|
# # # # LRRK2 inhibition represents one of several biological approaches to slowing the progression of Parkinson’s that is currently being clinically tested. Leading the charge in the development of LRRK2 inhibitors is a biotech company called Denali Therapeutics (in partnership with Biogen). Recently, the company provided news on the immediate future clinical development plans for their lead molecule BIIB122. In today’s post, we will look at what is going to happen next for LRRK2 inhibition. # # # # |
 Source: Denali
Source: Denali
Founded in 2013 by a group of former Genentech executives, San Francisco-based Denali Therapeutics is a biotech company which is focused on developing novel therapies for people suffering from neurodegenerative diseases.
In particular, they have been leading the charge on a new class of drugs for Parkinson’s called LRRK2 inhibitors.
What are LRRK2 inhibitors?
Leucine-rich repeat kinase 2 (or LRRK2 – pronounced ‘lark 2’) – also known as ‘Dardarin‘ (from the Basque word “dardara” which means “trembling”) – is an enzyme that has many functions within a cell – from supporting efforts to move things around inside the cell to helping to keep the power on (involved with mitochondrial function).
 The many jobs of LRRK2. Source: Researchgate
The many jobs of LRRK2. Source: Researchgate
The LRRK2 gene – the section of DNA that provides the instructions for making LRRK2 protein – is made up of many different regions. Each of those regions is involved with the different functions of the eventual protein. As you can see in the image below, the regions of the LRRK2 gene have a variety of different functions:
 The regions and associated functions of the LRRK2 gene. Source: Intechopen
The regions and associated functions of the LRRK2 gene. Source: Intechopen
Tiny genetic errors or variations within the LRRK2 gene are recognised as being some of the most common genetic risk factor for Parkinson’s, with regards to increasing ones chances of developing the condition (LRRK2 variants are present in approximately 1-2% of all cases of Parkinson’s).
 The structure of Lrrk2 and where various mutations lie. Source: Intech
The structure of Lrrk2 and where various mutations lie. Source: Intech
As the image above suggests, mutations in the PARK8 gene are also associated with Crohn’s disease (Click here and here for more on this) – though that mutation is in a different location to those associated with Parkinson’s. And one particularly common Parkinson’s-associated LRRK2 mutation – called G2019S – is also associated with increased risk of certain types of cancer, especially for hormone-related cancer and breast cancer in women – Click here to read more about this. If you have a G2019S mutation, no reason to panic – but it is good to be aware of this association and have regular check ups.
The G2019S variation (the name designates its location on the gene) is the most common LRRK2 mutations. In certain populations of people it can be found in 40% of people with Parkinson’s (Click here to read more about this).
What is the effect of having the G2019S variation?
If you look at the image above, you will see that this genetic variation sits within the kinase region of the LRRK2 gene.
What does the kinase region do?
A kinase is an enzyme that regulates the biological activity of other proteins. So LRRK2 can regulate the activity of other proteins.
Kinases function by transferring phosphate groups from high-energy, phosphate-donating molecules (like ATP) to specific target proteins – in a process called phosphorylation.
 Source: Bmglabtech
Source: Bmglabtech
Wait. What does any of that mean? What does phos…phory…late mean?
Phosphorylation of a protein is basically the process of turning it on or off – making it useful or inactivating it. From allowing a protein to fold in a particular manner to actually activating/deactivating the function of a protein, phosphorylation is a critical function in cellular biology.
 Phosphorylation of a kinase protein. Source: Nature
Phosphorylation of a kinase protein. Source: Nature
Phosphorylation occurs via the addition or removal of phosphates. Their addition or removal determines the state of the protein being phosphorylated.
So the kinase region of LRRK2 is important for turning on or turning off other proteins?
In a nut shell, yes.
And am I correct if I assume that the G2019S mutation stops this kinase activity?
No, that would be incorrect.
Rather, quite the opposite.
In the mid 2000s, researchers reported that the G2019S mutation actually increases the kinase activity of LRRK2:
 Title: Parkinson’s disease-associated mutations in leucine-rich repeat kinase 2 augment kinase activity.
Title: Parkinson’s disease-associated mutations in leucine-rich repeat kinase 2 augment kinase activity.
