|
It is often said that Parkinson’s is a ‘distinctly human’ condition. Researchers will write in their reports that other animals do not naturally develop the features of the condition, even at late stages of life. But how true is this statement? Recently, some research has been published which brings into question this idea. In today’s post, we will review these new findings and discuss how they may provide us with a means of testing both novel disease modifying therapies AND our very notion of what Parkinson’s means.
|
 Checking his Tinder account? Source: LSE
Checking his Tinder account? Source: LSE
Deep philosphical question: What makes humans unique?
Seriously, what differentiates us from other members of the animal kingdom?
Some researchers suggest that our tendency to wear clothes is a uniquely human trait.
![]() The clothes we wear make us distinct. Source: Si-ta
The clothes we wear make us distinct. Source: Si-ta
But this is certainly not specific to us. While humans dress up to ‘stand out’ in a crowd, there are many species of animals that dress up to hide themselves from both predator and prey.
A good example of this is the ‘decorator crab’ (Naxia tumida; common name Little seaweed crab). These creatures spend a great deal of time dressing up, by sticking stuff (think plants and even some sedentary animals) to their exoskeleton in order to better blend into their environment. Here is a good example:
Many different kinds of insects also dress themselves up, such as Chrysopidae larva:
 Dressed for success. Source: Bogleech
Dressed for success. Source: Bogleech
In fact, for most of the examples that people propose for “human unique” traits (for example, syntax, art, empathy), mother nature provides many counters (Humpback whales, bower birds, chickens – respectively).
So why is it that we think Parkinson’s is any different?
Wait a minute. Are there other animals that get Parkinson’s?
Well…., that depends on one’s definition of Parkinson’s.
If we go with the classic characteristics of the condition – that it is associated with:
- Aging
- The clustering (or aggregation) of the protein alpha synuclein
- The loss of dopamine neurons
Then new research suggests that humans are not the only animals to get “Parkinson’s”. This research report was recently published:
 Title: Age- and a-Synuclein-Dependent Degeneration of Dopamine and Noradrenaline Neurons in the Annual Killifish Nothobranchius furzeri.
Title: Age- and a-Synuclein-Dependent Degeneration of Dopamine and Noradrenaline Neurons in the Annual Killifish Nothobranchius furzeri.
Authors: Matsui H, Kenmochi N, Namikawa K.
Journal: Cell Rep. 2019 Feb 12;26(7):1727-1733.e6. doi: 10.1016/j.celrep.2019.01.015.
PMID: 30759385 (This report is OPEN ACCESS if you would like to read it)
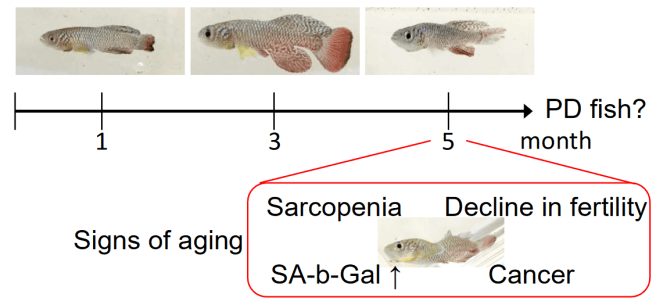
In this study, the researchers assessed the dopamine system of aged annual killifish (Nothobranchius furzeri). These fish have a short life span and exhibit a rather rapid aging process.
 Nothobranchius furzeri. Source: Noldus
Nothobranchius furzeri. Source: Noldus
This annual killifish is a species native to Mozambique, which has a very short life span of between 3 to 6 months. It is among the shortest-lived species of vertebrates. And given such a short lifespan, these fish are also among the fastest known sexual maturers (developing from hatchling to an breeding adult in only 14 days!). They don’t waste anytime.
N. furzeri fish reach a ful length of 6.5 cm (2.6 inches), and curiously 21% of the species’ DNA is made up of tandom repeats (a section of DNA where a pattern of one or more nucleotides is repeated), which is ridiculously high and believed to play a role in their short life spans (human DNA =3% tandom repeats).
Given their short life spans, researchers have previously used this species of fish to study aging, and now some Japanese researchers have asked whether these fish could be used to study Parkinson’s (the risk of which is associated with aging).
 Source: Cell
Source: Cell
The researchers firstly looked at the dopamine system of N. furzeri fish from 1, 3 and 5 months of age.
What is the dopamine system? And why look at it?
