|
# # # # In this end-of-year post, we review the Parkinson’s research that caught our attention at SoPD HQ in 2020. Month-by-month we will briefly discuss some of the major pieces of research/ announcements that have defined the year and advanced our understanding of Parkinson’s. The list is based on nothing more than the author’s personal opinion – apologies to any researchers who feel left out – and the contents should certainly not be considered definitive or exhaustive. It was just some of the stuff that made me say “wow” in 2020. And in the next SoPD post, we will conduct our annual horizon scan and consider what 2021 may have in store for us. # # # # |
 Source: PhysicsWorld
Source: PhysicsWorld
More than any other year, 2020 saw the best and worst of us.
It was a ridiculous 365 days (you couldn’t make up half the stuff that happened – e.g. “bleach”), and also one of the most humbling periods of our lives. In many ways we came together and stepped up to face challenges (e.g. the COVID-19 vaccine efforts), and yet at that same time if you listen to any of the 24-hour idiotic noise we have never been so divided. Some of people showed tremendous courage (e.g. the front line medics), and others of us were found to be wanting and learnt how little (if any) fortitude we truly have.
It was a dreadful year, but at the same time one that has been strangely fascinating to experience.
And despite the setbacks brought on by the COVID-19 situation, there has been remarkable progress in the arena of Parkinson’s research and in today’s post we will do both a short and long review of 2020, according to research-related events/publications that we here at the SoPD thought were of note.
THE SHORT REVIEW: A top five
If we had to select a top 5 Parkinson’s research-related events/highlights of 2020, they would be:
1. Biotech buyouts
There has previously been an outcry regarding the departure of certain members of the pharmaceutical industry from neurodegeneration (Click here to read an old SoPD rant about this).
Several major industry players were reeling on the back of large failed clinical trials and “brain” was being considered too hard as huge research departments were abondoned. In 2020, however, we saw the start of a reversal of that trend with the announcement of collaborations and aquisitions of a number of major biotech companies by big Pharma.
Some of the more prominent examples were:
- Biogen tied a knot with Denali (Click here to read an SoPD post about this)
- Bayer acquired Asklepios BioPharmaceutical who then merged with Brain Neurotherapy Bio -the GDNF gene therapy biotech firm (Click here to read a SoPD post about this – and don’t forget that Bayer purchased BlueRock Therapeutics – the stem cell transplantation company last year – click here to read a SoPD post about that).
- Bial purchased Lysosomal Therapeutics (Click here to read more about this)
- Eli Lilly acquired GBA-associated Parkinson’s gene therapy company Prevail Therapeutics (Click here to read a SoPD post on this)
- Inflammasome-focused biotech firm Inflazome was bought by Roche (Click here to read more about this).
All of this is exciting for the Parkinson’s community, but now we need to see the fruits of these marriages. The Pharmas will be able to bring considerable resources and a more long-term strategy to aid these research programmes, but this will not have material benefit for the companies if they can not speed up the clinical development of the experimental therapies being proposed.
2. A massive increase in funding
One of the big research events in 2020 was the announcement of the first funding awards from the Aligning Science Across Parkinson’s (or ASAP) initiative.
This is a return-to-basic/”first principles” effort for Parkinson’s, and in August the first round of major research grants were announced, providing $161 million in research funding to 21 projects (involving 96 research leaders from 60 institutions across 11 countries) for the next three years (Click here to read a SoPD post about this).
And almost immediately, the next round of grant applications were opened for submission.
To put this massive investment into perspective for you, in 2016 the total amount issued by the entire US Government for research into Parkinson’s (via NIH funding) was $161M:
 NIH Parkinson’s research funding. Source: NIH
NIH Parkinson’s research funding. Source: NIH
In other words, in just their first round of funding, the ASAP initiative have matched what the US Government spent on Parkinson’s research in 2016. And they are already open for business for next year’s round of funding.
I have been in this game for a while, but that ‘$160 million’ number kinda floored me. Few previous research funding rounds come even remotely close to that for a single neurodegenerative disease.
It is an amount of money that should seriously shift the dial. And it will be interesting to see what comes from the funded projects over the next 3 years.
3. First immunotherapy clinical trial results in Parkinson’s
Immunotherapy is a means of boosting the body’s immune system to target specific pathogens (such as a new virus or a troublesome protein). Early in 2020, the pharmaceutical company Roche and the biotech company Prothena Corp announced that their alpha synuclein-focused immunotherapy treatment Prasinezumab had not met the primary endpoint (a pre-designated measure of efficacy) of their Phase II PASADENA clinical trial.
 But when the companies conducted a deep dive, post hoc analysis of the data, they found hints of prasinezumab actually having an impact on motor progression in their study.
But when the companies conducted a deep dive, post hoc analysis of the data, they found hints of prasinezumab actually having an impact on motor progression in their study.