Authors: West AB, Moore DJ, Biskup S, Bugayenko A, Smith WW, Ross CA, Dawson VL, Dawson TM.
Journal: Proc Natl Acad Sci U S A. 2005 Nov 15;102(46):16842-7.
PMID: 16269541 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers discovered that the G2019S variation did not have any obvious effect on LRRK2 protein levels or localization within cells. But it did cause an increase in the phosphorylation and the autophosphorylation activity of LRRK2.
Autophosphorylation?
LRRK2 can phosphorylate itself. It can regulate its own activity.
Ok. Got it.
This finding led the investigators to conclude that the G2019S variation may result in a ‘gain-of-function’ mechanism that could be influential in the pathology of LRRK2-associated Parkinson’s.
Does this increase in LRRK2 activity occur with any other genetic risk factors for Parkinson’s?
Yes.
Increased LRRK2 kinase activity has also been reported to be induced by variations in another Parkinson’s-associated genetic risk factor: VPS35
 Title: The Parkinson’s disease VPS35[D620N] mutation enhances LRRK2-mediated Rab protein phosphorylation in mouse and human.
Title: The Parkinson’s disease VPS35[D620N] mutation enhances LRRK2-mediated Rab protein phosphorylation in mouse and human.
Authors: Mir R, Tonelli F, Lis P, Macartney T, Polinski NK, Martinez TN, Chou MY, Howden AJM, König T, Hotzy C, Milenkovic I, Brücke T, Zimprich A, Sammler E, Alessi DR.
Journal: Biochem J. 2018 Jun 6;475(11):1861-1883.
PMID: 29743203 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers found robustly elevated the phosphorylation of LRRK2 target proteins (Rab8A, Rab10, and Rab12 – more on Rabs below) in mouse and human cells with the Parkinson’s-associated VPS35 variant grown in culture.
And VPS35 is not the only PD-associated variant that increases LRRK2 activity:
 Title: LRRK2 Expression Is Deregulated in Fibroblasts and Neurons from Parkinson Patients with Mutations in PINK1
Title: LRRK2 Expression Is Deregulated in Fibroblasts and Neurons from Parkinson Patients with Mutations in PINK1
Authors: Azkona G, López de Maturana R, Del Rio P, Sousa A, Vazquez N, Zubiarrain A, Jimenez-Blasco D, Bolaños JP, Morales B, Auburger G, Arbelo JM, Sánchez-Pernaute R.
Journal: Mol Neurobiol. 2018 Jan;55(1):506-516.
PMID: 27975167 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers the researchers were interested in another gene that is associated with Parkinson’s – called PTEN-induced putative kinase 1 (or simply PINK1). PINK1 is a protein that has a number of different functions in cells, but it is best understood in the context of cellular waste disposal (Click here to read a recent post that explains what PINK1 does).
In their study, the researchers increased levels of PINK1 in cells and they noticed something interesting about LRRK2:
As levels of PINK1 increased, levels of LRRK2 activity decreased
And when they lowered PINK1 levels in cells, guess what happened: LRRK2 levels of activity went up!
This finding suggests that people with PINK1 mutations who have Parkinson’s may have increased levels of LRRK2.
Interesting. What about people with spontaneous PD? People with PD who have no genetic risk factor. Do they have increased levels of LRRK2?
Data has been published which indicates that some people with idiopathic (or spontaneous) Parkinson’s do have increased levels of LRRK2 activity.
This report was published in 2018:
Title: LRRK2 activation in idiopathic Parkinson’s disease
Authors: Di Maio R, Hoffman EK, Rocha EM, Keeney MT, Sanders LH, De Miranda BR, Zharikov A, Van Laar A, Stepan AF, Lanz TA, Kofler JK, Burton EA, Alessi DR, Hastings TG, Greenamyre JT
Journal: Science Translational Medicine 25 Jul 2018: 10 (451), eaar5429.
PMID: 30045977 (This report is OPEN ACCESS if you would like to read it)
In this study, the researchers reported that many cases of idiopathic Parkinson’s showed about a 4-6 fold increase in markers of LRRK2 activity (compared with the control brains). This result suggested to the researchers that normal (un-mutated) LRRK2 protein may be highly activated in Parkinson’s.
