|
The recent documents filed with the U.S. Securities and Exchange Commission by the biotech firm Prevail Therapeutics provides interesting insight into the bold plans of this company which was only founded in 2017. Even more recent news that the U.S. Food and Drug Administration (FDA) has accepted the company’s Investigational New Drug (IND) application for its lead experimental treatment – PR001 – suggests that this company is not wasting any time. PR001 is a gene therapy approach targeting GBA-associated Parkinson’s. In today’s post, we will discuss what GBA-associated Parkinson’s is, how Prevail plans to treat this condition, and discuss what we know about PR001.
|
 Caterina Fake. Source: TwiT
Caterina Fake. Source: TwiT
The title of this post comes is from a quote by Caterina Fake (co-founder of Flickr and Hunch (now part of Ebay)), but it seemed appropriate.
This post is all about dreaming big (curing Parkinson’s), the struggle to get the research right, and to create a biotech company: Prevail Therapeutics.
What is Prevail Therapeutics?
Prevail is a gene therapy biotech firm that was founded in 2017.
 The company was founded by Dr Asa Abeliovich:
The company was founded by Dr Asa Abeliovich:
 Dr Asa Abeliovich. Source: Prevail
Dr Asa Abeliovich. Source: Prevail
It was set up in a collaborative effort with The Silverstein Foundation for Parkinson’s with GBA (Click here to read a previous SoPD post about this organisation) and OrbiMed (a healthcare-dedicated investment firm).
What does Prevail Therapeutics do?
Prevail Therapeutics is a gene therapy company focused on neurodegenerative conditions, particularly GBA-associated Parkinson’s.
What is GBA-associated Parkinson’s?
GBA is a gene that provides the instructions for making a protein called Glucocerebrosidase (or GCase). GCase is an enzyme that helps with the digestion and recycling of various proteins (particularly glucocerebrosides) inside cells.
Genetic variations in the GBA gene are associated with a condition called Gaucher disease (Click here to read a previous SoPD post on GBA and Gaucher disease).
In the 1990s, physicians began to notice that patients with Gaucher disease appeared to have a higher risk of developing Parkinson’s. An example of this was a report published in 1996 that described individuals with Gaucher disease who also exhibited an early-onset, severe form of Parkinson’s with a greater element of cognitive decline:
 Title: Occurrence of Parkinson’s syndrome in type I Gaucher disease.
Title: Occurrence of Parkinson’s syndrome in type I Gaucher disease.
Authors: Neudorfer O, Giladi N, Elstein D, Abrahamov A, Turezkite T, Aghai E, Reches A, Bembi B, Zimran A.
Journal: QJM. 1996 Sep;89(9):691-4.
PMID: 8917744 (This article is OPEN ACCESS if you would like to read it)
In this study, the Israeli researchers report on 6 people with Type I Gaucher disease (which up until that point had not been considered neuronopathic). All six of the subjects also exhibited the hallmark of a rather severe form of Parkinson’s, which made its appearance in the 4th to 6th decade of life and displayed an aggressive progression and was largely unresponsive to conventional anti-Parkinson therapy (such as L-dopa).
Larger follow up studies have reported similar results (Click here and here for an example).
In healthy unaffected individuals, the enzyme Glucocerebrosidase (or GCase) helps to break down proteins and helps to maintain proper lysosomal function (lysosomes are small bags inside of cells where the enzyme conducts its job).
 Source: Prevail
Source: Prevail
In folks with GBA genetic variations, however, the enzymatic breakdown of proteins is disrupted, which results in the build up and accumulation of proteins. This can lead to inflammation and ultimately cell death. And this process is believed to give rise to the appearance of Parkinson’s symptoms in some of these individuals (GBA variants are quite common, but not everyone with them goes on to get PD).
Numreous efforts have been made to identify drugs which may be able to correct this situation, such as the Ambroxol clinical trial program being conducted in London (Click here to read a previous SoPD post on that topic).

Ambroxol. Source: Skinflint
The issue with drug treatments, however, is that the therapy requires the patient to continuously take the drug.
And this is where a gene therapy approach – like what Prevail is proposing – may provide a more convenient solution.
What is gene therapy?
Gene therapy is an experimental treatment approach that involves treating medical conditions with DNA rather than drugs.
