|
Millions of dollars in research funding for Parkinson’s has been poured into the biology and function of just one hyperactive protein. It is called Leucine-rich repeat kinase 2 (or LRRK2). Genetic mutations in the gene that gives rise to this abnormal version of the protein can leave carriers with a higher risk of developing Parkinson’s. All of that research funding has resulted in an incredible leap forward in our understanding of LRRK2, which has further led to clinical trials focused solely on LRRK2. Mutations in the LRRK2 gene occur in only 1-2% of the Parkinson’s population, however, which has led to some complaints that too much research is being focused on only a small fraction of the people affected by PD. New research published this week could silence those complaints. In today’s post we will discuss a new report suggesting that independent of any genetic mutations, LRRK2 may actually play a role in idiopathic (or spontaneous) forms of Parkinson’s, which means that the treatments being developed for LRRK2 could be beneficial for a wider section of the PD community.
|

This is Sergey Brin.
He’s a dude.
You may have hear of him – he was one of the founders of a small company called “Google”.
Having changed the way the world searches the internet, he is now turning his attention to other projects.
One of those other projects is close to our hearts: Parkinson’s.
Why is he interested in Parkinson’s?
In 1996, Sergey’s mother started experiencing numbness in her hands. Initially it was believed to be a bit of RSI (Repetitive strain injury). But then her left leg started to drag. In 1999, following a series of tests and clinical assessments, Sergey’s mother was diagnosed with Parkinson’s.
It was not the first time the family had been affected by the condition though: Sergey’s late aunt had also had Parkinson’s.
Given this coincidental family history of this particular condition, both Sergey and his mother decided to have their DNA scanned for any genetic mutations that increase the risk of Parkinson’s. And they discovered that they were both carrying a genetic variation on their 12th chromosome, in a gene (a section of DNA) called PARK8 – one of the Parkinson’s-associated genes (Click here to read more about the genetics of Parkinson’s and the PARK genes).
The PARK8 gene is also known as Leucine-rich repeat kinase 2 (or LRRK2 – pronounced ‘lark 2’).
What is LRRK2?
Also known as ‘Dardarin‘ (from the Basque word “dardara” which means “trembling”), LRRK2 is an enzyme that has many functions within a cell – from supporting efforts to move things around inside the cell to helping to keep the power on (involved with mitochondrial function).
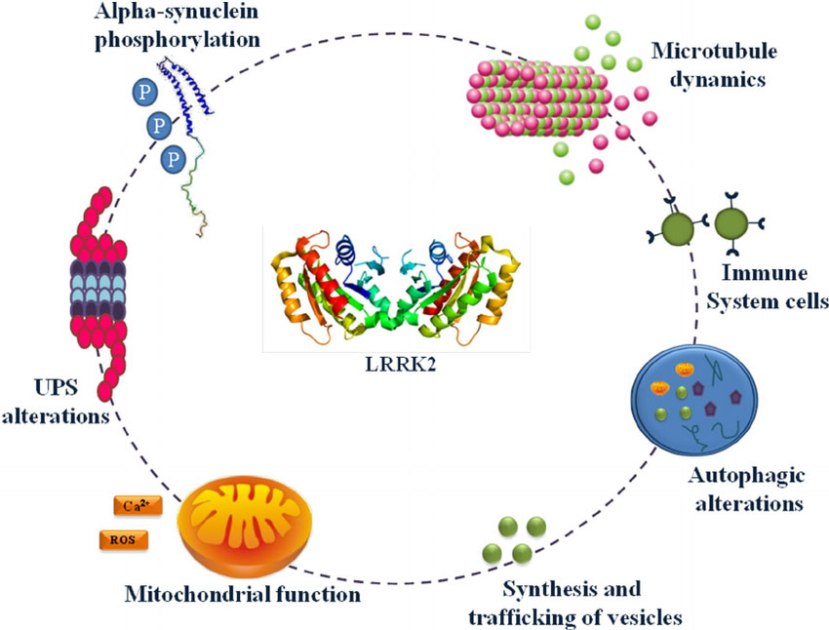
The many jobs of LRRK2. Source: Researchgate
The gene that provides the instruction for making the LRRK2 enzyme is made up of many different sections, each of which is involved with the different functions of the eventual protein.
