|
The cryptic title of this post will hopefully make sense by the time you have finished reading the material present here. This week, new research from the USA points towards an increased risk of Parkinson’s (PD) for people that suffer from inflammatory bowel disease (IBD). That same research, however, also points towards a clinically available treatment that appears to reduce the risk of Parkinson’s in individuals affected by inflammatory bowel disease. That treatment being: anti–tumor necrosis factor antibodies (TNF AB). Is that title making sense yet? If not, read on. In today’s post, we will outline what inflammatory bowel disease is, review what the new research found, and discuss what is known about TNF in Parkinson’s. |
Inflammatory bowel disease. Source: Symprove
Inflammatory bowel disease (or IBD) is one of these umbrella terms that is used to refer to a group of inflammatory conditions of the large and small intestine:
The large and small intestine. Source: Adam
The symptoms of IBD can include abdominal pain, diarrhoea, vomiting, rectal bleeding, severe internal cramps/muscle spasms in the region of the pelvis, and weight loss.
The most common forms of IBD are Crohn’s disease and ulcerative colitis.
There has been an increased incidence of IBD since World War II, which could be associated with increased awareness and reporting of the condition, but it could also be linked with increases in meat consumption (Click here to read more about this). For example, in 2015, an estimated 1.3% of U.S. adults (3 million) were diagnosed with IBD, which was a large increase on the levels in 1999 (0.9% or 2 million adults – Source: CDC).
This is delightful, but what does it have to do with Parkinson’s?
So this week, an interesting study was published on the Journal of the American Medical Association – Neurology edition website:
Title: Anti–Tumor Necrosis Factor Therapy and Incidence of Parkinson Disease Among Patients With Inflammatory Bowel Disease
Authors: Peter I, Dubinsky M, Bressman S, Park A, Lu C, Chen N, Wang A
Journal: JAMA Neurol. Published online April 23, 2018
PMID: 29710331
In this study, the researchers collected longitudinal data – from January 1, 2000 to March 31, 2016 – from more than 170 million people in two large administrative claims databases: the Truven Health MarketScan Commercial Database and the Medicare Supplemental Database. The investigators were looking for individuals who:
- had two IBD diagnoses
- were at least 18 years old
- had been enrolled for more than 12 months before the IBD diagnosis
- had at least 6 months of follow-up
- no prior diagnosis of Parkinson’s
The researchers found that 657 637 individuals received at least 2 IBD diagnoses, but of those people only 144 018 fit the criteria described above.
The researchers then compared the records of those 144 018 individuals with IBD with age- and sex-matched records from 720 090 unaffected controls.
The results were interesting:
- Curiously, individuals with IBD in this study were more likely to reside in the Northeastern regions of the US.
- More importantly, the incidence of Parkinson’s among people with IBD was 28% higher than that among unaffected matched controls.
- The increased rate of Parkinson’s was the same among the different types of IBD.
- A greater proportion of men with IBD developed Parkinson’s (0.34% vs 0.25%). There was very little difference between women (0.19% vs 0.16%).
This result is very similar to a previous population study conducted in Taiwan that was looking for any relationship between IBD and Parkinson’s:
Title: Association Between Parkinson’s Disease and Inflammatory Bowel Disease: a Nationwide Taiwanese Retrospective Cohort Study.
Authors: Lin JC, Lin CS, Hsu CW, Lin CL, Kao CH.
Journal: Inflamm Bowel Dis. 2016 May;22(5):1049-55.
PMID: 26919462
In this study, the researchers used the Taiwanese National Health Insurance Research Database, which covered the 23.22 million residents of Taiwan (aged 20 years or older). They compared the 8373 people diagnosed with IBD during 2000 to 2011 with IBD-free individuals. The records of 41,865 people were used in this retrospective study: 8373 people with IBD and 33,492 age- & sex-matched controls without IBD. The investigators found that the risk of developing Parkinson’s was 35% higher in the IBD group than in the non-IBD control group – a very similar number to the new American study.
But the American study took the analysis one step further, and this is where the research gets REALLY interesting.
What did they do?
So in the American study, of the 144 018 people with IBD that fit the criteria to be included in the analysis, 13 089 of them were exposed to anti-TNF therapy after being diagnosed with IBD.
What is anti-TNF therapy?
Tumor necrosis factor (or TNF) is a small cytokine that is involved in inflammation.