 Source: Baltimoresun
Source: Baltimoresun
Gene therapy basically involves introducing a new piece of DNA or replacing a faulty piece of DNA within a population of cells. DNA, as you may remember from high school science class provides the instructions for making proteins in the a cell and these proteins are the bits that actually do stuff.
By introducing a new piece of DNA into a cell, the cell can start to produce a functioning protein that it may not normally produce. In some diseases, a cell may normally produce a particular protein, but because the genetic instructions in the DNA (a section of the DNA called a gene) for that protein have a small error (a genetic mutation), a non-functioning version of the protein is actually being produced. The introduction of the new correct (functioning) version of that piece of DNA (or gene) into a cell can start the production of a functional version of the protein.
 Gene therapy. Source: yourgenome
Gene therapy. Source: yourgenome
Taking this approach one step further, we can take sections of DNA that contain the genes involved with the production of a proteins that would be beneficial for the cell, such as GDNF. By then injecting a virus with the DNA for GDNF into the brain, we can produce GDNF in any infected cells (it’s slightly more complicated than that, but you get the basic idea). And such a gene therapy approach has recently been tested in a clinical trial (Click here to read more about this).
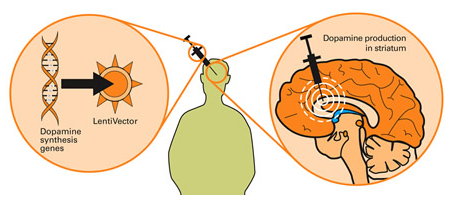
Gene therapy for Parkinson’s disease. Source: Wiki.Epfl
I’m sorry, but did you say viruses?
Yes, if you remove the viral DNA from inside a virus and replace it with something useful, then a virus becomes a very useful biological delivery system. Far superior to anything we humans have devised thus far. Viruses are easy to produce and manipulate, and they can even be engineered to target specific cell types.

Viruses – an ideal biological delivery system. Source: HuffingtonPost
And these viruses have been engineered NOT to replicate or cause disease. Their ability to replicate themselves has been completely removed.
They deliver the proposed DNA and that is all.
And there is a particular type of virus that Prevail is planning to use in their PR001 treatment.
What sort of virus?
An AAV virus.
What is an AAV virus?
Adeno-associated viruses (or AAV) are a kind of virus that are popular with researchers because A.) they readily infect human and primate cells, and B.) they produce little (if any) immune response, and C.) they are non-pathogenic (they don’t cause any known diseases).
Given these characteristics, AAVs have been used in most of the gene therapy clinical trials thus far:
 A few of the AAV-based gene therapy clinical trials. Source: Wikipedia
A few of the AAV-based gene therapy clinical trials. Source: Wikipedia
They were originally discovered in the preparation of another type of virus, called an adenovirus (hence the name ‘Adeno-associated’). They were believed to simply be a contaminant of that preparation. Further research, however, revealed that AAVs belong to the Dependoparvo genus of viruses, which in turn belongs to the family Parvoviridae.
AAVs are single-stranded DNA viruses, and they are one of the smallest viruses (approximately 22 nm in diameter) with a non-enveloped capsid. The capsid is the shell surrounding the genetic material of the virus. Viruses are either enveloped or non-enveloped. “Enveloped” means that a second casing surrounds the capsid, providing further protection for the virus, while “non-enveloped” viruses have only the capsid.

Enveloped (left) vs Non-enveloped (right) viruses. Source: Differencebtwn
Given the reduced amount of casing, non-enveloped viruses are generally more virulent (more infectious) than enveloped viruses (a good example of a non-enveloped virus is the influenza virus). Non-enveloped viruses do not survive outside of an organism for long though.

The AAV capsid. Source: Wikipedia
PR001 is an AAV9 virus.
Eleven different (or serotypes) of AAV have thus far been identified (Source). AAV9 are very effective at infecting cells in the brain (such as neurons – click here to read more about AAV viruses).
Have AAV9 viruses been used before in clinical trials?
Yes, they have.
There are at least 14 registered clinical trials using AAV9 viruses (Click here to read more about them).
In 2017, Prevail entered into an exclusive worldwide license agreement with REGENXBIO to develop and commercialise gene therapy products using REGENXBIO’s NAV AAV9 vector for the treatment of Parkinson’s and other related neurodegenerative conditions. These viruses are remarkably more robust and selective compared to previous generations.
