. These included:
- Hip replacement
- Osteoarthritis
- Carpal tunnel syndrome
- A variety of tendonopathies
- Cancer
- Cardiovascular disease
- Obesity-related conditions
- Gout and a variety of systemic inflammation (including inflammatory bowel disease)
- Allergies
Now this is where the researchers asked themselves a very simple question:
What do many of these conditions have in common?
And that was when they noticed that many of these conditions required immunosuppression medications.
This revelation led the researchers to conduct a follow-up study that was recently published:
Title: Immunosuppressants and risk of Parkinson disease
Authors: Racette BA, Gross A, Vouri SM, Camacho‐Soto A, Willis AW, Nielsen SS
Journal: Annals of Clinical and Translational Neurology; EPUB early release
PMID: N/A (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reanalysed the collected prescription data from the database of United States Medicare beneficiaries age 60–90 in 2009. They were looking for any immunosuppression medication and comparing its use with the incidence of Parkinson’s within their group. And to make the data more interesting and informative, the investigators broke the immunosuppressant medications down into six categories, which were:
- Calcineurin inhibitors (cyclosporine, tacrolimus)
- Inosine monophosphate dehydrogenase (IMDH) inhibitors (leflunomide, mycophenolate)
- Dihydrofolate reductase inhibitors (methotrexate)
- Biologics (abatacept, adalimumab, anakinra, certolizumab, etanercept)
- Corticosteroids (prednisone, prednisolone, methylprednisolone, dexamethasone, cortisone, hydrocortisone)
- Miscellaneous (hydroxychloroquine, sulfasalazine, mesalamine, interferon beta‐1a, thalidomide, lenalidomide, glatiramer acetate, flingolimod, and dimethyl fumarate).
Of these groups, two were significantly associated with a lower risk of PD:
- Corticosteroids (Relative risk = 0.80; 95% Confidence Interval 0.77–0.83)
- IMDH inhibitors (Relative risk = 0.64; 95% Confidence Interval 0.51–0.79)
This suggests that people who had been prescribed IMDH inhibitors had 1/3 less incidence of Parkinson’s.
Have been corticosteroids or IMDH inhibitors been associated with Parkinson’s before?
Last year (2017), a Korean research team published this research report:

Title: Hydrocortisone-induced parkin prevents dopaminergic cell death via CREB pathway inParkinson’s disease model
Authors: Ham S, Lee YI, Jo M, Kim H, Kang H, Jo A, Lee GH, Mo YJ, Park SC, Lee YS, Shin JH, Lee Y.
Journal: Sci Rep. 2017 Apr 3;7(1):525. doi: 10.1038/s41598-017-00614-w.
PMID: 28366931 (This article is OPEN ACCESS if you would like to read it)
Dr Lee and his colleagues began this study with cells were engineered to produce a bio-luminescent signal when a Parkinson’s-associated gene called PARKIN was activated. PARKIN is a Parkinson’s associated gene as genetic mutations in this gene can result in carriers developing a juvenile-onset/early-onset form of Parkinson’s disease.
The researchers then conducted an enormous screening experiment to find agents that turn on the PARKIN gene. They applied a library of 1172 FDA-approved drugs to these cells – one drug per cell culture – and looked at which cell cultures began to produce a bioluminescent signal. They found 5 drugs that not only made the cells bioluminescent, but also resulted in PARKIN protein being produced at levels 2-3 times higher than normal. Those drugs were:
- Deferasirox – an iron chelator (interesting considering our previous post)
- Vorinostat – a cancer drug (for treating lymphoma)
- Metformin – a diabetes medication
- Clindamycin – an antibiotic
- Hydrocortisone
Interesting things to note:
- Hydrocortisone is corticosteroid, which – according to the immunosuppression study discussed above – is a class of immunosuppressives that lowers the risk of Parkinson’s
- Hydrocortisone produced the highest levels of PARKIN (interestingly, hydrocortisone also did not increase the activity of PERK, an indicator of endoplasmic reticulum stress, while the other drugs did).
Hydrocortisone is the name for the hormone ‘cortisol’ when supplied as a medication.
Ok, so what is cortisol?
Cortisol is a glucocorticoid (a type of hormone) produced from cholesterol by enzymes in the cortex of the adrenal gland, which sits on top of the kidneys. It is produced in response to stress (physical or emotional)
The location of the adrenal glands. Source: Cancer
Cortisol helps us to deal with physical or emotional stress by reducing the activity of certain bodily functions – such as the immune system – so that the body can focus all of it’s energies toward dealing with the stress at hand.