What is a cytokine?
Cytokines (from the Greek: kýtos meaning ‘container, body, cell’; and kī́nēsis meaning ‘movement’) are small proteins that are secreted by certain cells in the body and they have an effect on other cells. Cytokines are a method of communication for cells.
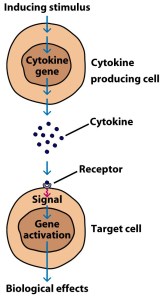
How cytokines work. Source: SBS
In the case of Tumor necrosis factor (or TNF) the signal that it passes on to the cells it attaches is one of inflammation.
What is inflammation?
Inflammation is an immune response to a situation in the body. The function of inflammation is to eliminate the initial cause of the problem (such as a damage causing pathogen like a virus), clear out dying cells and tissues damaged from the original insult, clear out dying cells and tissues damaged by the inflammatory process itself, and initiate tissue repair.
TNF is produced by blood cells (eg. macrophages and monocytes) during early inflammatory events. It triggers the production and release of a series of other inflammatory molecules/cytokines – further inflaming the situation.
So ‘anti-TNF therapy’ is a treatment against inflammation?
Exactly.
Anti-TNF therapy involves the use if artificially produced antibodies. And before you ask: Antibodies are a critical part of our immune system. They are Y-shaped proteins which act as ‘alert flags’ for the immune system.

Monoclonal antibodies. Source: Astrazeneca
When a pathogen (an agent that causes disease or damage) is detected in your body, the immune system will quickly determine that it is not ‘self’ (meaning that it is not part of ‘you’ yourself). This judgement will be made by the identification of antigens on the surface of the pathogen.
An antigen is defined as any substance or molecule that is capable of causing an immune response in an organism. If a molecule on the surface of the pathogen is not familiar to the immune system, it will be considered an antigen and an immune response will be initiated.
Antibodies bind to parts of the antigen and can do a pretty good job of stopping pathogens, by blocking them from attaching to cells or by sticking together and clustering the antigens to prevent them from doing anything bad. Now, a while back some clever researchers engineered artificial antibodies that could bind to TNF, and this stopped TNF from doing its job (causing inflammation), which resulted (‘surprise, surprise’) in less inflammation.
Anti-TNF therapy has become one of the most common treatments for IBD (as well as other inflammatory conditions like rheumatoid arthritis, etc). And Adalimumab (which is sold under the trade name Humira) is one of the most commonly used anti-TNF therapies for IDB.
Humira. Source: Humirapro
I see. So what did the researchers notice about the people with IBD exposed to anti-TNF therapy?
When the researchers looked at the 13 089 of the 144 018 people with IBD that were exposed to anti-TNF therapy after being diagnosed with IBD, they found that those individuals had a markedly lower incidence of Parkinson’s (0.08 per 1000 patient-years) than people with IBD who were not exposed to anti-TNF therapy (0.76 per 1000 patient-years).
That is to say, the investigators found a 78% reduction in the Parkinson’s incidence rate in the group of IBD sufferers who were exposed to anti-TNF therapy (compared to the IBD group not exposed).
Go back and read that sentence again please.
13 089 people with IBD were exposed to anti-TNF therapy according to this study, and there were only two cases that wen on to develop Parkinson’s.
Compare that to the 369 cases of Parkinson’s in the 130 929 in people with IBD that were NOT exposed to anti-TNF therapy.
Stop here and take a moment to fully appreciate this result.
Take as long as you want…
…
Ok, moving on:
While the result is exciting and very interesting, we should be cautious in not over-reading too much out of the results.
Admittedly the numbers involved in the study are quite small (only 371 cases of PD). Plus (as pointed out in the editorial connected to this study), given that more cases of IBD were reported to come from the North East region of the United States, there could be some influence from genetic variants that are associated with both PD and IBD (in particular, LRRK2 genetic variants within the Ashkenazi Jewish community on the East coast of the US):
Title: Functional variants in the LRRK2 gene confer shared effects on risk for Crohn’s disease andParkinson’s disease.