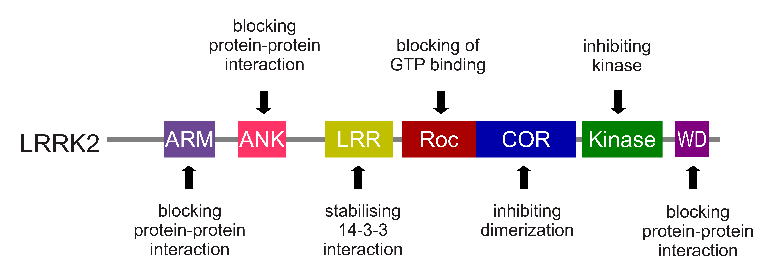
The regions and associated functions of the LRRK2 gene. Source: Intechopen
Genetic mutations (or variants) within the LRRK2 gene are recognised as being some of the most common with regards to increasing ones risk of developing Parkinson’s. But despite being one of the ‘most common’ genetic risk factors, variants in the LRRK2 gene are only present in approximately 1-2% of all cases of Parkinson’s. They are rather rare.
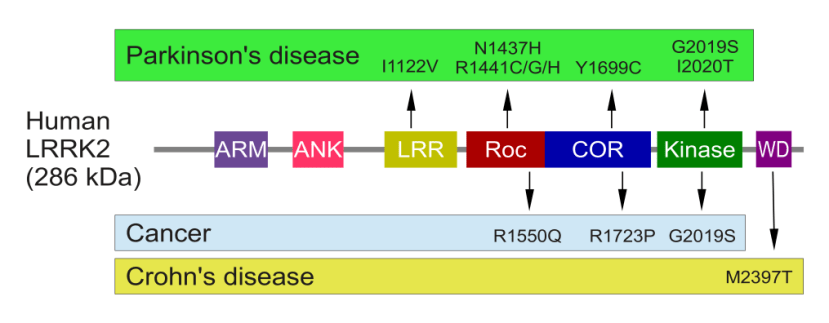
The structure of Lrrk2 and where various mutations lie. Source: Intech
As the image above suggests, mutations in the PARK8 gene are also associated with Crohn’s disease (Click here and here for more on this) – though that mutation is in a different location to those associated with Parkinson’s. And one particularly common Parkinson’s-associated LRRK2 mutation – called G2019S – is also associated with increased risk of certain types of cancer, especially for hormone-related cancer and breast cancer in women – Click here to read more about this. If you have a G2019S mutation, no need to panic, it is good to be aware of this association and to have regular check ups with your doctor.
The G2019S mutation (the name designates its location on the gene) is the most common LRRK2 mutation. In some populations of people it can be found in 40% of people with Parkinson’s (Click here to read more about this). But in general, LRRK2 mutations are very rare within the PD community.
What is interesting about this mutation is that it gives rise to a LRRK2 enzyme that is hyperactive.
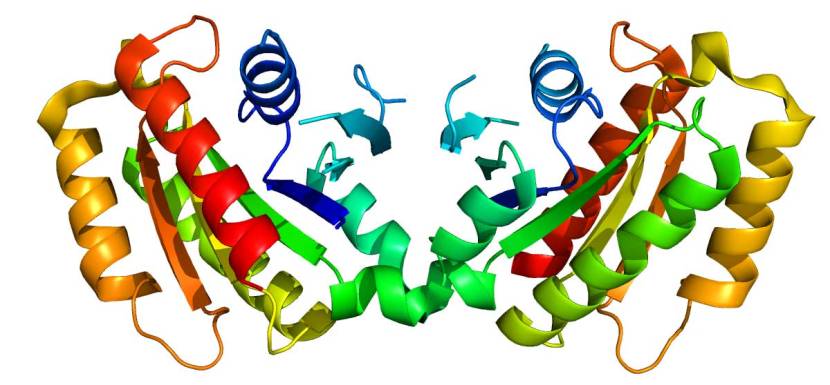
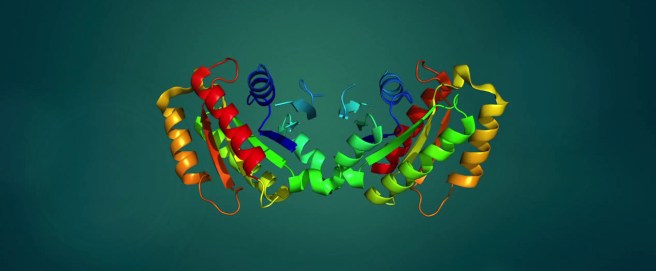
The structure of LRRK2 protein. Source: Wikipedia
As a protein, LRRK2 interacts with many different types of other proteins, and you can imagine that in a finely balanced environment like the cells that a mutant hyperactive form of LRRK2 is going to cause problems. And it is this hyperactive behaviour of the LRRK2 protein that is believed to be involved with the cellular dysfunction seen in LRRK2-associated Parkinson’s.