Dopamine is a chemical that is produced in our brains by specific populations of neurons, which we refer to as the “dopamine system”. Dopamine is necessary for many aspects of normal neurological functioning, particularly the regulation of movement.
The loss of these dopamine producing cells is one of the most characteristic features of the Parkinsonian brain, and their gradual disappearance is associated with the initiation of the motor symptoms of the condition.
Dopamine neurons in the human brain produce a dark pigment (called neuromelanin) which allows you to visualise the dopamine population with your bare eye. As you can see in the image below, the Parkinsonian brain has less dark pigmented cells (in the substantia nigra region of the midbrain). As dopamine neurons are lost, so too is the dark pigment of neuromelanin.
 The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinsonian brain (right). Source:Memorangapp
The dark pigmented dopamine neurons in the substantia nigra are reduced in the Parkinsonian brain (right). Source:Memorangapp
By looking at thedopamine system of the N. furzeri fish at 1, 3, and 5 months of age, the researchers found that there was a significant reduction in the number of dopamine neurons (labelled using a marker called Tyrosine hydroxylase or TH) over the life times of these fish. And dopamine (or DA) neurons were not the only cells affected, the researchers also reported significant loss of noradrenergic (or NE) neurons – another type of neuron that is badly affected in Parkinson’s.
 Source: Cell
Source: Cell
This result was interesting, but then the researchers observed something else that is associated with Parkinson’s: the clustering or aggregation of a protein called alpha synuclein.
What is alpha synuclein?
Alpha synuclein is one of the most common proteins in our brains. It makes up about 1% of all the protein in a neuron. When alpha synuclein protein is produced by a cell, it normally referred as a ‘natively unfolded protein’, in that is does not really have a defined structure. When it is first produced, alpha synuclein will look something like this:
 Alpha synuclein. Source: Wikipedia
Alpha synuclein. Source: Wikipedia
In this form, alpha synuclein is considered a monomer – which is a single molecule that can bind to other molecules. When it does bind to other alpha synuclein proteins, they form an oligomer (a collection of a certain number of monomers in a specific structure). In Parkinson’s, alpha synuclein also binds (or aggregates) to form what are called ‘fibrils’.

Microscopic images of Monomers, oligomers and fibrils. Source: Brain
And it is believed that the oligomer and fibril forms of alpha synuclein protein that cluster and aggregate together, and then go on to form what we call Lewy bodies.
 Parkinson’s associated alpha synuclein. Source: Nature
Parkinson’s associated alpha synuclein. Source: Nature
What are Lewy bodies?
Lewy bodies are dense circular clusters of alpha synuclein protein (and other proteins) that are found in specific regions of the brain in people with Parkinson’s (Click here for more on Lewy bodies).
 A cartoon of a neuron, with the Lewy body indicated within the cell body. Source: Alzheimer’s news
A cartoon of a neuron, with the Lewy body indicated within the cell body. Source: Alzheimer’s news
The aggregated alpha synuclein protein, however, is not limited to just the Lewy bodies. In the affected areas of the Parkinsonian brain, aggregated alpha synuclein can be seen in the branches (or neurites; see black arrow in the image below) of cells. In the image below, alpha synuclein has been stained brown on a section of brain from a person with Parkinson’s.
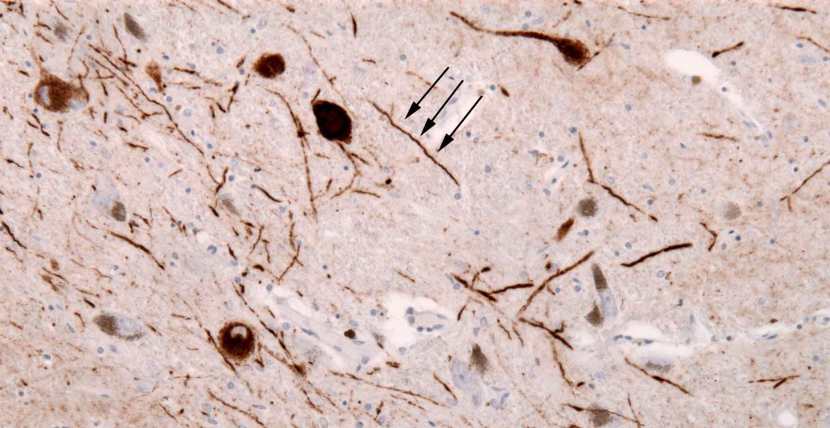
Examples of Lewy neurites (stained in brown; indicated by arrows) from a human brain. Source: Wikimedia
The Japanese researchers found that N. furzeri had abundant levels of alpha synuclein protein and that over time, it gradually aggregated into Lewy body-like structures. Interestingly, at 1 month, these structures were restricted to the spinal cord, but they gradually spread throughout the brain at 3 and 5 months.