Authors: Hui KY, Fernandez-Hernandez H, Hu J, Schaffner A, Pankratz N, Hsu NY, Chuang LS, Carmi S, Villaverde N, Li X, Rivas M, Levine AP, Bao X, Labrias PR, Haritunians T, Ruane D, Gettler K, Chen E, Li D, Schiff ER, Pontikos N, Barzilai N, Brant SR, Bressman S, Cheifetz AS, Clark LN, Daly MJ, Desnick RJ, Duerr RH, Katz S, Lencz T, Myers RH, Ostrer H, Ozelius L, Payami H, Peter Y, Rioux JD, Segal AW, Scott WK, Silverberg MS, Vance JM, Ubarretxena-Belandia I, Foroud T, Atzmon G, Pe’er I, Ioannou Y, McGovern DPB, Yue Z, Schadt EE, Cho JH, Peter I.
Journal: Sci Transl Med. 2018 Jan 10;10(423).
PMID: 29321258
In this study from earlier this year, the DNA of 2066 people with Crohn’s disease and of Ashkenazi Jewish background CD cases was compared with the DNA of 3633 healthy controls. The researchers found genetic variants in the Parkinson’s associated LRRK2 gene that increased risk for Crohn’s disease (the N2081D variant), as well as LRRK2 genetic variants that provided protection from Crohn’s disease (the N551K variant – see figure below).
Both of these variants are also associated with Parkinson’s.
Genetic variants in the LRRK2 gene, associated with PD and Crohn’s. Source: Nature
These genetic variations are very rare though, and it is unlikely that they can account for all of the effect observed in the TNF study.
There are additional limitations with the TNF study, however, such as the researchers who conducted the study did not have access to any lifestyle information, which have previously been found to impact PD and IBD in different ways. Smoking, for example, reduces the risk of developing Parkinson’s (Click here to read more about that), and it also reduces the risk and severity of ulcerative colitis. But conversely, smoking increases the risk and severity of Crohn’s disease (Click here to read more).
Thus, while the result is very interesting, it is too early to draw too many conclusion and go rushing to one’s doctor and asking for “some of that anti-TNF therapy please”.
Do we know anything about TNF in Parkinson’s?
Yes. There has been quite a lot of research on TNF in Parkinson’s.
Level of TNF are raised in the blood and cerebrospinal fluid (the liquid surrounding the brain) of people with Parkinson’s (when compared to healthy controls – Click here, here and here to read more about this).
In addition, there are associations between genetic variants in the TNF gene and Parkinson’s:

Title: Tumor necrosis factor gene polymorphisms in patients with sporadic Parkinson’s disease
Authors: Nishimura M, Mizuta I, Mizuta E, Yamasaki S, Ohta M, Kaji R, Kuno S.
Journal: Neurosci Lett. 2001 Sep 21;311(1):1-4.
PMID: 11585553
In this study, the researchers found a genetic variation (or a polymorphism) in the region of DNA that drives the activity of the TNF gene (this region is called the promoter). This variant results in higher than normal levels of TNF production. And it was found in a cohort of Japanese people with early-onset Parkinson’s (compared to late-onset PD patients and healthy controls). And that same variant was found in another independent cohort (Click here to read more about that).
Other genetic variants in the TNF gene have also been reported (Click here and here to learn more). But it should be noted here that these particular genetic variants are all extremely rare.
In addition, there is evidence that the introduction of TNF into the regions of the brain affected by Parkinson’s can cause cell loss:

Title: Intra-parenchymal injection of tumor necrosis factor-alpha and interleukin 1-beta produces dopamine neuron loss in the rat.
Authors: Carvey PM, Chen EY, Lipton JW, Tong CW, Chang QA, Ling ZD.
Journal: J Neural Transm (Vienna). 2005 May;112(5):601-12.
PMID: 15583962
In this study, the researchers injected TNF into the brains of rats. In particular, they were targeting the branches of the dopamine neurons. They found that the introduction of this pro-inflammatory cytokine could cause the loss of dopamine neurons by itself. The observed cell loss was dose-dependent, meaning the higher the dose, the more the cell loss.
So there is some evidence supporting the association between TNF and Parkinson’s. And for those interested, there is a good section on TNF and Parkinson’s in this review article.
Has anyone ever looked at blocking TNF in models of Parkinson’s?
Yes, they have.
But this research has been hampered by problems with actually getting anti-TNF antibodies and TNF inhibitors into the brain. You see, they are not very good at crossing the blood-brain barrier – the protective membrane surrounding the brain (Click here to read more about the issue of TNF treatments accessing the brain).
This setback, however, has not stopped the development of small molecule TNF inhibitors.