This hyperactive version of LRRK2 has been the focus of a great deal of research – some of which is out of this world (Click here to read more about this) – and pharmaceutical companies have been rushing to develop LRRK2 inhibitors with the goal of using them to treat people with Parkinson’s (Click here to read more about this).
And this is where Mr Brin comes back into the picture.
Source: Desktop-backgrounds
You see: Sergey Brin and his mother have the G2019S variant.
Now, to be very clear: Not everyone with this LRRK2 variant will go on to develop Parkinson’s. But Sergey has apparently decided that his chances are 50:50. And he does not appear to be wanting to take any chances.
Being one of the founders of a large company like Google has left Sergey with considerable resources at his disposal. And he has chosen to focus some of those resources on LRRK2 research (call it an insurance policy). He has done this via considerable donations to groups like the Michael J Fox foundation (he gives approximately $20 million per year in donations to the MJFF).
You will appreciate that this has shifted the needle on ‘LRRK2’ research rather considerably – if you go to the Pubmed search engine and type in ‘LRRK2’ you will see that there are approximately 1930 reports highlighted (as of 25th July 2018).
In 2004, there were only 3 reports.
But if LRRK2 genetic mutations only affect a small proportion of the Parkinson’s community, is this not a case of disproportionate distribution of research funds?
Until recently, this may have been a fair question. But in the last year or so, there has been a steady stream of research reports that suggest LRRK2 over activity in the Parkinson’s community may not be limited to those individuals with particular LRRK2 genetic variations.
For example, earlier this year (2018), this report was published:
Title: LRRK2 Expression Is Deregulated in Fibroblasts and Neurons from Parkinson Patients with Mutations in PINK1
Authors: Azkona G, López de Maturana R, Del Rio P, Sousa A, Vazquez N, Zubiarrain A, Jimenez-Blasco D, Bolaños JP, Morales B, Auburger G, Arbelo JM, Sánchez-Pernaute R.
Journal: Mol Neurobiol. 2018 Jan;55(1):506-516.
PMID: 27975167 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers the researchers were interested in another gene that is associated with Parkinson’s – called PTEN-induced putative kinase 1 (or simply PINK1). PINK1 is a protein that has a number of different functions in cells, but it is best understood in the context of cellular waste disposal (Click here to read a recent post that explains what PINK1 does).
In their study, the researchers increased levels of PINK1 in cells and they noticed something interesting about LRRK2:
As levels of PINK1 increased, levels of LRRK2 activity decreased
And when they lowered PINK1 levels in cells, guess what happened: LRRK2 levels of activity went up!
Thus, this study identifies a novel function for PINK1 – modulating the levels of LRRK2. It also suggested that people with PINK1-associated Parkinson’s may also benefit from new LRRK2-targeting therapies that are currently being developed.
Great. So how many people in the Parkinson’s community have PINK1 genetic mutations?
Less than those with LRRK2 mutations.
So it is still a case of a very small fraction of the community potentially benefiting from all this research and these new treatments?
Well, that may have been a fair question until this week, when this research report was published:
Title: LRRK2 activation in idiopathic Parkinson’s disease
Authors: Di Maio R, Hoffman EK, Rocha EM, Keeney MT, Sanders LH, De Miranda BR, Zharikov A, Van Laar A, Stepan AF, Lanz TA, Kofler JK, Burton EA, Alessi DR, Hastings TG, Greenamyre JT
Journal: Science Translational Medicine 25 Jul 2018: 10 (451), eaar5429.