“As previously reported, the study did not meet the primary objective, but signals of efficacy showing a reduction in disease progression were observed in both of the prasinezumab arms when compared to placebo” (Source)
The researchers found that prasinezumab “significantly reduced decline in motor function by 35% (pooled dose levels) vs. placebo after one year of treatment” on MDS-UPDRS Part III, and was associate with a “delayed time to clinically meaningful worsening of motor progression over one year”. The companies stated that the “results support further clinical development of prasinezumab” (Click here to read more about this and click here to read a SoPD post on this topic).
I’ll be completely honest, I had no expectations that this study would show any effect. This was not that I had/have no faith in the treatment, it is simply a survival mechanism on my part – I have seen too many failed trials to expect anything to work!
Given the disasterous landscape of immunotherapy trials for Alzheimer’s, it is encouraging that the first double-blind immunotherapy study for Parkinson’s showed any signs of life. That said, it must be remembered that post hoc analysis is supposed to be “hypothesis generating” and should not be considered evidence of efficacy.
It will be interesting to see the longer-term follow up of the Pasadena cohort and the results of the second Phase II immunotherapy clinical trial (the “SPARK study” being conducted by Biogen), which is scheduled to announce results in mid 2021 (see next post).
4. Lots of interesting clinical trial results
It used to be that only a few clinical trials were conducted each year for Parkinson’s, but as a recent review of the Parkinson’s clinical trial pipeline demonstrated (Click here to read more about that) there is now a lot of ongoing clinical trials. And in 2020 we received the results of a large number of those trials.
To highlight just a few:
- The results of the Phase II Ambroxol clinical trial in Parkinson’s were published (Click here to read a SoPD post about this)
- Results of Neuropore Therapies‘s Phase I trial of their drug NPT520-34 (a toll-like receptor 2 antagonist) were announced (Click here to read more about this)
- Denali Therapeutics announced Phase 1b study results for their LRRK2 inhibitor DNL201 (Click here to read more about this)
- The results of an open‐label pilot study of Ursodeoxycholic acid (UDCA) were published (Click here to read a SoPD post on the topic)
- Biotech firm Herantis Pharma announced topline results of Phase 1-2 CDNF clinical trial in Parkinson’s (Click here to read a SoPD post on this topic).
- The Phase III isradipine/”STEADY-PD” clinical trial results were published (Click here to read a SoPD post on this topic).
- Biotech firm resTORbio announced interim results from 3 cohorts for Phase 1b/2a trial of the TORC1 inhibitor RTB101 in Parkinson’s (Click here to read more about this).
- Biotech firm Theranexus announced that their drug THN102 met the primary efficacy endpoint of its Phase 2 clinical trial by significantly reducing excessive daytime sleepiness in Parkinson’s (Click here to read more about this).
- The results of the Stanford young plasma study in Parkinson’s were published (Click here to read a SoPD post on this topic)
- The initial results from the Phase II PD-STAT clinical trial assessing the cholesterol-lowering Simvastatin in Parkinson’s were announced (Click here to read more about this)
- The results of the Phase II NILO-PD clinical study of nilotinib in Parkinson’s were published (Click here to read more about this)
And in 2021 we will be finding out the results of a lot more (see next post).
5. Parkinson’s research during COVID-19
There were many examples of unsung heroic efforts in the Parkinson’s research arena made during the last 12-months. In my role as deputy director of research at the Cure Parkinson’s Trust, I have been made aware of a few cases that deserve mention in this review.
The global COVID-19 situation made life very difficult for everyone in 2020, bringing hardship in terms of loss of life and economic downturn. It also forced laboratories around the world to stop ongoing experiments and shut down during the lockdown periods. Remarkably – despite the obstacles – several ongoing Parkinson’s clinical trials were able to finish collecting data during this period, thanks to the amazing efforts of the researchers and participants involved.
Two examples of this are the UP (“UDCA in Parkinson’s”) study and the PD-STAT (Simvastatin) study here in the UK. At the start of the lockdown both studies were still active and collecting critical data. The coordinators of the studies had to quickly adapt and find safe solutions for their staff and participants to continue collecting data. Remarkably, everyone involved were able to safely work together to finish these studies with only a few data points missing in the end. Both studies were incredible efforts (and not alone – they are simply the examples that I am most familiar with).
More than anything, this last one was a highlight for me in terms of Parkinson’s research in 2020. It shows that despite everything that the last 12 months threw at us, we were able to make progress and keep the ball rolling. It also stimulated a great deal of activity around new ideas for remote assessment for clinical trials, with the goal of more real-world data collection that won’t be disrupted by future global events that could impact our normal way of life.
The future implications of all of these 2020 highlights are exciting and bode well for the future of Parkinson’s research.
# # # # # #
And now for the long review… maybe grab a cup of tea, this is rather long:
 On the 23rd January, the Chinese city of Wuhan was placed under quarantine, in which all public transport in and out of the city was suspended in efforts to control the spread of a new coronavirus, designated 2019-nCoV. This was the start of a global pandemic that would change our lives and come to define the entire year.