One example of this is a molecule called XPro1595:
Title: Blocking soluble tumor necrosis factor signaling with dominant-negative tumor necrosis factor inhibitor attenuates loss of dopaminergic neurons in models of Parkinson’s disease.
Authors: McCoy MK, Martinez TN, Ruhn KA, Szymkowski DE, Smith CG, Botterman BR, Tansey KE, Tansey MG.
Journal: J Neurosci. 2006 Sep 13;26(37):9365-75.
PMID: 16971520 (This article is OPEN ACCESS if you would like to read it)
XPro1595 was a TNF inhibitor that was engineered to disrupt TNF by binding to it and inhibiting its signalling. The researchers in this study, tested this new inhibitor by injecting it directly into the brain of a rodent model of Parkinson’s and they observed reduced the dopamine cell loss.
And this report has been followed by additional research that demonstrated that XPro1595 could be injected peripherally and was able to cross the blood brain barrier to rescue a model of Parkinson’s (Click here to read that research report).
So what does it all mean?
New research from the US suggests that people with inflammatory bowel disease have a higher risk of developing Parkinson’s. It is important to understand, however, that the chances of developing Parkinson’s is still very low for people with IBD. In addition, the new research also suggests that by treating the IBD with anti-TNF therapy, the risk of developing Parkinson’s can be reduced.
Another interesting aspect of the study: it is hard not to notice that four of the seven scientists named as authors of this new research report all work for a Pharmaceutical company called Abbvie.
Source: Abbvie
Foundered in 2013, from the break up of Abbott Laboratories , Chicago-based Abbvie has a very big interest in anti-inflammatory drugs, as they own some of the leading brands. Most prominent among them is Adalimumab (which I mentioned above). It is used to treat rheumatoid arthritis, Crohn’s disease, ulcerative colitis, and psoriasis (another inflammatory condition associated with Parkinson’s – Click here to read more about that).
Abbvie is a company that has active research interests in Parkinson’s, with their product Duodopa approved for use in the clinic and a follow up product ABBV-951 being clinically tested (Phase I). So it will be interesting and very encouraging to see if – based on this and future research – Abbvie expand their Parkinson’s-related research activities as a natural extension of their broader anti-inflammatory expertise. This would be particularly advantageous as we head towards future attempts to treat early or prodromal Parkinson’s.
We will be watching this with interest.
ADDENDUM: 28th April 2018
This addendum has been made to highlight that similar anti-TNF therapy findings have been made in another neurodegenerative condition: Alzheimer’s.
In 2016, this research report was published:

Title: Treatment for Rheumatoid Arthritis and Risk of Alzheimer’s Disease: A Nested Case-Control Analysis.
Authors: Chou RC, Kane M, Ghimire S, Gautam S, Gui J.
Journal: CNS Drugs. 2016 Nov;30(11):1111-1120.
PMID: 27470609 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers performed an analysis of more than 8.5 million commercially insured adults (aged ≥18 years) in the US states (and associated territories) in the Verisk Health claims database. They were seeking to determine the risk of developing Alzheimer’s among people with and without rheumatoid arthritis – a long-term inflammatory condition that causes pain, swelling and stiffness in the joints.
They found that Alzheimer’s was significantly more common among people with rheumatoid arthritis (0.79 %) than among those without (0.11 %). And when they looked at anti-TNF therapy… guess what they found?
Exposure to anti-TNF therapy (but not other immunosuppressive drugs) was associated with significantly lowered risk of Alzheimer’s among people with rheumatoid arthritis (0.45%).
And when this study looked more closely at the anti-TNF therapies, it found that of the three anti-TNF agents investigated, only Etanercept (trade name: Enbrel) was actually associated with a decreased risk of Alzheimer’s among people with rheumatoid arthritis.
Etanercept is different to many of the other anti-TNF therapies (which are antibodies), in that it is a fusion protein. Etanercept is half antibody/half receptor. It functions as a ‘decoy receptor’ that binds to TNF floating around in the body. Why it is apparently more efficient at reducing the risk of Alzheimer’s is not entirely clear.
Different types of anti-TNF therapies. Source: Researchgate
And interestingly, this result has (sort of) been independently replicated:
Title: The influence of TNF inhibitors on dementia incidence in patients with rheumatoid arthritis; an analysis from the BSRBR-RA.