PMID: 30045977
In this study, the investigators looked at LRRK2 activity on sections of postmortem brain from 7 individuals who had passed away with idiopathic (or spontaneous) Parkinson’s and compared them with section of brain from 8 healthy controls. Idiopathic Parkinson’s is any form of Parkinson’s that can not be immediately associated with a particular causal factor. Most forms of Parkinson’s are idiopathic, while individuals with Parkinson’s that results from genetic mutations are considered non-idiopathic.
The researchers used sections of brain from an area called the midbrain, which contains a region called the substantia nigra – the area of the brain where the dopamine-producing cells reside. These cells are particularly affected in Parkinson’s, with approximately 50% of them being lost by the time of diagnosis.
The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinson’s brain (right). Source:Memorangapp
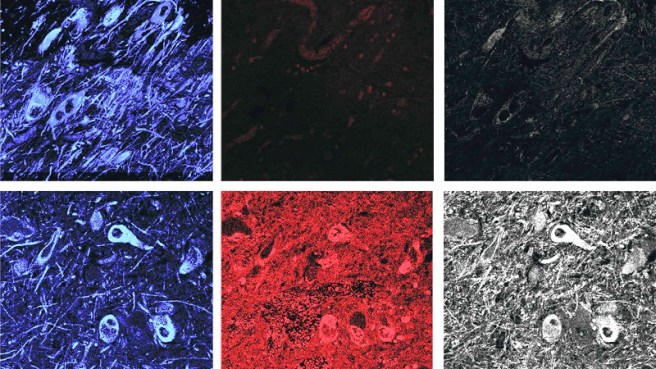
To the surprise of the researchers the remaining dopamine neurons of the cases of idiopathic Parkinson’s showed about a 4-6 fold increase in markers of LRRK2 activity (compared with the control brains). This suggested to the researchers that normal LRRK2 protein is highly activated in Parkinson’s.
Control brains (top) have low levels of LRRK2 activity (shown in red and gray in the center and right panels), while Parkinson’s brains (bottom) have high levels. The far left panels show dopamine neurons in blue. Source: STAT
Interestingly, this increase in LRRK2 levels was not isolated to neurons in the idiopathic Parkinson’s brain. Microglia – the resident immune cells in the brain also exhibited twice the normal levels of LRRK2 activity.
Different types of cells in the brain. Source: Dreamstime
Next, the investigators looked at what happens to LRRK2 activity levels in an animal model of Parkinson’s – specifically, a model that involves a pesticide toxin (called Rotenone – Click here to read a previous SoPD post on pesticides). When they looked at dopamine neurons in animals treated with rotenone, the researchers observed a 10-fold increase in certain markers of LRRK2 activity. They also reported disruption of cellular waste disposal (or autophagy) markers caused by rotenone treatment.
They also looked in a second animal model of Parkinson’s – this one involving very high levels of normal human alpha synuclein protein (caused by injection of genetically engineered viruses), and again, the researchers observed a 10-fold increase in certain markers of LRRK2 activity.
In a third test, the researchers caused oxidative stress on cells and observed an increase in LRRK2 activity, which could be reduced by treatment with anti-oxidants.
All of these results collectively suggest that the role of LRRK2 in Parkinson’s is potentially a lot greater than we previously thought. By using many of the factors associated with the development of Parkinson’s – such as exposure to pesticides, aggregated alpha synuclein protein, and oxidative stress – the researchers observed increased levels of LRRK2 activity.
Interesting. So what does it all mean?
No wait, there’s more!
Importantly, the researchers conducting this study gave some of the rotenone-treated rodents a drug called PF-360 (from the pharmaceutical company Pfizer).
PF-360 is a LRRK2 inhibitor.
This is a compound that bind to the LRRK2 protein and prevent it was doing its job.
And by treating the animals with PF-360, the investigators were able to reduce the levels of LRRK2 activity and improve the disturbed markers of autophagy.
Wow. Are there any LRRK2 inhibitors in clinical trials at the moment?
Yes there are.
The most advanced of the LRRK2 inhibitors being developed at the moment is DNL-201, which is being clinically tested by a San Francisco-based biotech firm called Denali Therapeutics.