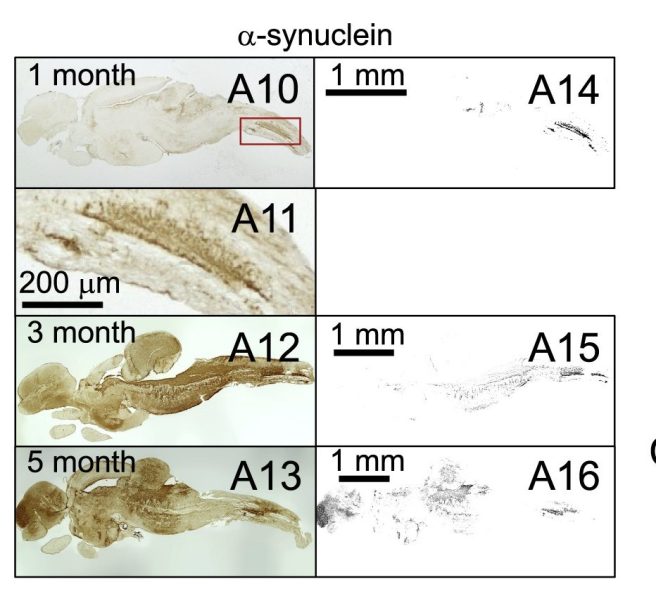 Alpha synuclein staining (in brown) of N. furzeri fish at different ages. Source: Cell
Alpha synuclein staining (in brown) of N. furzeri fish at different ages. Source: Cell
Naturally this situation made the researchers ask: what would happen if we remove the alpha synuclein?
So they genetically engineered N. furzeri fish (using CRISPR-Cas9 gene editing technology – Click here to read more about that) that have no alpha synuclein. And the result? Well, the fish with no alpha synuclein did not live longer,…
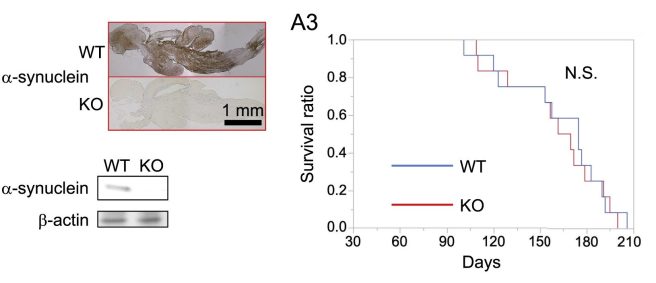 N. furzeri fish with no alpha synuclein (KO) did not live longer. Source: Cell
N. furzeri fish with no alpha synuclein (KO) did not live longer. Source: Cell
…but this genetic correction (and absence of alpha synuclein) did result in both the dopamine (DA) neurons and noradrenergic (NE) neurons surviving longer. There were significantly more DA and NE neurons at the 5 months of age stage. In the bar graphs below, note that the N. furzeri fish with no alpha synuclein (KO) have similar numbers of DA and NE neurons are 5 months (graphs on the right) to what they had at 1 month of age (graphs on the left).
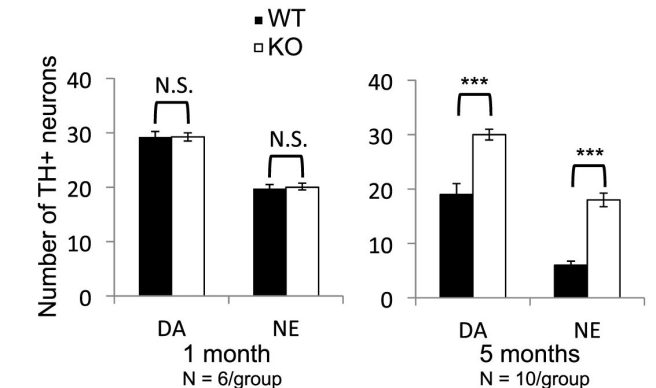 Alpha synuclein absence rescues neurodegeneration. Source: Cell
Alpha synuclein absence rescues neurodegeneration. Source: Cell
Do the fish have movement issues like Parkinson’s?
The researchers looked at this but it was difficult for them to differentiate between the motor issues that could be related to 40% loss of dopamine neurons, and all of the other issues associated with rapid aging (such as muscle pathology).