On the 23rd January, the Chinese city of Wuhan was placed under quarantine, in which all public transport in and out of the city was suspended in efforts to control the spread of a new coronavirus, designated 2019-nCoV. This was the start of a global pandemic that would change our lives and come to define the entire year.
In January, the results of the Ambrxol clinical trial in Parkinson’s (supported by The Cure Parkinson’s Trust, the Van Andel Institute, & John Black Charitable Foundation). The drug was found to be safe and well tolerated at the doses used, it also accessed the brain and raised levels of the target protein, glucocerebrosidase (Click here to read more about this study, click here for the summary and click here to read a SoPD post about this topic)
In other research, scientists in Korea reported that in the postmortem Parkinsonian brain there are a significant portion of “dormant dopamine neurons”. Rather than immediately dying, the investigators found that dopamine neurons in the substantia nigra region of the brain simply stop producing the neurotransmitter dopamine. They found that in both mild & severe Parkinson’s brains, dormant dopamine neurons could account for 54.7% & 27.0% of the dopamine neurons, respectively – raising the possibility that “dormant but recoverable” dopamine neurons are still alive in the brain in Parkinson’s (Click here to read more about this, click here to read the press release, and click here to read a SoPD post on this study).
 In addition, researchers in Scotland reported that the probiotic Bacillus subtilis protects against Parkinson’s-associated α-synuclein aggregation in microscopic roundworms (C. elegans), by changing the host sphingolipid metabolism (Click here to read more about this, click here for a press summary, click here for the Parkinson’s UK summary, and click here to read a SoPD post on this topic). Later in the year, it was announced that the researchers had been awarded £750,000 to further study the disease-modifying potential of a probiotic bacterium in Parkinson’s, including a clinical trial in Edinburgh (Click here to read more about this).
In addition, researchers in Scotland reported that the probiotic Bacillus subtilis protects against Parkinson’s-associated α-synuclein aggregation in microscopic roundworms (C. elegans), by changing the host sphingolipid metabolism (Click here to read more about this, click here for a press summary, click here for the Parkinson’s UK summary, and click here to read a SoPD post on this topic). Later in the year, it was announced that the researchers had been awarded £750,000 to further study the disease-modifying potential of a probiotic bacterium in Parkinson’s, including a clinical trial in Edinburgh (Click here to read more about this).

 In February, as the world was slowly becoming aware of the COVID-19 threat, astronomers quietly announced that Earth had acquired a second, slightly smaller moon. Designated 2020 CD3, a calculation of its orbit suggests that it has been orbiting Earth for approximately 3 years. But given that it is the size of a car, don’t expect a second moon landing any time soon (Click here to read more about this).
In February, as the world was slowly becoming aware of the COVID-19 threat, astronomers quietly announced that Earth had acquired a second, slightly smaller moon. Designated 2020 CD3, a calculation of its orbit suggests that it has been orbiting Earth for approximately 3 years. But given that it is the size of a car, don’t expect a second moon landing any time soon (Click here to read more about this).
In Parkinson’s news, two research reports published back-to-back suggesting that apolipoprotein E4 (APOE4) genotype – a major risk factor for neurodegenerative diseases, particularly Alzheimer’s – can affect α-synuclein pathology in mouse models & cases of α-synucleinopathy, like Parkinson’s. The first paper used genetically engineered mouse models of alpha-synucleinopathy & analyzed cognition in patients with Parkinson’s to demonstrate that APOE4 directly regulates α-synuclein pathology & is associated with faster cognitive decline (in 2 separate cohorts – click here to read this paper). And the second study also generated a mouse model of alpha-synucleinopathy & reported that APOE4 (but not APOE2 or APOE3) exacerbated Parkinson’s-associated α-synuclein pathology… independent of beta amyloid (click here to read more about this, click here to read the press release, and click here to read the SoPD post on this topic).
Biotech firm Herantis Pharma announced topline results of Phase 1-2 CDNF clinical trial in Parkinson’s. Confirms positive safety & tolerability of CDNF in advanced-PD, + encouraging PET imaging in some patients. CDNF is “Cerebral dopamine neurotrophic factor”, considered a neurotrophic factor though it functions differently to other NFs like GDNF. This study has involved implantation of tubes into the brain & monthly delivery of CDNF for 6+6 months (Click here to read the press release and click here to read a SoPD post on this topic).
Researchers also published the results of an open‐label, prospective, multiple‐ascending‐dose study of oral Ursodeoxycholic acid (UDCA) in people with Parkinson’s. The results of this small study (only 5 participants) suggest that the drug is safe and well tolerated in people with Parkinson’s. The treatment also resulted in modest increases in ATP (energy) levels (based on brain imaging techniques). The investigators concluded that the “findings warrant additional studies” (Click here to read more about this and click here to read a SoPD post on the topic). Based on these results, we look forward to seeing the findings of the larger “UP study” (UDCA in Parkinson’s) being conducted in the UK – results in 2021 (Click here to read a SoPD post about that).