Authors: McGuinness B, Holmes C, Mirakhur A, Kearsley-Fleet L, Vieira R, Watson K; BSRBR-RA Contributors Group; BSRBR-RA Control Centre Consortium, Hyrich K.
Journal: Int J Geriatr Psychiatry. 2018 Mar;33(3):556-558.
PMID: 29424111
Using the the British Society for Rheumatology Biologics Register for Rheumatoid Arthritis (BSRBR‐RA) and a database of people receiving conventional (non-antibody) anti‐rheumatic drugs (such as methotrexate, sulfasalazine, leflunomide, hydroxychloroquine, etc), the researchers conducting this study wanted to determine if the results from the previous study could be replicated in an independent analysis – is someone with rheumatoid arthritis at higher risk of Alzheimer’s, and can Etanercept reduce that risk?
In their analysis, the investigators found 13 474 individuals who were treated with Etanercept and 3774 individuals who were treated with conventional treatments. They also found a total of 46 cases of dementia.
There were 24 cases of dementia in the Etanercept-treated group, and 22 cases in the conventional treatment group.
Can you do the maths?
24 cases out of 13 474 individuals who were treated with Etanercept VS 22 cases out of 3774 individuals who were treated with conventional treatments.
That is 0.18% compared to 0.58%, respectively. While this difference was not significant, the trend is certainly supporting the idea that Etanercept treatment in cases of rheumatoid arthritis reduces the risk of Alzheimer’s.
And curiously, before any of this rheumatoid arthritis was published there was a series of clinical studies conducted that looked at etanercept in people with Alzheimer’s. Initially there were a couple of small pilot studies, which indicated that the drug was safe if injected directly into the cerebrospinal fluid – the liquid surrounding the brain (Click here and here to read more about those studies). And those studies were followed up by a Phase II clinical trial:

Title: Etanercept in Alzheimer disease: A randomized, placebo-controlled, double-blind, phase 2 trial.
Authors: Butchart J, Brook L, Hopkins V, Teeling J, Püntener U, Culliford D, Sharples R, Sharif S, McFarlane B, Raybould R, Thomas R, Passmore P, Perry VH, Holmes C.
Journal: Neurology. 2015 May 26;84(21):2161-8.
PMID: 25934853 (This article is OPEN ACCESS if you would like to read it)
This study differed from the initial pilot studies, however, in that rather than delivering etanercept into the cerebrospinal fluid, in this trial the researchers gave the drug peripherally (they simply injected it subcutaneously). The study was a randomised, double-blind, placebo controlled trial of etanercept in people with mild to moderate Alzheimer’s (Click here to read more about this trial).
The results of the study were inconclusive (“While there were some interesting trends that favored etanercept, there were no statistically significant changes in cognition, behavior, or global function”), which the researchers put down to the small size of the study – only 41 participants were involved in the study – recommending that a larger trial was required.
For a short, open-access review article on the topic of anti-TNF therapy in Alzheirmer’s (pre-clinical and clinical research) – Click here.
Now the obvious question that you will be asking is “Has Etanercept ever been tested in the context of Parkinson’s?”
And the answer is ‘Yes’.
But before you get too excited, please read on:
While there has been interesting results involving etanercept in cell culture models of Parkinson’s (Click here, here and here for examples), there has been only one in vivo study (that I am aware of), and the results were not so promising:

Title: Neuroprotection with a brain-penetrating biologic tumor necrosis factor inhibitor.
Authors: Zhou QH, Sumbria R, Hui EK, Lu JZ, Boado RJ, Pardridge WM.
Journal: J Pharmacol Exp Ther. 2011 Nov;339(2):618-23.
PMID: 21831964 (This article is OPEN ACCESS if you would like to read it)
In this study, the researchers (from the University of California and ArmaGen Technologies) used etanercept as a positive control in comparison to their own drug, a TNF inhibitor fusion protein called cTfRMAb-TNFR.
The key difference between the two drugs: Etanercept does not cross the blood brain barrier (the protective membrane covering the brain), while cTfRMAb-TNFR does cross the barrier.
And this difference in properties demonstrated itself in a mouse model of Parkinson’s (6-OHDA): Etanercept was found to have no protective effect on cell survival or motor behaviour.
This is just one study though. As with the IBD/PD report discussed in the post above, we should not draw too many conclusions based on just one result. It would be interesting to see further research on etanercept in other models of Parkinson’s, and perhaps using etanercept in conjunction with other potential treatments.