In June 2018, the U.S. FDA granted Fast Track designation to REGENXBIO for RGX-111, which is a novel, one-time investigational treatment for Mucopolysaccharidosis Type I (MPS 1). MPS 1 is a rare lysosomal storage disease caused by deficiency of an enzyme called iduronidase (IDUA). RGX-111 is a NAV AAV9 vector that is designed to deliver the human iduronidase (IDUA) gene directly to cells in the brain (Click here to read the press release).
That trial is now ongoing (Click here to learn more about this study).
So PR001 is an AAV9 virus. But what does it contain?
PR001 will deliver a section of DNA which will provide infected cells with the instructions for making a normal version of the GCase enzyme. AAV viruses integrate their cargo DNA into the DNA of the host cell, meaning that the GCase enzyme can be made continuously over time (better than taking a drug every few hours right?). The intended route of delivery for PR001 is via a single injection into the intra cisterna magna.
What is the intra cisterna magna?
Our brains sit in a solution called cerebrospinal fluid, and there are areas of our brains where this liquid can be accessed. The intra cisterna magna is one of these areas. It is at the base of our skulls (under the cerebellum of the brain), and a needle can be inserted at the back of our heads, near the top of the neck, to access it.
The cisterna magna is labelled “cisterna cerebellomedullaris” on the lower right side of the image below.
Source: Wikipedia
Cisterna magna delivery does require surgery, but it will not be as long or as invasive as some other experimental gene therapy approaches that have been previously discussed on this website (Click here for an example).
So by injecting a virus – containing the instructions for a normal version of the GCase enzyme – into the fluid surrounding the brain (via the base of the skull), Prevail are hoping to correct GBA-associated Parkinson’s?
That is the plan.
The virus will infect a high number of cells in the brain, and those cells will start to produce normal GCase enzyme and this will hopefully restore normal lysosomal function – in theory making the cells healthier and improving their survival.
 Source: Prevail
Source: Prevail
And this outcome will ideally have a disease modifying effect on the course of the condition, by slowing down or stopping the progression.
As I say, that is the plan.
Is there any evidence to suggest that this approach might work?
I am not aware of any published data on PR001 (happy to be corrected on this). And most of the publications listed on the Prevail website are related to the clinical aspects of GBA-associated Parkinson’s (Click here to read more about this). This is not a critique – most biotech companies keep their intellectual property close to their chests.
But Prevail Therapeutics has recently become a publically traded company. And as part of that process, the company had to file papers with the U.S. Securities and Exchange Commission (Click here to read those filing).
 Source: Wikipedia
Source: Wikipedia
In those filings, the company provided a great deal of information about PR001.
We shall review some of that data here – but please note, that this is unpublished data. Being present in a Government document it is in the public domain, but it has not yet been peer-reviewed or published in a scientific journal. In addition, this post is not an endorsement of Prevail Therapeutics as an investment opportunity (please see disclaimer in footnotes) – the author just thought that the science behind this company is interesting.
Moving on.