So going back to your original question – this is why biotech companies like Denali are developing inhibitors of LRRK2 activity.
 LRRK2 protein. Source: Youtube
LRRK2 protein. Source: Youtube
|
# RECAP #1: LRRK2 is a protein that functions as a kinase – an enzyme that regulates the biological activity of other proteins Genetic variations in the LRRK2 gene can result in a hyperactive version of the protein. There is also data indicating that people with spontaneous PD have elevated levels of LRRK2 activity. Biotech companies are developing inhibitors of LRRK2 as potential disease modifying treatments for PD. # |
Ok so Denali have been developing LRRK2 inhibitors for Parkinson’s. Have they tested any of them yet?
Yes they have been clinically testing two of their inhibitors.
At the 2021 International Association of Parkinsonism and Related Disorders Virtual Congress, Dr Danna Jennings (Senior Medical Director at Denali) gave a poster presentation on some of the Phase I clinical work that Denali has conducted on one of their LRRK2 inhibitors.
 Source: IAPRD
Source: IAPRD
This was abstract of the presentation:
Title: LRRK2 inhibition by BIIB122/DNL151 in double-blind, placebo-controlled Phase 1 healthy volunteer and Phase 1B Parkinson’s disease trials
Authors: Jennings D, Wetering de Rooij J, Vissers M, Heuberger J, Groeneveld G, Maciuca R, Kay A, Borin M, Wong B, Daryani V, Huntwork-Rodriguez S, Cruz-Herranz A, Chin P, Ho C, Troyer M
Poster: P54 (The abstract for this poster is available here)
The poster presented data from two clinical trials. Both studies were Phase I investigations evaluating the safety, tolerability, pharmacokinetics, and pharmacodynamics of Denali’s LRRK2 inhibitor, DNL151 (also now known as BIIB122).
What is pharmacokinetics and pharmacodynamics?
In clinical trials, researchers are interested in investigating the pharmacodynamics and pharmacokinetics of a treatment.
Pharmacodynamics explores how a drug affects an organism (eg. mechanism of action) – basically, the researchers is asking what does the drug do to the body?
Pharmacokinetics, on the other hand, looks at how the organism affects the drug (eg. how well is it absorbed and excreted) – basically, the researchers is asking what does the body do to the drug.
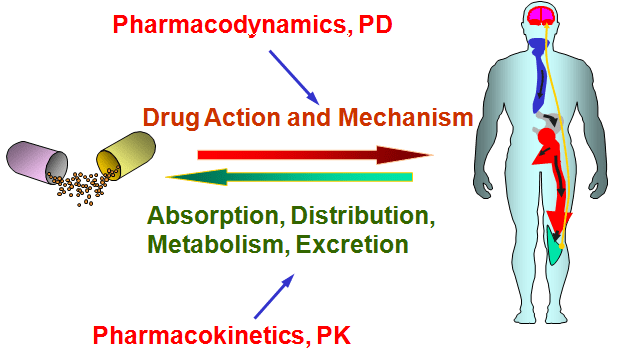 Source: Pinterest
Source: Pinterest
The first study (DNLI-C-0001) was a double-blind, placebo-controlled study, assessing single- and multiple ascending doses of DNL151 in healthy volunteers up to 28 days. This study was conducted in the Netherlands, and the single doses ranged from 15 mg to 300 mg for up to 28 days, while the twice daily doses went up to 400 mg for 14 days (Click here to read more about the details of this study).
The second study (DNLI-C-0003) was an international multicenter, double-blind, placebo-controlled, Phase 1b study evaluating 3 different dose levels of DNL151 in people with Parkinson’s over 28 days. The doses in this study were just once daily, up to 300 mg (Click here to read more about the details of this study).
Collectively, the studies involved 184 healthy volunteers (145 administered with DNL151 & 39 with placebo) and 36 people with Parkinson’s (26 administered with DNL151, 10 with placebo), so a lot of data was collected regarding the pharmacokinetics and pharmacodynamics of DNL151.