Despite the restraints that COVID-19 lockdown forced on large parts of the world, there were many who tried to make us feel better. One example was Daniel Matarazzo with his Coronarvirus-parody of “Supercalifragilisticexpialidocious“:
In Parkinson’s research, researchers conducted an analysis of the longest postmortem trophic factor gene therapy Parkinson’s cases reported to date and their results were very interesting! The clinical trial of AAV2-neurturin (CERE120) from a decade ago did not meet its primary endpoint, but this new data suggests that there was a biological effect. In the putamen, TH+ fibres were observed in areas of detectable neurturin expression; In the nigra, melanized neurons displayed intense TH & RET expression in the patient where CERE120 was directly delivered to the nigra; Interestingly, there was no difference in the degree of Lewy pathology. The results suggest that the neurturin gene therapy only covered a relatively small areas of the putamen (3-12% – click here to read more about this, click here to read a lay summary, and click here to read a SoPD post about it).
In France researchers demonstrated the feasibility of directly administering a stable form of dopamine (“A-dopamine”) into the brain in a primate model of Parkinson’s (MPTP). They reported improved dopa-responsive motor and cognitive symptoms, and no dyskinesia under continuous A-dopamine delivery (vs peripheral L-dopa). A biotech company – called InBrain Pharma – has been set up to clinically develop this new approach, and clinical trials have been initated (Click here to read more about this and click here to read a SoPD post on this topic).
 Researchers also published a remarkable report presenting “GTCA” (“genetically targeted chemical assembly”) as an experimental means of restorative therapy for the brain. Combining genetic engineering & polymer chemistry, researchers at Stanford University directly leveraged complex cellular architectures of living organisms to synthesize and assemble bioelectronic materials. These functional artificial structures make connection between neurons, stimulating active and enabling better survival of cells (Click here to read more about this, click here to read the press release, and click here to read a SoPD post about this).
Researchers also published a remarkable report presenting “GTCA” (“genetically targeted chemical assembly”) as an experimental means of restorative therapy for the brain. Combining genetic engineering & polymer chemistry, researchers at Stanford University directly leveraged complex cellular architectures of living organisms to synthesize and assemble bioelectronic materials. These functional artificial structures make connection between neurons, stimulating active and enabling better survival of cells (Click here to read more about this, click here to read the press release, and click here to read a SoPD post about this).
March also saw the initiation of online Parkinson’s research seminar series that were focused on supporting early career researchers, giving them an opportunity to share their research during the COVID lockdown period. These included 3P Seminars (co-ordinated by the Van Andel Institute, World Parkinson’s Congress, and The Cure Parkinson’s Trust) and also PD seminars (which is supported by Parkinsons’s UK).
 While certain billionaires were playing down the threat of COVID, other billionaires were stepping up to help. For example, Jeff Bezos (Amazon founder) donated $100 million to Feeding America, a nonprofit that helps food banks feed families in need (Source).
While certain billionaires were playing down the threat of COVID, other billionaires were stepping up to help. For example, Jeff Bezos (Amazon founder) donated $100 million to Feeding America, a nonprofit that helps food banks feed families in need (Source).
 Billionaire Jack Ma (founder of Ali Baba) donated 1.1 million COVID-19 testing kits, 6 million masks and 60,000 medical use protective suits/face shields to Africa.
Billionaire Jack Ma (founder of Ali Baba) donated 1.1 million COVID-19 testing kits, 6 million masks and 60,000 medical use protective suits/face shields to Africa.
And billionaire Jack Dorsey (founder of Twitter) announced he was donating $1 billion (28% of his net wealth) to help fund COVID-19 relief.
 For more on how billionaires are supporting the COVID-19 effort – click here.
For more on how billionaires are supporting the COVID-19 effort – click here.
As for Parkinson’s research, in April the Phase III isradipine/”STEADY-PD” clinical trial results were published. This was a major study, involving 300 participants with early-stage Parkinson’s being treated with either the blood pressure medication isradipine or placebo over a 36 months period. The conclusion of the study: “Long-term treatment with immediate-release isradipine did not slow the clinical progression of early-stage Parkinson’s” (Click here to read more about this, click here to read a lay summary, and click here to read a SoPD post on this topic).
In other clinical trial news, the pharmaceutical company Roche & biotech firm Prothena announced that the Phase 2 PASADENA alpha synuclein immunotherapy (prasinezumab) trial in Parkinson’s did not meet the primary objective, but showed signals of efficacy. These results were from Part 1 of the study (52 week, double-blind, randomised, 3-arm study that enrolled 316 participants with recently diagnosed Parkinson’s, who were allocated to placebo, high dose, or low dose). Part 2 (52 week extension) is ongoing. Treatment was well tolerated & safe, but the key sentence in the announcement: “Signals were observed on multiple prespecified secondary & exploratory clinical endpoints” (Click here to read more about this, click here to read a SoPD post on this topic, and click here for an interview with the investigators from the Michael J Fox Foundation).