Now generally, the functions of cortisol are supposed to be short-lived – long enough for the body to deal with the offending stressor and then levels go back to normal. But the normal levels of cortisol also fluctuate across the span of the day, with levels peaking around 8-9am:
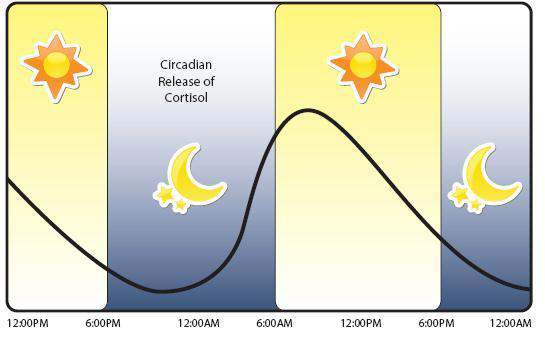
A graph of cortisol levels over the day. Source: HealthTap
Ok, so what did the Korean researchers do next?
Dr Lee and his colleagues gave the hydrocortisone drug to cell cultures which they then stressed (causing cell death). Hydrocortisone protected the cells from dying, and (importantly) it achieved this feat in a manner that was dependent on PARKIN activation. In cells that do not have PARKIN, hydrocortisone was found to have no effect on cell survival.
Next the researchers treated mice with hydrocortisone before they then modelled Parkinson’s using a neurotoxin (6-OHDA). Hydrocortisone treatment resulted in approximate a two-fold increase in levels of PARKIN within particular areas of the brain. Without hydrocortisone treatment, the mice suffered the loss of approximately 45% of their dopamine neurons. Mice pre-treated with hydrocortisone, however, demonstrated enhanced dopamine neuron survival.
The researchers concluded that a sufficient physiological supply of hydrocortisone was required for protection of the brain, and that hydrocortisone treatment could be further tested as a means of maintaining high levels of parkin in the brain.
So what do we know about cortisol in Parkinson’s?
So this is where the story gets interesting;

Title: Cortisol is higher in parkinsonism and associated with gait deficit.
Authors: Charlett A, Dobbs RJ, Purkiss AG, Wright DJ, Peterson DW, Weller C, Dobbs SM.
Journal: Acta Neurol Scand. 1998 Feb;97(2):77-85.
PMID: 9517856
The researchers who conducted this study were interested in the role of cortisol in Parkinson’s. They measured cortisol levels in the blood of 96 subjects with Parkinson’s and 170 control subjects. They found that cortisol levels were 20% higher in the subjects with Parkinson’s, and that MAO-B inhibitor treatment for Parkinson’s (Selegiline) reduced cortisol levels.
And MAO-B inhibitors are not the only Parkinson’s medication associated with reduced levels of cortisol:

Title: Acute levodopa administration reduces cortisol release in patients with Parkinson’s disease.
Authors: Müller T, Welnic J, Muhlack S.
Journal: J Neural Transm (Vienna). 2007 Mar;114(3):347-50.
PMID: 16932991
In this study the researchers found that cortisol levels started to decrease significantly just 30 minutes after L-dopa was taken. Whether this lowering of cortisol levels may have any kind of detrimental effect on Parkinson’s is yet to be determined and requires further investigation.
Is hydrocortisone or cortisol used in the clinic?
Yes it is.
Hydrocortisone is used to treat rheumatism, skin diseases, and allergies.
Hydrocortisone tablets. Source: Wisegeeks
Thus, there is the potential for another example of drug repurposing here. But the drug is not without side effects, which include:
- Sleep problems (insomnia)
- Mood changes
- Acne, dry skin, thinning skin, bruising or discoloration;
- Slow wound healing
- Increased sweating
- Headache, dizziness, spinning sensation;
- nausea, stomach pain
For the full list of potential side effects – click here.
Has Hydrocortisone been tested in the clinic for Parkinson’s?
Not that I am aware (and I am happy to be corrected on this).
There is, however, a clinical study currently being conducted in Oregon (USA) that is measuring Cortisol levels in people with Parkinson’s. The study is called CORT-PD, and the goal of the study is to measure stress levels in people with PD (Click here to read more about this clinical study).
Could immunosuppression be a good idea for the long term treatment of Parkinson’s?
Long time readers will know that I am very cautious when it comes to discussing treatments that could potentially be used for treating Parkinson’s. They will be aware that I like to point out where the dangers lie.
That said, you should be able to guess the tone of what is coming up next:
Long-term use of immunosuppressive drugs has the potential to cause immunodeficiency, which weakens your bodies ability to fight off infection. This situation can obviously cause increased susceptibility to opportunistic viruses, etc. But what is less obvious is that this can also lead to decreased cancer immunosurveillance. Our immune system is always on the look out for rogue cancerous cells and is continually getting rid of such trouble makers.
FACT: Approximately 10-20% of transplant recipients go on to develop at least one cancer after 10 years of immunosuppressive therapy (Click here, here and here for examples of this).