The results of both studies indicate that DNL151 was safe and generally well tolerated. Only 4 individuals discontinued treatment during the studies. No serious adverse events were observed and the majority of treatment-emergent adverse events reported were mild in nature and resolved after termination of treatment. Importantly, there were no clinically meaningful changes in pulmonary or renal function in either study. Overall, a positive outcome considering that this is a new drug class in humans.
Is safety and tolerability all the assessed in the study?
Phase I trials are usually short assessments of the safety of a treatment, but they can also provide the investigators with an opportunity to explore ‘target engagement’ to determine if the therapy is doing what it is supposed to.
The researchers in these studies also investigated various biomarkers of LRRK2 activity. For example, levels of the phosphorylated form of LRRK2 – known as pS935 LRRK2 – were measured in blood samples, and found to be less than half that observed in the placebo treated group across all of the three doses tested in the Parkinson’s patients (80 mg, 130 mg, and 300 mg given once daily for 28 days):
 QD means ‘once a day’. Source: Denali
QD means ‘once a day’. Source: Denali
This result suggested that DNL151 could reduce the hyperactivity of LRRK2. And this finding was supported by measures of another biomarker, phosphorylated RAB10 (pRAB10), which was less than half of the levels that were reported in the placebo group across all of the three doses tested in the Parkinson’s patients:
 QD means ‘once a day’. Source: Denali
QD means ‘once a day’. Source: Denali
What is phosphorylated Rab10?
RAB proteins are a group of proteins that sit on the inside of cell membranes. There are many types of RAB proteins and each of them has unique subcellular membrane distribution and take part in various functions.
LRRK2 is known to act on RAB proteins, but considerable research has focused on RAB10 in particular, since it was identified as a potential biomarker of LRRK2 activity in 2016:
 Title: Phos-tag analysis of Rab10 phosphorylation by LRRK2: a powerful assay for assessing kinase function and inhibitors.
Title: Phos-tag analysis of Rab10 phosphorylation by LRRK2: a powerful assay for assessing kinase function and inhibitors.
Authors: Ito G, Katsemonova K, Tonelli F, Lis P, Baptista MA, Shpiro N, Duddy G, Wilson S, Ho PW, Ho SL, Reith AD, Alessi DR.
Journal: Biochem J. 2016 Sep 1;473(17):2671-85.
PMID: 27474410 (This report is OPEN ACCESS if you would like to read it)
Basically, by phosphorylating RAB10 protein, LRRK2 blocks the ability of it to interact with other proteins, thereby trapping the phosphorylated RAB protein on the membrane and unable to performing its task (RAB10 appears to have important roles in lysosomal function – click here to read more about this). DNL151 treatment reduced levels of phosphorylated RAB10, allowing this protein to function normally.
Treatment with DNL151 was also associated with a dose-dependent reduction in di-22:6-BMP, a specific species of bis[monoacylglycerol] phosphate (BMP), which is another marker of lysosomal function (lysosomes are key components of the waste disposal system inside of cells):
 Source: Denali
Source: Denali
This data suggests that not only was DNL151 reducing LRRK2 activity, but this was also having downstream effect on LRRK2 targets in the blood of people with Parkinson’s over the 28 day time frame that the drug was tested. Overall an encouraging result.
One of the most intriguing aspects of the data shared thus far is that phosphorylated Rab10 levels were approximately 2x higher in patients with sporadic Parkinson’s (as well as in those with LRRK2 mutations) compared with healthy volunteers. This is another indication of elevated LRRK2 activity in idiopathic PD, which suggests that LRRK2 inhibition could be potentially useful in a wider PD cohort than simply those carrying LRRK2 mutations.
The data supports further clinical investigations, suggesting a once daily oral dosing regime. It will be interesting to see how DNL151 is tolerated over a longer period of treatment time and (of course) whether it can impact the progression of PD.
|
# # RECAP #2: LRRK2 inhibition is being clinically tested as a means of slowing the progression of Parkinson’s. Biotech company Denali Therapeutics recently presented safety and tolerability results from Phase I studies evaluating their LRRK2 inhibitor DNL151. The results indicate that the drug was safe and generally well tolerated over 28 days of treatment, and provide evidence of target engagement demonstrating that DNL151 is lowering levels of LRRK2 activity. # # |
What is Denali planning to do next?