Founded in 2013 by a group of former Genentech executives, Denali is focused on developing novel therapies for people suffering from neurodegenerative conditions. Although they have product development programs for other condition (such as Amyotrophic Lateral Sclerosis and Alzheimer’s), Parkinson’s appears to be their primary interest.
And their first drug, DNL-201, is a selective, orally-available, brain-penetrant, reversible small molecule LRRK2 inhibitor which is currently being tested in Phase I ‘safety’ trials in healthy adults (we have previously discussed the Phase I clinical trial results for DNL-201 – Click here to read that post).
On top of the ongoing testing of DNL-201, Denali submitted a Clinical Trial Authorisation (or CTA) for DNL-151 (another LRRK2 inhibitor) to the Netherlands Health Authority in October 2017 and it was accepted in November. The company dosed its first participant in a clinical trial of DNL-151 in March of this years and that trial is ongoing.
After completion of the Phase I clinical trials of both DNL-201 and DNL-151, Denali next plans to start testing one of the drugs (DNL-201 or DNL-151) in a Phase Ib study that will involve testing the safety of the drug in people with LRRK2-associated Parkinson’s.
We are currently awaiting the final results of the DNL-201 phase I trial (some time in the next couple of months – expect a SoPD post on those results).
In addition to Denali, pharmaceutical company GlaxoSmithKline recently announced a $300 million equity investment in the DNA analysis company 23andMe (Click here to read more about this).

The companies are forming a 4 years partnership, which will involve contributing preclinical programs on a 50-50 basis. As part of that deal, GSK is contributing its LRRK2 inhibitor program, and they maybe hoping to use 23andMe’s database of people who know their LRRK2 genetic status. Such access may help their Parkinson’s clinical program enroll participants quicker in their planned clinical trial of their LRRK2 inhibitor.
So what does it all mean?
Genetic mutations in the LRRK2 gene are a major risk factor for Parkinson’s.
In many cases of these genetic variants, the resulting LRRK2 protein is hyperactive. Many cells may not be able to handle this constantly active form of LRRK2 and it is believed that this results in the cell dying. New research this week suggests that high levels of LRRK2 activity may not be associated solely with people that carry LRRK2 genetic mutations, but it may be a common feature of Parkinson’s in general.
By reducing the level of LRRK2 activity in vulnerable cells, researchers had previously hoped that they would be able to slow down the progression of Parkinson’s in individuals with LRRK2 mutations. But the new research published this week suggests that such a treatment approach may be beneficial to many folks in the Parkinson’s community.
Thus targeting the hyperactive version of LRRK2 with carefully designed inhibitors represents a promising therapeutic approach. One concern regarding this inhibitor idea, however, is that LRRK2 is involved with many functions and by blocking some of them, are there going to be debilitating side effects from the use of such a drug (thus far from the Denali study this does not appear to be the case).
Another question that has not been properly addressed yet is what (if any) effect LRRK2 inhibitors like DNL-201 have on LRRK2’s sibling LRRK1 (which we discussed in a previous post – Click here to read that post). In this new study, the resesarchers did not look at levels of LRRK1 activity to determine if it too is increased in the PD brain or models of Parkinson’s. It would be interesting (and ultimately necessary) to address this matter and also determine if DNL-201 is affecting LRRK1.
The take home message of this new study, however, is that LRRK2 appears to be more involved in the biology of Parkinson’s (not just specific genetic forms of the condition) than we previously thought, which bodes well for the overall community as novel therapies targeting this protein are being developed.
EDITOR’S NOTE: Denali Therapeutics and GlaxoSmithKline are publicly traded companies. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. Denali Therapeutics and GlaxoSmithKline have not requested that this material be produced, nor has the author had any contact with the companies or any associated parties. This post has been produced for educational purposes only.
The banner for today’s post was sourced from Youtube.







Thank you. Another informative article that is easy to understand.
LikeLike
Hi AllFramed,
You’re welcome. Glad you like the content.
Kind regards,
Simon
LikeLike
Excellent. I am a Physiologist and am working with a client to help reduce anxiety and tremors. Guided visualisation helps meditation, mindfulness etc. However, he likes to know the science and neuro physiology behind what is going on. So, Thanks!
LikeLike
Hi David,
You are very welcome. Thanks for the comment.
Kind regards,
Simon
LikeLike