Thus, we need to be very careful in how we approach long-term use of immunosuppression as a component of any future treatment for Parkinson’s. Having said that, the monitoring for and treatment of cancers is improving every year and raising this concern may ultimately be me simply being overly cautious.
But it is better for people to be aware.
So what does it all mean?
An interesting analysis of a large medical health dataset has recently been published, and it highlights the decreased incidence of Parkinson’s in people being treated with particular types of immunsuppressive medications. Longer-term data needs to be looked at to determine whether this association is maintained over a wider period of time (than the 5 years of the current study), but the results are very interesting.
It will be intriguing to see if these associations are replicated in other large medical databases (such as those of Taiwan and the Scandiavian nations).
Whether some of these immunosuppressive drugs could be re-purposed for Parkinson’s still needs to be determined with further research. And given the risks associated with long-term use of immunosuppressant drugs and immunodeficiency/reduced cancer immunosurveillance such investigations will need to be extra vigilant.
What we need is another Gertrude Elion!
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from Nurseslabs








Thank You, Simon
LikeLike
You are welcome 🙂
LikeLike
I don’t really understand the overly cautious attitude towards experimental treatment. It’s not like standard treatment has much to offer PD patients. Heck, the medical professionals generally start off giving patients a hefty nocebo effect by telling the patients they have an incurable, degenerative disease that will lead to them losing function and dignity… why not let people get excited about a possible new treatment. I mean, I don’t really care if my chance of cancer goes up when I’m looking at being incontinent and in a wheel chair in a few years…
LikeLike
Hi Em,
Thanks for your comment. It’s a very fair question, but it is all too easy to take a reckless “let’s-just-try-it” attitude without considering the consequences. I think it is always better to balance the fragments of positive research news with some sober realities (even if, for example, the risk of cancer is only 10%). Better to make people aware, so they can be on the lookout for these things is the policy.
It is also important for readers to appreciate that some of the compounds we are discussing on this website have VERY powerful effects on the body, even if we are not consciously aware of them. And that has to be respected when attempting to re-purpose them. In addition, not all of these treatments work the same way for everyone (for example, a couple of years ago it was discovered that a significant portion of the Norwegian population have a genetic variation that means simple pain killers work differently for them – https://geminiresearchnews.com/2016/07/50000-norwegians-require-more-morphine-than-the-average-joe/).
And overriding all of this, is the situation I am desperately trying to avoid in sharing all of the information on this site. I never want to have to face the situation of someone having a negative outcome trying a compound that they read about on this website, being left worse off, and saying “But Simon said…”. I have read – with increasing alarm – comments on forums that taken what I have written completely out of context (and I mean COM-PLETE-LY!). It kind of freaks me out at times. And I have become more and more careful with what I write (and don’t write) as a result, which is probably to no ones benefit.
Thanks for the thought provoking comment. I really appreciated it. It is good to put these thoughts down in words. I hope you can understand my position – I certainly appreciate your side of the situation. And I am happy to discuss this further.
Kind regards,
Simon
LikeLike
Fair enough, and I appreciate the concern that people might do really dangerous things… but on the other hand, that is somewhat infantalizing the patients. I don’t have PD but my husband does. We’re both PhD scientists, and have read a lot of the literature. Obviously we are not MDs or PhDs in neuroscience, but we’ve been rather shocked by how much of the literature the neurologists are unaware of, and they are very dismissive when you bring up potential new or experimental treatments. An example: many people are reporting cessation of progression using low-dose Naltrexone, but trying to get this from your doctor, in the US at least, is impossible, and they will likely mock you or get angry with you for asking for it. (This is a summary from many reports from an alternative treatment group that I follow). Why? Yes, we’d all love to have a massive double-blind trial to prove something works, but we don’t have that time, and in the case of something like naltrexone, there is very little interest in even doing the study. If we understand the risks and potential side-effects, it seems it ought to be up to the patient to try something (especially when it has reasonable scientific logic behind it).
LikeLike
Interesting comments.
LikeLike
FINAL OUTCOME
The results of this study were negative for the efficacy of naltrexone for the treatment of impulse controls disorders (such as compulsive gambling, buying, sexual behavior and eating) in Parkinson’s disease (PD) using a clinician rating of general improvement, which was the primary outcome measure for the study. However, using a patient-completed, PD-specific assessment of impulse control disorder symptom severity, naltrexone treatment was associated with a significant decrease in symptoms compared with placebo treatment. The results of this preliminary study support further research with naltrexone or other opioid blockers for the treatment of these disorders in PD. In addition, other novel treatments to be considered for this condition include medications targeting glutamate (another brain chemical) and the possibility of deep brain stimulation.
LikeLike