Cellivery Therapeutics presented new data on their cell-permeable Parkin protein (iCP-Parkin), that rescues neurons from accumulation of damaged mitochondria & pathological α-synuclein. The researchers report iCP-Parkin autoubiquitination and cytoprotective activities do not require PINK1; it also induces mitophagy & mitochondrial biogenesis, & demonstrates neuroprotection in toxin-induced & α-synuclein–induced models of PD (Click here to read more about this and click here to read a SoPD post on this topic).
 In May, the age of commercial human spaceflight officially began with the crewed launch of the SpaceX Crew Dragon Demo-2, carrying astronauts Bob Behnken and Doug Hurley to the International Space Station.
In May, the age of commercial human spaceflight officially began with the crewed launch of the SpaceX Crew Dragon Demo-2, carrying astronauts Bob Behnken and Doug Hurley to the International Space Station.
May also brough news for the Parkinson’s community about a N=1 case study involving a personalized cell transplantation operation. The results of the study were published in the New England Journal of Medicine. The researchers involved collected skin cells from a gentleman with Parkinson’s, coverted them into stem cell, that they further differentiated into dopamine neurons. They tested these cells in a humanized mouse model, before growing more cells and bilaterally implanting them into the gentleman in question “without the need for immunosuppression”. Brain imaging (PET) indicates that the grafted cells have survived and the clinical data suggests improvement – though a placebo response may be at play (Click here to read more about this, click here to read the STATnews article about it – click here for the follow up article questioning the ethics of the study – and click here to read a SoPD post on this topic).

Researchers involved in the Phase 2 GDNF trial proposed the “Parkinson’s Disease Comprehensive Response” (PDCORE) – a novel weighted composite endpoint integrating changes measured in 3 established outcomes. The “results are not described to provide post-hoc evidence of the efficacy of GDNF but rather are presented to further the debate of how current regulatory approved rating scales may be combined to address some of the recognized limitations of using individual scales in isolation” (Click here to read more about this and click here to read a SoPD post on this topic).
Researchers put a manuscript on the preprint database, bioRxiv that presented a novel high-throughput & hypothesis-free approach to detecting the existence of Parkinson’s genetic risk linked to any particular biological pathway. Utilising polygenic risk scores, gene-set burden analyses, cell type expression specificity enrichment, & summary-data based MR, they “highlight several promising pathways & genes for functional prioritization & provide a cellular context in which such work should be done”. As part of this study, they also created a foundational resource for the Parkinson’s community that can also be applied to other neurodegenerative diseases with complex genetic etiologies: https://pdgenetics.shinyapps.io/pathwaysbrowser (Click here to read more about this).
 In June, as a Siberian town – Verkhoyansk – broke the record for the highest temperature ever recorded within the Arctic Circle – it hit 38 degrees celsius (that is 100.4F), the Parkinson’s research community received news regarding a potential vaccine for Parkinson’s.
In June, as a Siberian town – Verkhoyansk – broke the record for the highest temperature ever recorded within the Arctic Circle – it hit 38 degrees celsius (that is 100.4F), the Parkinson’s research community received news regarding a potential vaccine for Parkinson’s.
In June, the Austrian biotech firm AFFiRiS published the results of their Phase 1 safety & immunogenicity work of the α-synuclein active immunotherapeutic PD01A in patients with Parkinson’s. The results demonstrated that over multiple years of assessments, the treatment was found to be safe and well tolerated. It also caused the immune system to generate antibodies targetting the aggregated form of alpha synuclein, and the investigators saw a reduction in the levels of this protein. Important to remember that this is an open-label study, but the researchers also reported no change in MDS-UPDRS part 3 scores or brain imaging data. The study was not designed to assess clinical efficacy of the immunotherapeutic, which “requires further exploration”. A Phase 2 trial is now being prepared ( to read the report, click here to read a press release, and click here to read a SoPD post of this result).
Genetic risk factors puts you at risk, while the genetic modifiers influence the level of risk. In June, researchers in New Zealand and Australia published a report exploring the effect that common genetic variations might have on the Parkinson’s-associated GBA gene. They identified common noncoding variants both within and outside of the GBA gene that may act as genetic modifiers of GBA activity. They speculated that some of these mutations could explain aspects of the variability between individuals with regards to age of symptom onset ( to read more about this and click here to read an SoPD post about this research).
In June, researchers report that Fyn-dependent regulation of Kv1.3 channels may play a role in augmenting the neuroinflammatory response in Parkinson’s, identifying Kv1.3 as a potential therapeutic target. They found that administration of Kv1.3-specific small-molecule inhibitor “PAP-1” significantly inhibited neurodegeneration & neuroinflammation in multiple animal models of Parkinson’s ( to read more about this).