They summarized their study by suggesting that “use of N. furzeri model established in this study may contribute to identification of novel pathological mechanisms in PD. Furthermore, the small size of this fish will facilitate conducting drug trials and screening for PD therapy”
Interesting, but do other animals like primates develop Parkinson’s?
For a long time, it was argued that other primates do not develop any of the features of Parkinson’s (unless exposed to environmental toxins), even in old age. But recently that idea has been challenged as well.
In 2012, this research report was published:
 Title: Increased α-synuclein phosphorylation and nitration in the aging primate substantia nigra
Title: Increased α-synuclein phosphorylation and nitration in the aging primate substantia nigra
Authors: McCormack AL, Mak SK, Di Monte DA.
Journal: Cell Death Dis. 2012 May 31;3:e315.
PMID: 22647852 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers compared dopamine neurons in the substantia nigra of 4 young adult (<10yrs of age) vs 3 old (>16 yrs of age) squirrel monkeys. Their analysis suggested an increase in both normal and modified forms of alpha synuclein protein with aging. There were more dopamine neurons with high levels of alpha synuclein at the older time points.
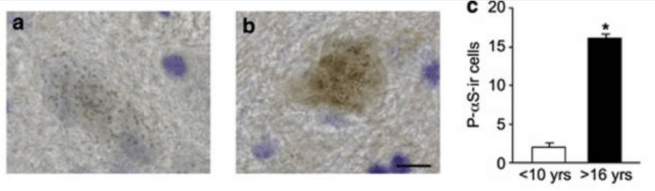 Source: PMC
Source: PMC
But the investigators did not find any aggregated alpha synuclein in these brains.
Perhaps it’s different in a different species of monkey?
Good question. And this has been explored by other researchers, such as in this report:
 Title: Age-dependent α-synuclein aggregation in the Microcebus murinus lemur primate
Title: Age-dependent α-synuclein aggregation in the Microcebus murinus lemur primate
Authors: Canron MH, Perret M, Vital A, Bézard E, Dehay B.
Journal: Sci Rep. 2012;2:910.
PMID: 23205271 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers looked at the brains of gray mouse lemur (Microcebus murinus) – these are some of the smallest primates (they look more like a possum than a chimpanzee). Previously, it had been reported that abundant levels of Alzheimer’s-associate β amyloid plaques and Tau pathology had been found in the aged brains of these creatures (Click here, here and here to read more about that), so naturally the investigators behind this study were curious to investigate Parkinson’s-associated alpha synuclein.
The analysis of the investigators suggests that yes, there is aggregation of alpha synuclein in the brains of these primates, but only in the aged animals (particularly in the cortex regions of the brain – see panels D & J in the image below, where alpha synuclein is stained in brown).
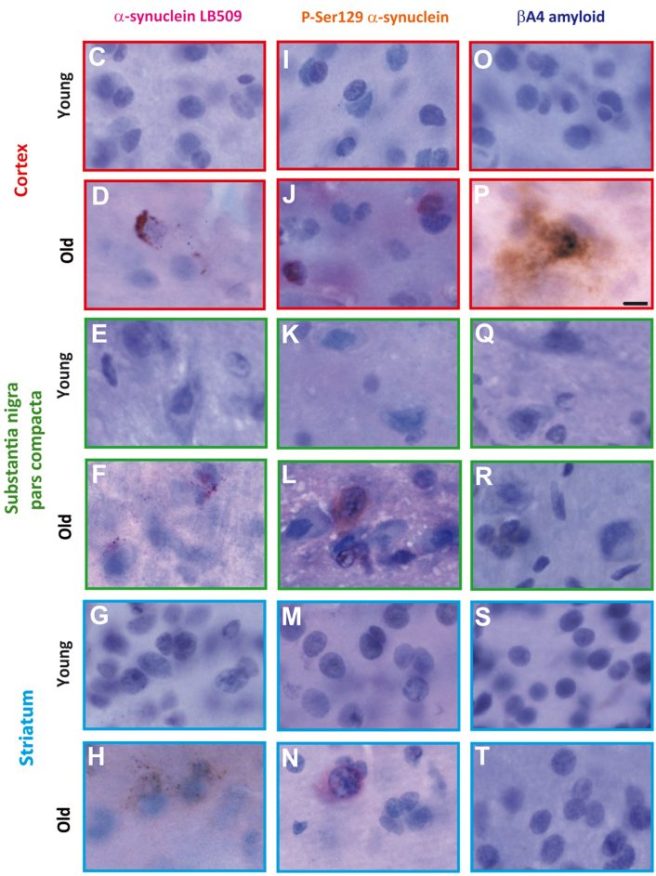 Source: PMC
Source: PMC
And this was not the only species of non-human primate that has exhibited alpha synuclein protein aggregates.