The company used a mouse model of GBA-associated Parkinson’s which involves daily delivery of a compound called conduritol-ß-epoxide (or CBE). CBE is a pharmacological inhibitor of GCase. The mice were subsequently given a single treatment of one of 3 different doses of PR001 (denoted by the number of vector genomes, or vg):
- Low dose of 3,200,000,000 vector genomes, or 3.2 x 109 vg
- Medium dose of 10,000,000,000 vector genomes, or 1.0 x 1010 vg
- High dose of 32,000,000,000 vector genomes, or 3.2 x 1010 vg
All three doses were found to be well tolerated and resulted in the detection of vector genomes in the brain and spinal cord five weeks after delivery. GCase activity, which was reduced in CBE-treated mice, was found to be increased by the highest dose of PR001 in the brain at five weeks. The CBE-treated mice were also found to have raised levels of the toxic glycolipids, GluCer and GluSph, in the brain, which was reduced by PR001 administration in a dose-dependent manner (see image below):
 Source: SEC
Source: SEC
And PR001 was found to have positive impact on the behaviour of the mice. While mice treated with just CBE had reduced performance on the tests of motor function (rotarod and tapered beam), the highest dose of PR001 caused a statistically significant improvement in motor function (see image below):
 Source: SEC
Source: SEC
The company also looked at neuroinflammation in CBE treated mice, and found that CBE induced reactive astrogliosis (as determined by glial scarring) and microgliosis (as determined by a marker called Iba1). Remarkably, PR001 treatment significantly reduced both reactive astrogliosis and microgliosis – in a dose-dependent manner (see image below):
 Source: SEC
Source: SEC
All of the data above came from experiments that only lasted 5 weeks. In a longer-term assessment of PR001 in CBE treated mice, the researchers tested the treatment over a 6 month period and “observed persistent and durable effects” – GCase activity levels remained significantly increased in the cerebral cortex of treated mice, and there was a corresponding reduction of lipid accumulation (see image below):
 Source: SEC
Source: SEC
The researchers also investigated levels of aggregated protein in mice that have been genetically engineered to produce high levels of human alpha synuclein protein which carries the A53T genetic mutation. When these mice were treated with PR001, there was a reduction in levels of alpha synuclein aggregates (see graph below):
 Source: SEC
Source: SEC
And finally the company conducted PR001 safety and biodistribution testing in healthy non-human primates. While I appreciate some readers will object to this type of experimentation, it is a regulatory requirement before any novel therapy can be tested in humans. In addition, the company “plans to use the safety and biodistribution results from these studies to inform dose selection in our clinical trials“.
The animals were treated with either:
- Control solution
- PR001 low dose (2.1 x 1010 vg/g brain, or vector genomes per gram of brain mass)
- PR001 high dose (8.0 x 1010 vg/g brain)
Six months after PR001 administration, levels of GCase in the brain were found to be significantly raised (see graph below).
 Source: SEC
Source: SEC
The effect in the graph may not look extremely impressive (a 10-20% increase), but remember these animals are normal – they do not have any lysosomal dysfunction or any GCase deficit. In addition, this data suggests that increasing GCase levels does not have a deterimental effect over a 6 month period.
There is a lot more data in the SEC filing for readers who are interested to learn more (Click here to read more about it).
So when does clinical testing start?
On the 4th June, 2019, Prevail Therapeutics announced that the U.S. Food and Drug Administration (FDA) has accepted the company’s Investigational New Drug (IND) application for PR001.
This news gives the company a green light to “initiate a Phase I/II clinical trial that will investigate the safety and tolerability of PR001, and will also measure key biomarkers and exploratory efficacy endpoints, in patients with PD-GBA. The Company plans to begin dosing patients in the trial this year” (Source).
We currently do not know anything more about a planned clinical trial beyond this, but we can assume that it will be primarily focused on individuals with GBA-associated Parkinson’s.
Interesting. What else are Prevail doing?
On their website, the company has this table outlining their pipeline of projects:
 Source: Prevail
Source: Prevail
The thing that catches one’s eye is PR004, which is described as utilising “an AAV9 vector to deliver the GBA1 gene, which encodes glucocerebrosidase (GCase), and a molecule that suppresses expression of α-Synuclein”. I will be interested in learning more about this in the near future.
So what does it all mean?
A relatively young biotech company is making significant progress towards testing a novel gene therapy approach for a subtype of Parkinson’s. The limited information that we currently have has been gleamed from SEC filings, but looks very interesting.
If nothing else the urgency with which this effort has got to this point is extremely impressive (you might remember that we were hoping to hear some news regarding Prevail this year in our 2019 outlook – click here to read that post).
The information provided in this post blows away any expectations this author might have had.
 But as always with new experimental therapies, we must be guarded in how we manage expectations. The philosophy promoted here at the SoPD is to have zero expectations, and in this manner we will never be overly disappointed.
But as always with new experimental therapies, we must be guarded in how we manage expectations. The philosophy promoted here at the SoPD is to have zero expectations, and in this manner we will never be overly disappointed.
Easy to write, but hard to practise, of course.
And I have to admit to being a little bit excited by the pending Prevail Therapeutics clinical trial.
EDITOR’S NOTE: The information provided by the SoPD website is for information and educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
In addition, some of the companies mentioned in this post are publicly traded companies. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. None of the companies mentioned have requested that this material be produced, nor has the author had any contact with the companies. This post has been produced for educational purposes only.
The banner for today’s post was sourced from Prevail.



8 thoughts on “Dream, struggle, create, Prevail”