 In July 2020, Capt “Sir” Tom Moore was knighted – what a legend! (Click here to read more about this).
In July 2020, Capt “Sir” Tom Moore was knighted – what a legend! (Click here to read more about this).
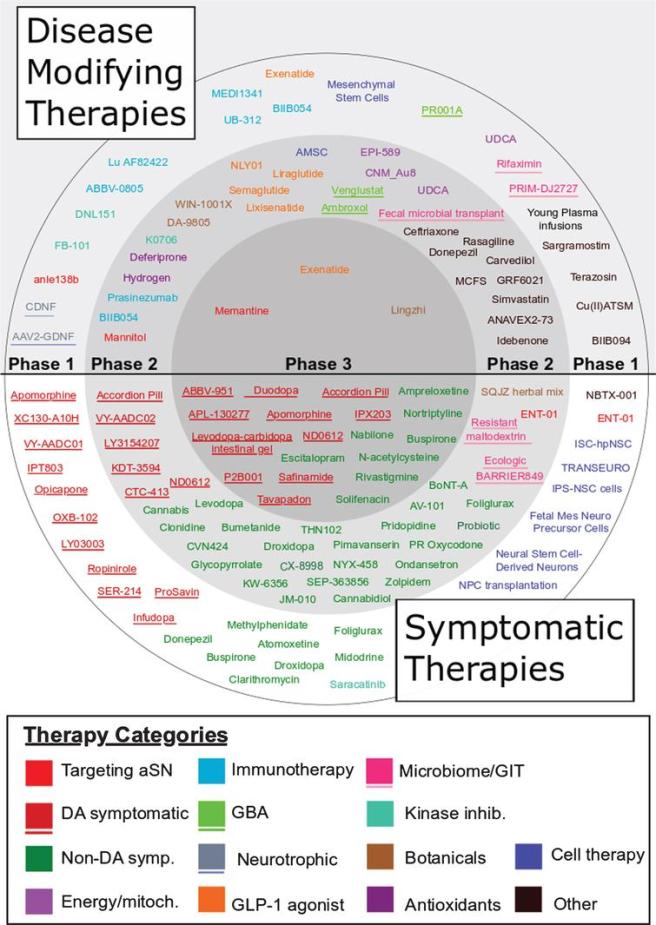
In July, an indepth analysis of the clinical trial landscape from PD research (super) advocates Kevin McFarthing (@InnovationFixer) Sue Buff (@SueBCal1), & Gary Rafaloff (@grafaloff) and The Cure Parkinson’s Research team in July. It provided an overview & analysis of 2019-2020 clinical drug development pipeline for Parkinson’s – both symptomatic & disease modifying. The study explored data from 145 Parkinson’s clinical trials that were ongoing during 2019-2020, breaking them down into phase & agent type to give the PD community a broad view of the clinical research landscape. Some very interesting trends appear in the data – such as 57 trials (39%) of all the trials were focused on long-term disease modifying therapies. (Click here to read more about this and click here to read a SoPD post about it).
In July, researchers in California reported the results of their safety study exploring young plasma infusions in Parkinson’s. The study involved 15 participants, and it found that the treatment was safe, feasible, well tolerated and warranted “further therapeutic investigations in PD” ( to read more about this and click here to read a SoPD post on this topic)
In August, the appropriately named Furnace Creek (in Death Valley, California – population of 24) logged a day time high temperature of 130° Fahrenheit (54.4° Celsius) – it was the “highest temperature ever reliably recorded on Earth“.
In August, the San Francisco-based biotech firm Denali Therapeutics – which is leading the development of LRRK2 inhibitors for Parkinson’s – firstly announced that they have signed an agreement to co-develop & co-commercialise their small molecule LRRK2 inhibitors with the pharmaceutical company Biogen ( to read more about this). In addition, Denali announced that their LRRK2 inhibitor DNL151 has been selected to progress to late stage studies in Parkinson’s patients with a LRRK2 kinase activating genetic mutations & in sporadic PD cases. Denali & Biogen are finalizing plans for the LRRK2 program & intend to commence two separate Parkinson’s clinical trials in 2021 (Click here to read more about this and click here to read an SoPD post on the topic).
An interesting report published in August focused on the use of a particular protein as a biomaker for Parkinson’s. Alpha synuclein is a protein that aggregates in the brain of many people with Parkinson’s, and it can be detected in blood, leading to suggestions that it could be a biomarker in PD. Researchers from the Systemic Synuclein Sampling Study, however, provided class III evidence that total cerebrospinal fluid (CSF) alpha synuclein does not accurately distinguish Parkinson’s from healthy controls. They found lower total alpha synuclein levels in the CSF in PD, but the specificity of this result was low. Plus alpha synuclein monoclonal antibody staining on skin & submandibular gland samples is specific for PD, but (again) sensitivity is low. The researchers also pointed out that “relationships within subjects across different tissues & biofluids could not be demonstrated“. Thus, alpha synuclein does not appear to be an ideal biomarker ( to read more about this).