In July 2020, the US FDA cleared an Investigational New Drug (IND) application for DNL151 enabling expansion of the clinical trial program.
And then August, Denali signed an agreement with the pharmaceutical company Biogen to co-develop and co-commercialise DNL151 and other small molecule inhibitors of LRRK2 for Parkinson’s (Click here to read the press release).
 To partner up, Biogen made an upfront payment of US$560 million to Denali Therapeutics, and they also took a US$465 million equity position in the company (13.3 million newly issued shares, representing 11.2% of the total shares). In addition, there are potential milestone payments (up to US$1.125 billion), profit sharing and royalties are part of the deal.
To partner up, Biogen made an upfront payment of US$560 million to Denali Therapeutics, and they also took a US$465 million equity position in the company (13.3 million newly issued shares, representing 11.2% of the total shares). In addition, there are potential milestone payments (up to US$1.125 billion), profit sharing and royalties are part of the deal.
Biogen and Denali will share responsibility and costs for global development (60% Biogen; 40% Denali), as well as profits and losses for commercialisation in the U.S. (50% Biogen; 50% Denali) and China (60% Biogen; 40% Denali). Outside of the U.S. and China, Biogen will be responsible for commercialisation and (if all goes well) pay Denali tiered royalties.
On the 10th January of this year, Denali Therapeutics provided some details on their plans for the future development of BIIB122/DNL-151 (Click here to read the press release).
Specifically, they are planning two late stage clinical trials – which will be called LIGHTHOUSE and LUMA.
And these studies will be very different in their design.
The LIGHTHOUSE study will be a global Phase 3 clinical trial and it will be seeking to recruit 400 people with Parkinson’s who also carry a LRRK2 gene variant. These individuals will be treated with either BIIB122 or placebo for at least 96 weeks.
The LUMA study will be a large Phase 2b clinical trial of 640 people with Parkinson’s. Importantly, the study participants will NOT have any variant in the LRRK2 gene and the study treatment period of BIIB122 or placebo will be 48 weeks.
In both studies, clinical symptoms of participants will be assessed to determine whether there is a decrease in the rate of progression of Parkinson’s in those individuals treated with BIIB122.
The company has not provided a start date for either study, but we can assume that they will be keen to get started very shortly (next 6 months).
 Source: Centogene
Source: Centogene
In collaboration with the genotyping company Centogene, Denali has been preparing for the LIGHTHOUSE study by screening people with Parkinson’s for LRRK2 genetic variants in the Rostock International Parkinson’s Disease (ROPAD) Study (Click here to read a previous SoPD post about this study).
Denali has also set up a website (EngageParkinson’s) for anyone seeking to learn more about their LRRK2 inhibitor program and the future clinical trials:
Are other companies developing LRRK2 inhibitors as well?
Yes, this area of research is fast becoming rather crowded and very busy.
One pharmaceutical company with a major LRRK2 inhibitor research program is GlaxoSmithKline. In 2018, the company signed on for a 4 years partnership with the DNA analysis company 23andMe.
 As part of that deal, GSK is contributing its LRRK2 inhibitor program, and they may be hoping to use 23andMe’s database of people who know their LRRK2 genetic status (Source). GSK also initiated an observation clinical study exploring LRRK2-associated biomarkers at King’s College in London. That study was scheduled to complete in early 2021 (Source), and given that the 4 year deal is coming to an end, we will hopefully be learning more about the future plans for the GSK LRRK2 program in 2022.
As part of that deal, GSK is contributing its LRRK2 inhibitor program, and they may be hoping to use 23andMe’s database of people who know their LRRK2 genetic status (Source). GSK also initiated an observation clinical study exploring LRRK2-associated biomarkers at King’s College in London. That study was scheduled to complete in early 2021 (Source), and given that the 4 year deal is coming to an end, we will hopefully be learning more about the future plans for the GSK LRRK2 program in 2022.
Other biotech companies building a LRRK2 inhibitor program include Cerevel Therapeutics.