In 2016, this research report was published:
 Title: α-Synuclein aggregation in the olfactory bulb of middle-aged common marmoset
Title: α-Synuclein aggregation in the olfactory bulb of middle-aged common marmoset
Authors: Kobayashi R, Takahashi-Fujigasaki J, Shiozawa S, Hara-Miyauchi C, Inoue T, Okano HJ, Sasaki E, Okano H.
Journal: Neurosci Res. 2016 May;106:55-61.
PMID: 26643383
In this study, the researchers compared alpha synuclein levels in the brains of young (2-years-old) and middle-aged (6-years-old) common marmoset (Callithrix jacchus) primates. The results reported alpha synuclein aggregation were present in the olfactory bulb (the smell oriented part of the brain) in middle-aged animals, but not the young brains.
The researchers also found alpha synuclein protein accumulation in the hippocampus of the middle-aged group. The hippocampus is a regions associated with memory formation, and alpha synuclein in this region was mainly found in the tips of the branches of the neurons, as opposed to the main cell body. Interestingly, they did not find any alpha synuclein aggregation in the substantia nigra region (where the dopamine neurons reside) at any time point.
But the finding of alpha synuclein aggregates in these non-human primates return us to square one: is Parkinson’s a distinctly human condition. I would suggest that jury is still out.
It has to be remembered that all of these animals are being maintained in sterile laboratory conditions. It could be interesting to investigate Parkinsonian features of the brains of wild (or zoo) primates. In addition, the definition of “aged” in these studies may not reflect the human version of aged. It is possible that these non-human primate brains are not under that same pressures as the human equivalent (Click here to read more about this).
So what does it all mean?
One of the biggest problems in preclinical research is the models we use to test novel therapies. Because other animals ‘do not develop Parkinson’s’, we have been limited to artificial models involving neurotoxins or ridiculously high levels of pathogenic proteins. Regardless of how one feels about the use of animals in research, the basic biology of all drugs in the clinic begins with pharmacology in these creatures to determine if they are safe before we shift to human testing. This process would be easier if we had proper models of Parkinson’s.
Reading through this post, one can not help but again note that our ideas about “Parkinson’s” still require some work. While there appears to be a strong genetic component to young onset Parkinson’s (diagnosed under the age of 40 years of age), idiopathic Parkinson’s – that is the spontaneous version of the condition – still needs an explanation.
It will be interesting to see independent replication of the Nothobranchius furzeri fish research, and then use of that model for the screening of novel therapeutic approaches for Parkinson’s.
The banner for today’s post was sourced from Alto

Thanks for this most interesting article!
These laboratory-raised primates might be suffering from nutritional deficiencies which humans also suffer from, but which wild primates don’t. They might also be suffering from low vitamin D due to lack of sun exposure, as do most humans. Comparative studies with wild and lab primates would be highly valuable. Vitamin D and boron levels should be assessed, along with calcium, potassium and various other minerals.
I don’t have time or space here to give proper references, but I suggest that boron deficiency is a common cause of PD, AD and other neurodegenerative diseases involving Tau and/or alpha-synuclein – perhaps the biggest single cause of all. It seems that high levels of these misfolded proteins and/or the brain’s inability to cope with higher levels of them is the cause of the lasting damage to neurons. (Many people have plenty of Lewy bodies, but their neurons seem to cope just fine.)
Chronic inflammation is strongly implicated in AD, PD, and as far as I know similar pathologies such as dementia with Lewy bodies. Research indicates that boron is essential to proper regulation of inflammatory processes.
AD and PD sufferers are more likely than the general population to also suffer from osteoporosis.
https://www.sciencedirect.com/science/article/pii/S1873959817302466
https://www.aan.com/PressRoom/Home/PressRelease/353
https://link.springer.com/article/10.1007/s00415-012-6697-x
My reading makes me think that osteoporosis is caused, to a large degree, by boron deficiency – though this is not widely enough recognised. I recall that the bone density in a study group was only correlated to a few percent with the subject’s calcium intake. Boron is known for helping people cope with inadequate vitamin D levels, and adequate vitamin D is well known to be essential to maintaining high bone density as we age.