September started with researchers announcing that they have found a way to float toy boats under a levitating liquid (Click here to read more about this and click here to read a press summary).
In September, the initial results from the Phase II PD-STAT clinical trial assessing the cholesterol-lowering Simvastatin in Parkinson’s were announced. The study coordinators indicated that the drug “does not slow progression of PD” (Click here to read more about this).

The results of another double‐blind, randomised placebo‐controlled trial were reported in September – this time evaluating Helicobacter pylori eradication therapy in Parkinson’s. The study found no improvement in clinical outcomes in Parkinson’s after 12 months; “there is no justification for routine HP screening or eradication with the goal of improving PD”. 80 participants were randomised at the start of the study; 67 included in the final full‐analysis set (32 treatment group patients, 35 placebo patients); assessment were undertaken at weeks 6, 12, 24, & 52. But there was no significant improvement in any motor, nonmotor, or quality‐of‐life outcomes (Click here to read more about this). This was a follow up study to previous research (Click here to read that study report).
In September there was also a report providing data supporting the existence of “brain-first” and “body-first” subtypes of Parkinson’s. The study found that individuals with de novo Parkinson’s with & without REM sleep behaviour disorder are very similar clinically, but they display strikingly different profiles on a multimodal imaging battery (Click here to read more about this and click here to read the press summary).
In October, it was announced that Emmanuelle Charpentier and Jennifer Doudna had been awarded the 2020 Nobel Prize for Chemistry “for the development of a method for genome editing” (aka CRISPR technology – Click here to read more about this).
Researchers also reported in October that use of DPP4 inhibitors &/or GLP-1 agonists (such as exenatide) were associated with a lower rate of Parkinson’s in people with diabetes compared to the use of other oral antidiabetic drugs (Click here to read more about this, click here for the press summary, and click here to read an SoPD post on this topic).
New research published in October highlighted the potential of IL6 as progression marker in PRKN/PINK1-associated PD. Increased levels of circulating cell-free mitochondrial DNA in serum from patients with PRKN/PINK1 mutations was also reported. The researchers pointed towards the cGAS-STING pathway as being potentially involved (Click here to read more about this, click here to read the press summary, and click here to read a SoPD post on this topic).
In October there was also a research report indicating that plasma caffeine concentrations are lower in people with Parkinson’s (vs control subjects; p<0.001), more so among LRRK2+ carriers (-76%) than among LRRK2- subjects (-31%). Similar results were also observed for caffeine metabolites. Given that caffeine intake has previously been associated with a reduced risk of developing Parkinson’s, could raising caffeine levels in people with Parkinson’s have beneficial effects? (Click here to read more about this).
 In November, it was reported that multiple COVID vaccine trials had given positive results:
In November, it was reported that multiple COVID vaccine trials had given positive results:
In addition, the results of a large Phase II clinical trial of ANAVEX®2-73 (also called blarcamesine – being developed by Anavex Life Science) were reported and they suggested that the treatment was well tolerated (in oral doses up to 50 mg once daily) and it showed clinically meaningful, dose-dependent, & statistically significant improvements in cognitive tests (Click here to read more about this). The study was a 14 week double-blind, multicenter, placebo-controlled trial involving 132 participants. It will be interesting to see the results of the 48 week extension study (ongoing – Click here to read more about this).
In November, a biorxiv manuscript was made available which presented an unbiased, automated, deep learning phenotypic profiling platform for analysing cells in culture. The investigators applied it to cells collected from 91 people (including 46 cases of Parkinson’s). The trained machine learning models captured individual variations, allowing for the accurate identification of cells from specific people (even across different experiments & different plate layouts). The models also confidently separated LRRK2 & idiopathic Parkinson’s cells from cells collected from control cases. There is serious potential for drug screening applications here (Click here to read more about this and click here to read an SoPD post on this topic).
In addition to that preprint, a medrxiv manuscript outlined a new platform: the Accelerating Medicines Partnership Parkinson’s (AMP PD) genetic resource – a solution to democratise data access/analysis for Parkinson’s research. Version 1 is now available and contains Whole Genome Sequencing data derived from 3,941 participants from 4 separate cohorts, including 2,005 idiopathic PD cases, 963 unaffected controls, 64 prodromal cases, 62 SWEDDs & 705 participants with GBA variants or LRRK2 variants, in which 304 were affected. An amazing resource and congrats to all the researchers who made it possible (Click here to read more about this and click here to see the website).
Oh, and while not specific to Parkinson’s, another achievement announced in November will have important implications for the condition: it was announced that the latest version of AlphaFold – an AI deep-learning program from Google’s DeepMind – made a gigantic step forward in solving the “protein folding problem”. More accurately determining the 3D shapes of proteins represents a transformational shift in biology. The accuracy of the software is measured on a range of 0-100. The latest version of AlphaFold has shifted the median score from the mid 60s in the last version to 92.4 across all targets (Click here to read more about this or watch the video below).