 This is a biotech firm that was started by Bain Capital and the Pharmaceutical company Pfizer (Click here to read more about this). Cerevel has taken on many of the neuroscience treatments that Pfizer was clinically testing until it shut down their neuroscience division in early 2018. In addition to those clinically tested assets, Cerevel have also quietly added ‘LRRK2 inhibitor’ to their preclinical ‘lead development’ area of research (Click here to read more about this).
This is a biotech firm that was started by Bain Capital and the Pharmaceutical company Pfizer (Click here to read more about this). Cerevel has taken on many of the neuroscience treatments that Pfizer was clinically testing until it shut down their neuroscience division in early 2018. In addition to those clinically tested assets, Cerevel have also quietly added ‘LRRK2 inhibitor’ to their preclinical ‘lead development’ area of research (Click here to read more about this).
A biotech company called E-Scape Bio is also developing a LRRK2 inhibitor program (Click here to read more about this).
 E-Scape Bio have a genotyping study in collaboration with Sano Genetics to help identify individuals with Parkinson’s and a LRRK2 variant in North America, and they are seeking to start clinical trials in the near future (Click here to read more about this).
E-Scape Bio have a genotyping study in collaboration with Sano Genetics to help identify individuals with Parkinson’s and a LRRK2 variant in North America, and they are seeking to start clinical trials in the near future (Click here to read more about this).
And there is a collaboration between Servier and Oncodesign to develop novel LRRK2 inhibitors (Click here to read more about this).
And then there is of course the Biogen LRRK2 inhibition program.
The one with Denali?
No. The other Biogen LRRK2 inhibition program.
There’s more than one?!?
One interesting question raised by the collaboration between Biogen and Denali is how it affects the Biogen LRRK2 inhibition program with another biotech company called Ionis Pharmaceuticals, which is developing a novel LRRK2 approach in collaboration with Biogen.
 This company is conducting a Phase I clinical trial to evaluate their lead candidate, called BIIB094.
This company is conducting a Phase I clinical trial to evaluate their lead candidate, called BIIB094.
BIIB094 is an antisense oligonucleotide – this is an approach that blocks LRRK2 RNA before it can be used to make LRRK2 protein (we have discussed antisense oligonucleotides in a previous SoPD post – click here to read that post). The trial is a Phase I clinical trial to evaluate the safety and tolerability of single and multiple doses of BIIB094 (Click here to learn more about that trial).
Biogen appears to be doubling down on LRRK2 and placing some major bets that reducing the activity of this protein is the path forward for treating Parkinson’s.
Let’s hope those bets pay out.
So what does it all mean?
News of a pair of late stage clinical trials – on the back of positive Phase 1 clinical trial results provides encouraging signs of progress in the field of LRRK2 inhibition for Parkinson’s. But there is still a long way to go in clinical testing for this therapeutic class and caution must be taken on that journey.
LRRK2 is a complex protein. It has two enzymatic activities and a number of additional functions. Certain genetic mutations in the LRRK2 gene are associated with increased risk of developing Parkinson’s, but not all of these mutations have the same effect on the activity of the subsequent LRRK2 protein. In addition, the future trials of experimental therapies like BIIB122/DNL151 may be difficult clinical studies to conduct, given the slower rate of progress in PD symptoms in LRRK2 mutation carriers (longer studies may be required in order to see an effect on disease progression).
Apologies if I am raining on the parade here, but expectation must be managed. LRRK2 inhibitors represent an exciting new class of possible therapeutics for Parkinson’s, but it is still early days in their development and we have a great deal to learn.
All of that said, we will be keeping a keen eye out for any news about the start dates for the LIGHTHOUSE and LUMA clinical trials later this year.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE: Denali Therapeutics and other companies mentioned in this post are publicly traded companies. The material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. Denali Therapeutics and other companies have not requested that this material be produced, nor has the author had any recent contact with these companies or associated parties. This post has been produced for educational purposes only.
The banner for today’s post was sourced from lighthouseglobal






Hello Simon. great post.
“The LUMA Phase 2b study will recruit about 640 patients without LRRK2 mutations”
Are they excluding people with mutations on GBA gene?
Isn’t it that reducing LRRK2 activity in people with GBA mutations could be detrimental?
LikeLike