Importantly, the results of the Alzheimer’s research points towards anti-TNF therapy having a beneficial impact on another neurodegenerative condition, raising the idea that targeting inflammation may be a useful treatment approach that could be use at the same time as administering therapies which target actual disease mechanisms.
Hopefully all of this research will stimulate further research in this area.
The banner for today’s post was sourced from Eternalplants













Simon – thanks for the informative report. A couple of thoughts:
The IBD association may play into the theory that (some?) PD has an auto-immune etiology. Certainly there is a large focus on triggered auto-immunity being at (or near) the root cause of IBD and CD – with various hypotheses regarding the trigger(s). In this area, I think we’ll see increasing focus on microbiome (sounding familiar ?), and possibly gluten.
Your article highlights an observed significant increase in incidence of IBD from 1999 to 2015. It could be interesting to see if this correlates with increasing incidence of self-reported gluten sensitivity – why? Because, as Sarah Ballentyne and others have made a strong argument that sub-chronic gluten sensitivity can, in and of itself, lead to potentiation/activation of auto-immune response in the wall of the gut (where does PD start?), which could cascade into a whole slew of auto-immune or inflammation-linked conditions – of which IBD and CD are certainly the most causally linked and obvious, but where PD is also one.
LikeLike
Hi Alex,
Thanks for your comment – glad you found the post interesting.
I am not familiar with any association between guten and Parkinson’s (happy to be corrected on this). Interesting food for thought though (no pun intended). There are some genetic variants that appeared to be shared between PD and some autoimmune conditions (https://www.ncbi.nlm.nih.gov/pubmed/28586827), but these would not explain all cases. I also mentioned in the post that there is believed to be an increase in IBD based on increased meat consumption, and I mentioned in a previous post that meat is considered to be a negative type of food for people with PD (https://scienceofparkinsons.com/2017/09/18/food/).
I agree that there will probably be an increase in the microbiome research, but I’m not sure that this will explain the core problems of PD. Given that similar anti-TNF therapy results are seen in Alzheimer’s (see the addendum), I suspect that things like the microbiome and IBD are simply influential components of the condition rather than the actual source of the problem. Just that is just my gut feeling (again, no pun intended).
Kind regards,
Simon
LikeLike
Another thought –
The Peter et al. 2018 longitudinal study observed a 10-fold reduction of incidence of PD in IBD patients treated with “conventional” anti-TNF therapy – which you report as unable to cross the blood-brain barrier. IF there is a causal link, then it would be by a systemic reduction in TNF. Why wait to design new BBB-compatible molecules? Should we not instead run a clinical trial using “conventional” re-purposed anti-TNF (= FDA fast-track) to see if it supports the longitudinal study data ?
LikeLike
Alex Leonard wrote: “to see if it supports the longitudinal study data”
Or perhaps more directly put: “to see if the PwPs taking the anti-TNF drug experience significant symptomatic relief (easier to measure), and/or significant slowing of disease progression (harder to measure)”?
LikeLike
Hi Jeffreyn,
Thanks for the comment. I would probably play it safe in designing a clinical trial to look at anti-TNF therapy for PD and make ‘safety’ the primary end point (even easier to measure), with some efficacy/symptomatic measures being secondary end points. It would be easier to justify and have a higher chance of being funded/successful.
Kind regards,
Simon
LikeLike
You are right, of course, Simon (think “nilotinib”). And the chances of even that kind of Phase 1 trial actually happening are pretty low, unfortunately.
LikeLike
Hi Alex,
I am inclined to agree with that line of thinking. Why not test if lowering peripheral inflammation levels can help? But I would probably be inclined to make this part of a multi-treatment approach (anti-TNF therapy in conjunction with another disease targeting treatment), maybe as part of a multi-arm drug trial (one group on anti-TNF therapy, being compared to another group on anti-TNF therapy plus drug X).
Kind regards,
Simon
LikeLike
Hi Simon, while anti-TNF does not cross the BBB, TNF does. Could there be an equilibrium effect, so a reduction in circulating TNF draws more TNF out of the brain? Any follow up studies may want to consider looking at TNF levels in cerebrospinal fluid.