 In December, as the SpaceX Starship prototype SN8 made a first test flight…
In December, as the SpaceX Starship prototype SN8 made a first test flight…
…the results of the Phase II NILO-PD clinical study of nilotinib in individuals with moderately advanced Parkinson’s found that the treatment was acceptable safety & tolerability, but data “indicate that nilotinib should not be further tested in PD“. The researchers reported that “the low cerebrospinal fluid exposure & lack of biomarkers effect combined with the efficacy data trending in the negative direction indicate that nilotinib should not be further tested in Parkinson’s“. There was no difference in the clinical measures (change of MDS-UPDRS-3 OFF) from baseline to 6 months between the treatment groups. Cerebrospinal fluid/serum ratio of nilotinib concentration was 0.2% to 0.3% (Click here to read more about this).
In December the GDNF gene therapy company Brain Neurotherapy Bio also merged with Asklepios Bio Pharmaceutical to jointly advance central nervous system gene therapies, for conditions like Parkinson’s (Note: AskBio is a subsidary of the Pharma company Bayer – click here to read more about this and click here to read an SoPD post about this).
 And a research report was published in December in which it was found that activating the kinase Akt1 (with chlorogenic acid, which is found in coffee) prevents neurodegeneration in two mouse models of Parkinson’s. The scientists reported that Akt1 activates the CREB target RNF146 which subsequently inhibits PARP1-dependent cell death in both mouse & human neuronal cultures. “Furthermore, dysregulation of the Akt1-CREB pathway was observed in postmortem brain samples from patients with Parkinson’s” (Click here to read more about this and click here to read a SoPD post about this).
And a research report was published in December in which it was found that activating the kinase Akt1 (with chlorogenic acid, which is found in coffee) prevents neurodegeneration in two mouse models of Parkinson’s. The scientists reported that Akt1 activates the CREB target RNF146 which subsequently inhibits PARP1-dependent cell death in both mouse & human neuronal cultures. “Furthermore, dysregulation of the Akt1-CREB pathway was observed in postmortem brain samples from patients with Parkinson’s” (Click here to read more about this and click here to read a SoPD post about this).
# # # # # # #
These were some of the main events for Parkinson’s research in 2020 (according to yours truly). In the next post, we will look at where things are heading in 2021 in our annual “Road ahead” post.
 All of the material on this website is licensed under a
All of the material on this website is licensed under a
Creative Commons Attribution 4.0 International License
You can do whatever you like with it!
EDITOR’S NOTE – The author of this post is an employee of the Cure Parkinson’s Trust. The Trust is supporting many of the clinical trials mentioned in this post (including the Exenatide study, the Ambroxol study, the Australian Parkinson’s Mission, and the Linked Clinical Trials initiative). The Trust has not requested the production of this post, but the author considered the information interesting and important to share with the Parkinson’s community.
Many of the companies mentioned in this post are publicly traded companies. That said, the material presented on this page should under no circumstances be considered financial advice. Any actions taken by the reader based on reading this material is the sole responsibility of the reader. None of the companies have requested that this material be produced, nor has the author had any contact with any of the companies or associated parties. This post has been produced for educational purposes only.
In addition, the information provided by the SoPD website is for educational purposes only. Under no circumstances should it ever be considered medical or actionable advice. It is provided by research scientists, not medical practitioners. Any actions taken – based on what has been read on the website – are the sole responsibility of the reader. Any actions being contemplated by readers should firstly be discussed with a qualified healthcare professional who is aware of your medical history. While some of the information discussed in this post may cause concern, please speak with your medical physician before attempting any change in an existing treatment regime.
The banner for today’s post was sourced from Special events









































In related trial news, inhibikase went public raising 18 million dollars. That would indicate to me that a trial initiation is right around the corner
LikeLike
Highlight for me was reading “Brain Fables ” by Alberto Espay. As a result I have more confidence in shifting the focus of my DIY cocktail for my idiopathic PD away from mitos and oxidative stress and towards inflammation. Most intriguing article reviewed the similarities between a-syn, other amyloids and innate immunity mediators such as cathelicidin:https://www.frontiersin.org/articles/10.3389/fimmu.2020.01629/full Innate immunity promoted by “vitamin”D is important in reducing severity of Covid-19 as this Open Letter summarises: https://vitamindforall.org/letter.html
LikeLike
What an amazing amount of information you have shared this year. I am in awe of your ability to collate so much useful information and present it in such a user friendly way. Your blog is my ‘go to’ for information on all things scientific in relation to Parkinson’s. Keep up the good work and thank you! Alison
LikeLike