Boron supplementation is widely used to protect against arthritis. This article gives some background to the work of Rex E. Newnham, who researched it extensively and had a thriving business with borax tablets, based entirely on word-of-mouth:
Click to access Boron-and-Arthritis.pdf
https://www.tandfonline.com/doi/abs/10.3109/13590849009003147
I believe that potassium and boron are the two most significant seriously under-recognised nutritional deficiencies affecting humans.
I also suspect that boron deficiency is the primary or sole cause of tooth-resorption in domestic cats. The pattern of feline tooth resorption has become well recognised and highly prevalent in recent decades but was rarely observed in the 1950s. The etiology is officially unknown, and the advice of vets is to pull out all the teeth as they become destroyed by the resorptive processes, which accompanies periodontal disease. This can leave the elderly cat with no teeth at all. My wife Tina and I are giving our 5yo ginger tabby boron supplements and so far, by our estimation (the vet hasn’t seen him since he removed the first two affected teeth last year) his gums and teeth look better.
https://dspace.library.uu.nl/bitstream/handle/1874/42710/booij-vrieling.pdf (PhD thesis on Tooth Resorption in Cats.)
https://www.researchgate.net/publication/236192416_Boron_Enhances_Odontogenic_and_Osteogenic_Differentiation_of_Human_Tooth_Germ_Stem_Cells_hTGSCs_In_Vitro
I can’t find the research which showed that cats which are let outside are less at risk of feline tooth resorption. They would eat prey which is not boron depleted while indoor cats rely entirely on more likely to be boron depleted grain-fed chicken, bovine and pig meat byproducts of the mass-scale human food production system. (Fish?)
There is research linking periodontal disease with inadequate boron:
https://www.sciencedirect.com/science/article/pii/S1991790216300198
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4712861/
There’s more than enough reasons to take supplemental boron.
I strongly suspect that doing so will protect against AD, PD etc. It would take a few decades of research to establish this. AD, PD etc. are really similar diseases – its just a matter of which groups or neurons are damaged or destroyed, which is a minor distinction since the effects are utterly devastating in the end.
Just because the pathology is varied and complex, and just because there are dozens of mid-level mechanisms which can be studied and perhaps altered with drugs, doesn’t mean that the overall primary root cause is not a simple nutritional deficiency. This can be the case despite all the tantalising clues from genetic patterns which result in greater risk. With adequate nutrition, most of these predisposing genetic factors might only lead to minor levels of disease.
If you got thousands of cars, old and new, made by different companies – with different engines using carburetors and fuel injection, and with different firmware in the fuel injection systems – and ran them all on the same mix of contaminated fuel for a few years, then you would get quite a variation in symptoms – by make, engine type, temperature, driving conditions etc. The spluttering, lack of power, spark-plug fouling, lubrication oil fouling, corrosion, valve-burning and other mechanisms of engine destruction would vary enormously. Mechanics could tweak all sorts of things to reduce the symptoms, and spend lots of time comparing notes on how to do this. But if they knew the problem was contaminated fuel, than that is what needs to be fixed, and it would be unethical to charge the customer for adjusting the carbies, replacing valves, changing parameters in the engine management system etc.
LikeLike
Thanks for trying to make this basic research almost comprehensible to the lay person. i would like to hear more about functional or behavioral correlates of these abnormal cells. I m sure everyone else would like that too.
LikeLike
Fascinating post. In additional to the importance of animals for modeling and testing, the ways in which they differ from humans in expressing what for humans might be signs of nascent PD, without however developing the illness itself, could give us insight into the factors that cause the disease to become fully expressed in humans. It could be factors in the human vs. animal environment, or the specific stresses placed on the human brain, or the extension of our lifespans beyond the lengths which our evolution has prepared us to easily endure.
LikeLike
Simon, This is a very interesting post. So did someone tried to feed the aged fish with the l-dopa to see if it would move better?
LikeLiked by 1 person
Those fish are cute; I’d like to get some (and maybe put fish doxy in their tank and see if they live longer).
LikeLike
This topic reminded me on an interesting Ted talk on human vs animal brains by Suzana Herculano-Houzel (2013):
She found that primates proportionally have more neurons than other vertebrates. Further, humans have larger brains relative to body size than the other great apes since we have behavioral adaptations that allow us to extract more calories from our environment. Cooking is discussed in the video [I think our diet is perhaps(?) less specialized and more flexible than the other great apes, and tool making is important for hunting and processing of foods].
LikeLike