LikeLike
Hi Kevin,
That is a really interesting idea: comparing TNF levels in both bloods and CSF if testing an anti-TNF therapy in PD. It would be interesting to see over time if both levels drop despite the fact that the inhibitor may not be able to cross the blood brain barrier. My understanding is that most TNF when generated is usually local to the site of trouble, but this could be different in chronic, long term situations. As I have mentioned in the addendum, this could be the situation in the Alzheimer’s data. And perhaps – given the raised TNF levels observed in the bloods/CSF of PwPs mentioned above – it could be interesting to test an anti-TNF therapy in conjunction with other treatments that target disease-mechanism.
Thanks for the interesting thought.
Kind regards,
Simon
LikeLike
Also, I was just doing some idle Googling and came across this small pilot, open label study in Rasmussen’s encephalitis showing effects of adalimumab. Something must be happening inside the brain, despite the inability to cross the BBB. https://onlinelibrary.wiley.com/doi/full/10.1111/epi.13387
LikeLike
Hi Kevin,
There is a lot of evidence suggesting that there is an increase in blood-brain barrier permeability with ageing, inflammation, or disease (for a review on the topic: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4543304/), so I would not be surprised if some of these anti-TNF therapies are finding their way into the central nervous system in some of these conditions. This could be one possible explanation.
Thanks for the comments.
Kind regards,
Simon
LikeLike
Ibuprofen has anti-TNF properties, good epidemiological evidence, and is available now very cheaply. Why the delay?
Time has a cost. This should be factored into clinical practice. All clinical research has a probabilistic nature. I think it is reasonable to ask, on the basis of current knowledge, what is the probability that ibuprofen has benefits for PwP and to compare this to the risks.
John
LikeLike
Although I’ve never used it, I’m sure I’ve seen ibuprofen on sale in my local supermarket.
Given the low probability of any formal trial getting up, maybe this is a valid candidate for a platform such as CliniCrowd?
LikeLike
Hi Jeffreyn,
Thanks for your comment. Interesting idea, but while ibuprofen is cheap and readily available, I for one would be reluctant to see it used in an uncontrolled situation like a Clinicrowd platform. I really like the Clinicrowd project (https://clinicrowd.online/), but there are certain drugs that I don’t think should be tested without clinicians keeping a close eye on things. Ibuprofen is one of those drugs. It would be very easy for people to start using too much of it, without any kind of read-out of effectiveness.
Kind regards,
Simon
LikeLike
Hi Simon,
Thanks very much for your reply (and your reply to John Turner). You’ve convinced me.
Actually, I’ve moved on from that (ibuprofen/CliniCrowd) idea. I’m now looking into the (possible) anti-inflammatory benefits for PwPs of (certain) dietary supplements, after reading several recent articles, such as the one linked below.
https://womensbrainhealth.org/wishful-thinking/curcumins-cognitive-benefits-look-convincing
I think this might provide a better candidate for a platform such as CliniCrowd.
Warm Regards,
Jeff
LikeLike
Hi John,
Thanks for your question. It is a very fair question and I will be addressing it in an upcoming post looking specifically at ibuprofen. But until then there is one warning: Over use of nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen can cause ulcers, bleeding, or even holes in the lining of the intestine. And this can be dangerous (and even fatal), thus I am hesitant to encourage too many people to thinking about using them. Probably best to discuss this matter with ones doctor. Having said that, it would be interesting to see a carefully controlled clinical study of ibuprofen for Parkinson’s.
Kind regards,
Simon
LikeLike
I have been researching about the TNF levels for Fibromyalgia, right now there is a study in Boston Massachusetts General hospital involving BCG Tokyo Vaccine to change the way the cytokines and other types of white blood cells react to inflammation and I came a crossed this page because I added Parkinson’s in my search since my dad has a onset of the Disease. I am now thinking could the study for fibromyalgia be linked in helping people with Parkinson’s Disease. So far it has helped people with Diabetes 1 patients. Some how I believe the immune system can get damaged by food poisoning or viruses, I remember back in the 80’s we all got sick from food poisoning (bad pork roast) and I have been developing signs of Fibromyalgia, My mother died from colon cancer 20 years ago. My Dad has been developing Parkinson’s Disease. My older sister had breast cancer about 10 years ago. My younger sister had a 19mm polyp removed from her colon 2 years ago. I just wonder if it is all related in the immune system TNF levels and that maybe the BCG study’s are going in the right direction for the cure for many Disease related to inflammation. Thank you for reading.
LikeLike